* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Studies on the Phosphorylation of the 58000 Dalton Early Region
Endogenous retrovirus wikipedia , lookup
Biochemistry wikipedia , lookup
Cryobiology wikipedia , lookup
Western blot wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Lipid signaling wikipedia , lookup
Biochemical cascade wikipedia , lookup
Paracrine signalling wikipedia , lookup
Two-hybrid screening wikipedia , lookup
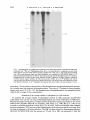
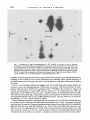
J. gen. Virol. (1983), 64, 1069-1078. Printed in Great Britain 1069 Key words: Ad5/early protein/phosphorylation pattern Studies on the Phosphorylation of the 58000 Dalton Early Region 1B Protein of Human Adenovirus Type 5 By P A U L M A L E T T E , S I U - P O K Y E E AND P H I L I P E. B R A N T O N * Department o f Pathology, M c M a s t e r University, Hamilton, Ontario, Canada (Accepted 20 December 1982) SUMMARY The 58000 dalton (58K) protein coded for by early region 1B of human adenovirus type 5 (Ad5) was found to be phosphorylated. At least three major tryptic phosphopeptides were identified and the average number of phosphates per 58K molecule was estimated to be between two and three. Thus, it was possible that each phosphopeptide contained just one phosphate group. The ratio of phosphoserine to phosphothreonine was about 2 to 1 on average and essentially no phosphotyrosine was detected. No evidence was found to suggest that cAMP-dependent protein kinase was involved in the phosphorylation of 58K. Previous studies have shown that 58K was phosphorylated when immunoprecipitates containing Ad5 early region 1 proteins were incubated in vitro with [~-32p]ATP. Analysis of the phosphopeptides of 58K labelled under these conditions indicated a large number of phosphorylation sites which differed from those found in vivo. Thus, the action of kinases in the in vitro phosphorylation of 58K in immunoprecipitates did not mimic the enzymic activity responsible for 58K phosphorylation in vivo. INTRODUCTION Transformation of cells by human adenovirus type 5 (Ad5) involves the expression of only about 11 ~ of the viral DNA, located at the extreme left end of the genome (Graham et al., 1974; Gallimore et aL, 1974). Complementation studies with host range (hr) mutants have shown that this portion of the viral genome is composed of two early transcriptional units (Harrison et, al., 1977; Graham et al., 1978) which have been designated E1A (1.5 to 4.5 map units) and E1B (5.5 to 11 map units) (Flint, 1977; Berk & Sharp, 1978; Chow et al., 1979; Galos et al., 1979). The E1B region produces three mRNA species of 2.2, l-0 and 0.5 kilobases (kb), of which the smallest codes for the late Ad5 polypeptide IX (Alestrom et al., 1980). Recently, it has been shown that the large 2.2 kb mRNA species codes for two polypeptides with predicted molecular weights of about 55 000 and 19 000 (Bos et al., 1981). The 1-0 kb mRNA species appears to code for a 19000 dalton (19K) protein believed to be identical to the 19K protein from the 2.2 ~:b mRNA (Bos et al., 1981). A 58 000 dalton protein (termed 58K) has been detected using anti-tumour sera in both Ad5infected and -transformed cells (Levinson & Levine, 1977 a, b; Johansson et al., 1978; Ross e t at., 1978; Lassam et al., 1979a, b) and it is certain that this protein is the product of the 2.2 kb E1B mRNA. It is produced in vitro by translation of E 1B-selected mRNA (Halbert et al., 1979; Lewis et al., 1979; van der Eb et aL, 1979). Cells transformed by the XhoI C fragment (0 to 15.6 map units) of Ad5 D NA express 58K (Schrier et aL, 1979; D. T. Rowe & F. L. Graham, unpublished results). All of the hr mutants that map in EIB (group II mutants) are defective in 58K synthesis (Lassam et al., 1979a, b). And now we have shown that antiserum raised against a synthetic peptide corresponding to the predicted carboxy terminus of the large product of the 2-2 kb E 1B mRNA immunoprecipitates 58K (S.-P. Yee, D. T. Rowe, M. L. Tremblay, M. McDermott & P. E. Branton, unpublished results). Neither the biological role nor biochemical function of 58K is clear at present. It appears to play some role in transformation, as Ad5 group II hr mutants fail to generate transformed foci Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 0022-1317/83/0000-5455$02.00 0 1983 SGM 1070 P. MALETTE, S . - P . YEE AND P. E. BRANTON (Graham et al., 1978), yet the nature of this role remains obscure because transfection with D N A isolated from these same mutants produces transformed cells with a broad range of oncogenic potential (D. T. Rowe & F. L. Graham, unpublished results). We have also found that E1A protein synthesis (but not E1A transcription) is shut down during lytic infection when 58K synthesis commences and thus it could regulate translation (D. T. Rowe, P. E. Branton & F. L. Graham, unpublished results). In addition, immunoprecipitates containing 58K possess protein kinase activity. Incubation of such immunoprecipitates with [~-32p]ATP results in the phosphorylation of certain exogenous substrates and a protein which co-migrates with 58K on SDS-polyacrylamide gels (Lassam et al., 1979c; Branton et al., 1979, 1981). This protein kinase activity is produced in infected cells with the same kinetics as 58K (D. T. Rowe, F. L. Graham & P. E. Branton, unpublished results) and it is deficient in cells infected with group II hr mutants (Lassam et al., 1979c; Branton et al., 1979). At present, it is still not clear whether this protein kinase activity is intrinsic to 58K, or is due to a protein which is associated with 58K. Previous reports have suggested that 58K is a phosphoprotein (Ross et al., 1978). We have begun to characterize the phosphorylation of 58K, because the biological activities of a wide variety of proteins are known to be regulated by phosphorylation (Rubin & Rosen, 1975; Krebs & Beavo, 1979). The protein was found to possess, on average, two to three phosphates located at a minimum of three sites on serine and threonine residues at a ratio of about two to one. METHODS Cell culture and virus infection. Human KB cells were generally grown at 37 °C in 150 x 15 mm plastic Petri dishes (Lux Scientific) using alpha minimal essential medium (~-MEM) supplemented with 4 ~ foetal calf serum and 1~ L-glutamine (Gibco). For some experiments, suspension cultures of KB cells were grown in c~-MEM containing 8 % calf serum at 37 °C in Belco spinner bottles. Cells were infected with wild-type Ad5 at 35 p.f.u./cell. Serum. Immunoprecipitation of 58K was carried out using 14B antiserum which was obtained from hamsters bearing tumours induced by the Ad5-transformed 14B hamster cell line (Sambrook et al., 1974). Cell extracts and immunoprecipitation. Ad5-infected and mock-infected KB cells were washed with phosphatebuffered saline (PBS) and were scraped into cold PBS. The cells were collected by low-speed centrifugation and were washed twice more with cold PBS. For cytoplasmic extracts, the washed cell pellet was resuspended in 200 mi-Tris HCl pH 7.0 containing 137 mM-NaCI, 1 mi-CaC12, 0-4 mi-MgC12, 10~ (v/v) glycerol and 1% (v/v) Nonidet P40 at a concentration of about 2 x 107 cells/ml and the mixture was incubated on ice for 20 rain. Nuclei and unlysed cells were removed by centrifugation at 12500 g for 20 min at 4 °C and the supernatant was dispensed into 1 ml aliquots. For whole cell extracts, the washed cell pellet was resuspended at 2 x 107 cells/ml in RIPA buffer which consisted of 0-05 M-Tris-HCI pH 7-2, containing 150 mM-NaC1, 0.1% (v/v) SDS, 1~ (v/v) sodium deoxycholate, 1~ (v/v) Triton X-100 and 2000 IU/ml aprotinin. The mixture was incubated for 20 min on ice, it was sonicated for 1-5 min at maximum setting on a Biosonik IV probe sonicator (VWR Scientific) and then centrifuged at 12500 g for 20 rain at 4 °C. The supernatant was removed and dispensed into 1 ml aliquots. For immunoprecipitation, 1 ml aliquots of whole cell or cytoplasmic extracts were combined with 10 ~tl of 14B antiserum and 250 ~il of Protein A-Sepharose beads (Pharmacia) suspended (1 : 10, v/v) in the appropriate lysing buffer and the mixture was incubated for 3 h at 4 °C with constant mixing. The Sepharose beads with bound antibody were collected by centrifugation at 30g for 30 s and then washed three times with 1 m1100 mM-Tris-HCl pH 7.0 containing 200 mM-LiCl and 0-1% (v/v) 2-mercaptoethanol. SDS polyacrylamide gel eleclrophoresis (SDS-PAGE). Proteins were separated by SDS-PAGE using a system based on that originally described by Laemmli (1970). The running gels consisted of 10 or 12 ~ polyacrylamide, or were a gradient of 7-5 to 15 ~ polyacrylamide with a 5 % polyacrylamide stacking gel. Unless otherwise stated, gels were run either at a constant current of 35 mA or a constant voltage of 90 V. They were stained with Coomassie Brilliant Blue and destained with isopropanol-acetic acid prior to drying under vacuum. Autoradiography was carried out using Kodak X-Omat XRP-I or XAR-5 film. Radioactive labelling. For routine labelling with 32p, cells were incubated for 2 to 3 h in phosphate-free medium (no serum) containing [32p]orthophosphate (New England Nuclear; carrier-free in H20) at 1 mCi/plate in 4 ml of medium. For the purpose of phosphoamino acid analysis or tryptic peptide mapping, Ad5-infected cells (usually six plates) were incubated with 4 mCi [32p]orthophosphate for 5 or 6 h. In radiochemical studies designed to determine the molar ratio of phosphate groups in 58K, infected cultures were incubated from immediately after the adsorption period (0.5 h) to 16 h post-infection in 8 ml of complete ~-MEM containing 4% dialysed calf serum in the presence of [32p]orthophosphate at a known specific activity of 0.2402 Ci/mmol, and either [3H]valine or [3H]leucine (New England Nuclear) was also present at a final specific activity of 0-1208 or 0-1186 Ci/mmol respectively. After immunoprecipitation and SDS-PAGE, the band corresponding to 58K was located by autoradiography and excised, the labelled protein was eluted (see below) and the amount of 3H and 32p present Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 Phosphorylation o f Ad5 5 8 K 1071 was determined taking into account the counting efficiency and the quench correction for each isotope. The 32P/3H ratio was used to calculate the number of phosphates per 58K molecule using the number of valine (39) or leucine (37) residues predicted from the corresponding nucleotide sequence ofAd5 (Maat & van Ormondt, 1979; Maat et al., 1980; Bos et al., 1981). In vitro phosphorylation using immunoprecipitates. Cytoplasmic extracts were immunoprecipitated using 14B antiserum as described above, lmmunoprecipitates were washed and then incubated with [y-3zp]ATP exactly as previously described (Branton et al., 1981). Phosphoamino acid determination. The area on SDS polyacrylamide gels containing the 58K band was excised, the gel piece was diced into small pieces and the protein was eluted for 24 h at 37 °C with constant shaking into a small volume of 0.05 M-ammonium bicarbonate containing 0.1% SDS. The gel fragments were then incubated for another 16 h in fresh solution. The combined solutions were precipitated with 10% trichloroacetic acid (TCA) in the presence of 200 ~tg bovine serum albumin (BSA) carrier. The precipitate was removed by centrifugation (1000O g for 10 min) and it was washed twice with 95 % acetone. The 32P_labelled 58K was hydrolysed in 5 M-HCI for 2 h at 110 °C under nitrogen. The hydrolysate was lyophilized and dissolved in a small volume of water (25 lal) and then combined with non-radioactive marker phosphoserine, phosphothreonine (Sigma) and phosphotyrosine (prepared according to the method of Mitchell & Lunan, 1964). Phosphoamino acids were separated by twodimensional electrophoresis on cellulose thin-layer plates (20 × 20 cm Polygram CEL300, 0.1 mm thick; Brinkman) by the method of Hunter & Sefton (1980) exactly as described previously (Branton et al., 1981). The positions of the phosphoamino acid markers were identified by ninhydrin staining and those of the labelled amino acids by autoradiography. The corresponding spots were removed by scraping and the material eluted with 1 MHCI and counted in a liquid scintillation counter. Tryptic peptide mapping. 32p-labelled 58K was immunoprecipitated and separated by S D S - P A G E and the corresponding area on dried, fixed gels was excised and diced into small pieces. The fragments were incubated at 37 °C for 24 h in 3 to 4 ml of 0.05 M-ammonium bicarbonate containing 50 ~tg/ml tolylsulphonyl phenylalanyl chloromethyl ketone-trypsin. The supernatant was removed and replaced with 2 to 3 ml of fresh trypsin and incubation was continued for 12 to 16 h. The supernatants were pooled, they were oxidized on ice with fresh performic acid, and then they were combined with 5 vol. H20 and lyophilized. The peptides were dissolved in about 40 ~tl of 10~o formic acid, spotted (usually 1000 to 4000 ct/min) onto cellulose thinqayer plates (Polygram CEL300) and separated by the method of Walter & co-workers (Walter & Flory, 1979; Scheidtmann et al., 1981). Electrophoresis was carried out in the first dimension for 50 rain at 1000 V using a buffer consisting of formic acidacetic acid-pyridine-water (6:1.25:0.25:92.5), pH 1-9. The plates were rotated 90° and a second dimension of separation was performed by ascending chromatography in n-butanol-pyridine-acetic acid-water (50:40 : 10:40). The positions of 32p-labelled phosphopeptides were determined by autoradiography. Analysis oftryptic peptides by ion-exchange chromatography. Tryptic peptides of 32p-labelled 58K were prepared as described above except that, after performic acid oxidation, the sample was combined with 2 mg of peptide carrier. The latter was prepared using BSA and V-8 protease (Miles Laboratories). The mixture was lyophilized and the sample was dissolved in 250 ~tl of 0.05 M-pyridine acetate buffer (pyridine acetic acid-water; 1 : 17.4:231.6, pH 3.2) containing 10~ formic acid. The sample was applied using an injection valve (Beckman) to a 9 x 250 mm glass column (Beckman) containing packed Aminex A5 cation exchange resin in the sodium form (Bio-Rad) which had been pre-equilibrated with pyridine acetate buffer. The peptides were eluted using a 300 ml gradient of 0.05 M(pH 3.2) to 1 M (pH 4.9) pyridine acetate under 200 to 400 lbf/in 2 with a flow rate of 0.5 ml/min and 2 ml fractions were collected. The column was washed with 2 M-pyridine acetate pH 5.0. Fractions were assayed for pH and pyridine concentration and then were dried in an oven at 100 °C. Radioactivity was measured using ACS counting fluid (New England Nuclear). RESULTS The Ad5 E1B 58K antigen is a phosphoprotein As shown in Fig. 1 (e), the E1B 58000 dalton protein was readily immunoprecipitated from Ad5-infected cells labelled with [3SS]methionine, using serum from hamsters bearing tumours induced by Ad5-transformed 14B hamster cells. Previous studies had suggested that 58K was phosphorylated (Ross et aL, 1978) and, as shown in Fig. 1 (c), a 32p-labelled species which comigrated with 3sS-labelled 58K was also precipitated from Ad5-infected cells by 14B serum. No 58K was detected in mock-infected cells (Fig. 1 a, b) or with non-immune serum (Fig. 1 d, f). Identification of the phosphoamino acids in 58K The phosphoamino acids present in 58K were determined by isolating 32p-labelled 58K from gels and subjecting this material to acid hydrolysis and two-dimensional electrophoresis. Fig. 2 Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 1072 P. MALETTE, S.-P. YEE AND P. E. BRANTON (a) (b) (e) (d) (e) (f) 92-5-66.2-- 58K 45-- 31-- 21.5-- Fig. 1. Autoradiogram of an SDS-polyacrylamide gel of immunoprecipitate. Ad5-infected cells were incubated with 3 2 p o 4 o r [35Slmethioninefrom 10 to 12 h post-infection. Cytoplasmic extracts from these cultures and from mock-infected cultures labelled with 32PO4 for 2 h were immunoprecipitated with 14B or non-immune serum and the precipitates were analysed by SDS-PAGE using a 12% polyacrylamidegel. At the left are shown the positions of migration of the followingtool. wt. markers: phosphorylase B (92500), bovine serum albumin (66200), ovalbumin (45000), carbonic anhydrase (31000), soybean trypsin inhibitor (21500) and lysozyme (14400). (a to d) 32P-labelledcells: mockinfected cells with 14B serum (a) and non-immune serum (b), Ad5-infected cells with 14B serum (c) and non-immune serum (d). (e, f) 3sS-labelledAd5-infected cells with 14B (e) and non-immune (f) sera. shows that 32p was present in the position of the phosphoserine and phosphothreonine markers, but virtually none was detected in phosphotyrosine. The ratio of 32p present in these phosphoamino acids was 2.35:1.00 : <0-1 for phosphoserine, phosphothreonine and phosphotyrosine respectively (average of four experiments). Estimation of the average number of phosphates per 58K molecule To estimate the average n u m b e r of phosphates present on 58K molecules, double-label experiments were carried out using [3:p]orthophosphate and either [3H]valine or [3H]leucine. Because the amino acid sequence of 58K had been deduced by sequencing studies on the 2-2 kb m R N A and E1B D N A (Maat & van Ormondt, 1979; Maat et al., 1980; Bos et al., 1981) it was possible to establish phosphate content from 32p/3H ratios. Cells were labelled with complete medium containing 32po 4 and one of the labelled amino acids at k n o w n specific activities, from the time of infection to 16 h post-infection. The 58K was then isolated by immunoprecipitation, separated by S D S - P A G E and the amount of 32p and 3H in 58K was measured. U n d e r the Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 Phosphorylation of Ad5 58K 1073 Fig. 2. Phosphoamino acids Of 58K. Ad5-infected cells were labelled with 3 2 p o 4 from 10 to 16 h postinfection and the phosphoamino acids present in 58K were analysed as described in Methods. The positions of the labelled species after two-dimensional separation were determined by autoradiography and those of the phosphoserine, phosphothreonine and phosphotyrosine markers by ninhydrin staining (see hatched lines). The radioactivity present in these three phosphoamino acids for the experiment shown was 2741, 1429 and 29 ct/min respectively, a ratio of 1-9 : 1.0:0-02. Over the course of four such experiments the average ratio was 2.35(+0.6):1-00:<0.1 respectively. Table 1. Estimate of the number of phosphate groups in 58K Labelling conditions (number of expts.) [3H]Leucine/32po4 double label (4) [3H]Valine/32po4 double label (3) Total double label (7) Number of phosphates per 58K molecule ( + standard error)* 2-7 + 0.7 2-6 _+ 0.8 2-7 + 0-8 * The number of phosphates on 58K was estimated as described in the text. present conditions, synthesis of 58K does not commence until after 4 h post-infection and is not m a x i m a l until 8 to 12 h postqnfection (P. Malette & P. E. Branton, unpublished data) and thus both phosphate and amino acid pools should have equilibrated by the time most of the 58K was synthesized. Because the endogenous pools of phosphate and amino acids were small relative to the large excess present in the medium, the specific activities of cellular pools at equilibrium should have been those present in the medium. Table 1 shows that, using this approach, the values obtained with [3H]leucine and [aH]valine were 2.7 and 2.6 respectively. Thus, in lytically infected cells, 58K molecules contain, on average, two to three phosphate groups. Analysis of tryptic phosphopeptides To examine further the number of phosphorylation sites, 32p-labelled 58K was isolated from preparative polyacrylamide gels and exhaustively digested with trypsin. As shown in Fig. 3 (a), following two-dimensional separation on cellulose thin-layer plates, four 32p-labelled species were resolved. One of these migrated towards the cathode (peptide 1), a second stayed near the origin (peptide 2), a third migrated slightly towards the anode (peptide 3) and a fourth migrated significantly towards the anode (peptide 4). On occasion, there were a few other minor species as Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 P. MALETTE, S.-P. YEE AND P. E. BRANTON 1074 C m C " O'~e J Fig. 3. Tryptic phosphopeptides of 58K. Ad5-infected cells were incubated with 3 2 p o 4 from 12 to 16 h post-infection and this labelled 58K was separated by immunoprecipitation and SDS-PAGE. The band containing 58K was excised, digested with trypsin and analysed by two-dimensional separation on cellulose thin-layer plates involving electrophoresis at pH 1.9 followed by chromatography in the second dimension, as described in Methods. In addition, some pure 32po 4 was also spotted onto the same thin-layer plate such that the 58K and 3 2 p o 4 profiles were mirror images of each other. (a) 32p_ labelled 58K; (b) 32PO4. well, and in some cases the patterns for peptides 2 and 3 suggested multiple spots (see below, a n d Fig. 5a). The fourth species was probably inorganic PO 4 because pure 3 2 p o 4 h a d similar migration properties (Fig. 3 b). Peptide 1 was usually labelled at lower levels t h a n peptides 2 and 3. Tryptic phosphopeptides were also separated by ion-exchange c h r o m a t o g r a p h y on A m i n e x columns. As shown in Fig. 4, four major 32p.labelled species were also resolved by this method. Peak A which was present in the void volume was undoubtedly inorganic phosphate, as 3 2 p o 4 run alone had similar properties (data not shown). Three other major species, peaks B, C and D, eluted at about 0.3, 0-4 and 0.6 M-pyridine acetate respectively. Peak C m a y have contained more than one species, because a slight shoulder was detected. Thus, at least three major tryptic phosphopeptides were resolved by these two methods. Phosphorylation o f 58K in immunoprecipitates Incubation of immunoprecipitates containing 58K with [~-3Zp]ATP in vitro has been found to result in the phosphorylation of a protein which co-migrated with 58K (Branton et al., 1981). In order to determine whether such phosphorylation reflected that observed in vivo, the tryptic phosphopeptides of 58K labelled in vivo and in vitro in immunoprecipitates were compared. As Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 1075 Phosphorylation of Ad5 58K I I 14! ! t t IC 1 ! I t 1 I l|l 1 1 12 .0 × 10 / f f 5.0 4.5 L 6 :4.0 ~& ~ 4 3'5 2 j t-i ...........l, ....... I-~ 20 40 60 80 Gradient Fraction number 100 I'*" 2M wash Fig. 4. Separation of the tryptic phosphopeptides of 58K by ion-exchange chromatography. 32p. labelled 58K was isolated by immunoprecipitation and SDS-PAGE and the labelled protein was eluted from the gel and precipitated with TCA. This material was applied to an Aminex ion-exchange column and chromatographed, as described in Methods. O, 32p-labelled material; - - - , pyridine acetate concentration; II, pH. shown in Fig. 5, there was no relation between the two profiles. None of the three major phosphopeptides found in vivo (Fig. 5 a) were labelled with the immunoprecipitates (Fig. 5 b); however, a larger number of other 32p.labelled species were observed. Thus, the phosphorylation of 58K in vitro using immunoprecipitates took place at sites other than those normally found in vivo. DISCUSSION These studies have clearly demonstrated that the Ad5 E 1B 58K antigen is a phosphoprotein. Tryptic peptide analysis showed that 58K contained at least three major phosphopeptides. At pH 1.9, one of the peptides (no. 1) was positively charged, one was neutral (no. 2), and one was negatively charged (no. 3). It should be pointed out that in some two-dimensional analyses, there was a suggestion that multiple species were present in peptides 2 and 3 (e.g. see Fig. 5a). However, over the course of several experiments these peptides virtually always resolved as single spots and it may be that differences in migration may have resulted from differences in oxidation prior to analysis. Analysis of the tryptic peptides by ion-exchange chromatography also suggested three major phosphopeptides (see Fig. 4), although a slight shoulder was present on one of the peptide peaks (C). Estimates of the average number of phosphates on 58K molecules gave values of about 2 to 3 and thus it is possible that each of the major tryptic phosphopeptides contained only one phosphate group. These values depended on equilibrium of the phosphate and amino acid pools in the 8 to 12 h prior to maximal 58K synthesis and thus they could have been somewhat underestimated. In most experiments less 32p was found in peptide 1 than in peptides 2 and 3 and thus the site present on peptide l may have been phosphorylated less frequently or may have been more susceptible to dephosphorylation by phosphoprotein phosphatases than the other sites. Phosphoserine and phosphothreonine were found in 58K at a ratio of about 2 to 1, and no phosphotyrosine was detected. All of these data are consistent with the presence of three phosphorylation sites on 58K, two of which are serine residues and one threonine. However, the number of sites could have been underestimated if multiple phosphopeptides tended to co-migrate with both separation techniques used, or if the phosphoDownloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 1076 P. MALETTE, S.-P. YEE AND P. E. BRANTON (a) Qo ,,..C It Fig. 5. Comparison of tryptic phosphopeptides of 58K labelled in vivo and in vitro in immunoprecipitates. 32P-labelled 58K was prepared from Ad5-infected cells incubated with 32po 4 from 12 to 16 h post-infection as described in Methods. Cytoplasmic extracts were also prepared from Ad5infected cells harvested at 12 h post-infection and these were immunoprecipitated with 14B serum. The precipitates were incubated with [2:-32p]ATPas described in Methods and 58K was separated by SDSPAGE. Labelled 58K was digested with trypsin and separated on cellulose thin-layer plates as in Fig. 5. (a) 58K labelled in vivo; (b) 58K labelled in immunoprecipitates in uitro. peptides contained multiple sites but that most o f these sites in general were not phosphorylated, resulting in an average of two to three phosphates per molecule. More precise m a p p i n g o f phosphorylation sites will require the use of various proteases and other techniques to resolve this issue. W e have also obtained evidence that suggests that cyclic A M P - d e p e n d e n t protein kinases do not play a role in the phosphorylation of 58K (data not shown). Cell extracts devoid of endogenous A T P and cyclic A M P were incubated with [3,-3Ep]ATP i n i h e presence or absence of various concentrations of cyclic A M P and the amount of 32p incorporated into 58K was determined. Cyclic A M P was found to have virtually no effect on the phosphorylation of 58K. This result was not surprising, as the proposed a m i n o acid sequence of 58K does not contain the usual phosphorylation sites of cyclic A M P - d e p e n d e n t kinase, Arg-Arg-X-Ser or Lys-Arg-X-XSer ( K e m p et al., 1977; K r e b s & Beavo, 1979). W h e n immunoprecipitates containing 58K are incubated with [y-32p]ATP, phosphorylation of a protein which co-migrates with 58K takes place (Branton et al., 1981). The p r e s e n t Study has indicated that the pattern of tryptic phosphopeptides obtained from this material is completely different from that seen with 58K labelled in vivo. Thus, either this 58000 dalton protein is not 58K or such in vitro phosphorylation of 58K occurs at sites other than those found in vivo. The latter could be caused by action of a protein kinase present in the i m m u n o p r e c i p i t a t e which normally does not use 58K as a substrate, or simply by artefacts induced by the in vitro system. Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 Phosphorylation of Ad5 58K 1077 Phosphorylation could regulate the action of 58K, either by controlling the activity of a single biological function or by providing a multifunctional potential. Little is known about the biological function(s) of 58K. It appears to be located both in the nucleus and the cytoplasm (D. T. Rowe, F. L. Graham & P. E. Branton, unpublished results), and thus phosphorylation could affect its intracellular location by altering its interaction with other macromolecules. We have also found that some 58K is present in large complexes in transformed cells (S.-P. Yee & P. E. Branton, unpublished results). Whether this complex represents polymers of 58K or interactions with other proteins, as has already been suggested (Sarnow et al., 1982), is still not known, but phosphorylation could play a role here as well. It is hoped that future studies will pinpoint more precisely the location of phosphate groups on 58K and their precise role in the regulation of 58K function. We are indebted to Gernot Walter for m a n y helpful discussions. We wish to t h a n k Sylvia Grinbergs, Aileen Hogan, Joceline Otis and Joe Luckshis for their expert help with these experiments. This work was supported through grants from the Medical Research Council of C a n a d a and the National Cancer Institute of Canada. P.M. had an M R C Studentship. P.E.B. is an N C I C Research Associate. REFERENCES ALESTROM,P., AKUSJARVI,G., PERRICAUDET,M., MATHEWS,M., KLESSIG, D. & PETTERSSON, U. (1980). T h e gene for polypeptide IX of adenovirus type 2 and its unspliced messenger R N A . Cell 19, 671-681. BERK, A. J. & SHARp, P. A. (1978). Structure of the adenovirus 2 early m R N A s . Cell 14, 695-711. Bos, J. L., POLDER, L. J., BERNARD,R., SCHRIER, P. I., VAN DEN ELSEN, P. J., VAN DER EB, A. J. & VAN ORMONDT, H. (1981). The 2.2 K b m R N A of the E 1B region of h u m a n adenovirus type 5 and 12 directs the synthesis o f two major antigens from different A U G triplets. Cell 27, 121-131. BRANTON,P. E., LASSAM,N. J., GRAHAM,F. L., MAK,S. & BAYLEY,S. T. (1979). T-antigen-related protein kinase activity in cells infected and transformed by h u m a n adenovirus 5 and t2. Cold Spring Harbor Symposia on Quantitative Biology 44, 487-491. BRANTON,P. E., LASSAM,N. J., DOWNEY,J. F., YEE, S.-P., GRAHAM,F. L., MAK,S. & BAYLEY,S. T. (t981). Protein kinase activity immunoprecipitated from adenovirus-infected cells by sera from tumor-bearing hamsters. Journal of Virology 37, 601-608. CHOW, L. T., BROKER, T. R. & LEWIS, J. B. (1979). Complex splicing patterns of R N A s from the early regions of adenovirus 2. Journal of Molecular Biology 141, 249-265. FLINT, S. J. (1977). Two species of adenovirus early m R N A in transformed cells. Journal of Virology 23, 44-52. GALLIMORE,P. H., SHARP, P. A. & SAMBROOK,J. (1974). Viral D N A in transformed cells. II. A study of the sequences of Ad2 D N A in nine lines of transformed rat cells using specific fragments of the viral genome. Journal of Molecular Biology 89, 49-72. GALOS, R. S., WILLIAMS, J., BINDER, M. & FLINT, S. J. (1979). Location of additional early gene sequences in the adenoviral chromosome. Cell 17, 945-956. GRAHAM, F. L., VAN DER EB, A. J. & HEIJNEKER, H. L. (1974). Size a n d location of the transforming region in h u m a n adenovirus D N A . Nature, London 251, 687-691. GRAHAM, F. L., HARRISON, T. & WILLIAMS,J. (1978). Defective transforming capacity of adenovirus 5 host range mutants. Virology 86, 10-21. HALBERT, D. N., SPECTOR, O. J. & RASKAS,H. J. (1979). In vitro translation products specified by the transforming region of adenovirus type 2. Journal of Virology 31, 621-629. HARRISON,T., GRAHAM,F. L. & WILLIAMS,J. (1977). Host-range m u t a n t s of adenovirus type 5 defective for growth in HeLa cells. Virology 77, 319-329. HUNTER, T. & SEFTON, B. M. (1980). Transforming gene product of Rous sarcoma virus phosphorylates tyrosine. Proceedings of the National Academy of Sciences, U.S.A. 77, 1311-1315. JOHANSSON,K., PERSSON,H., LEWIS, A. M., PETTERSON,U., TIBBETTS,C. & PHILIPSON, L. (1978). Viral D N A sequences and gene products in hamster cells transformed by Ad2. Journal of Virology 27, 628-639. KEMP, B. E., GROVES, D. J., BENJAMINI,E. & KREBS, E. G. (1977). Role of multiple basic residues in determining the substrate specificity of cyclic A M P - d e p e n d e n t protein kinase. Journal of Biological Chemistry 252, 4888-4894. KREBS, E. G. & BEAVO,J. A. (1979). Phosphorylation-dephosphorylation of enzymes. Annual Review of Biochemistry 48, 923-959. LAEMMLI, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, London 277, 680-685. LASSAM,N. l., BAYLEY,S. T. & GRAHAM,F. L. (1979a). Transforming proteins of h u m a n adenovirus type 5: studies with infected and transformed cells. CoM Spring Harbor Symposia on Quantitative Biology 44, 477-49l. LASSAM,N. L, BAYLEY,S. T. & GRAHAM,F. L. (1979b). T u m o r antigens of h u m a n Ad5 in transformed cells and in cells infected with transformation-defective host-range mutants. Cell 18, 781-791. LASSAM,N. J,, BAYLEY, S. T., GRAHAM, F. L. & BRANTON,P. E. (1979C). Immunoprecipitation of protein kinase activity from adenovirus 5-infected cells using antiserum directed against tumor antigens. Nature, London 277, 241243. Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43 1078 P. M A L E T T E , S . - P . YEE AND P. E. B R A N T O N LEVINSON, A. & LEVINE,A. J. (1977a). The isolation and identification of the adenovirus group C tumor antigens. Virology 76, 1-11. LEVINSON, A. & LEVINE, A. J. (1977b). The group C adenovirus tumor antigens: identification in infected and transformed cells and a peptide map analysis. Cell 11, 871-879. LEWIS, J. B., ESCHE,H. E., SMART, J. E., STILLMAN,B., HARTER, M. L. & MATHEWS,M. B. (1979). The organization and expression of the left third of the adenovirus genome. Cold Spring Harbor Symposia on Quantitative Biology 44, 493-500. MAAT, J. & VAN ORMONDT, H. (1979). The nucleotide sequence of the transforming Hind I I I - G fragment of adenovirus type 5 DNA. The region between map positions 4.5 (Hpa I site) and 8.0 (Hind I I I site). Gene 6, 7590 MAAT,J., VANBEVEREN,C. P. & VANORMONDT,H. (1980). The nucleotide sequence of adenovirus type 5 early region E l : the region between map positions 8.0 (Hindlll site) and 11.8 (SmaI site). Gene 10, 27-38. MITCHELL,H. K. & LUNAN,K. D. (1964). Tyrosine-O-phosphate in Drosophila. Archives of Biochemistry and Biophysics 106, 219-222. ROSS,S. R., LINZER, D. I. H., FLINT,S. J. & LEVINE,A. J. (1978). The adenovirus and SV40 tumor antigens detected in adenovirus and adenovirus-SV40 hybrid virus transformed cells. In 1CN-UCLA Symposium on Persistent Viruses, vol. 11, p. 469. Edited by J. Stevens, G. Todaro & C. F. Fox. New York: Academic Press. RUmN, C. S. & ROSEN, O. M. (1975). Protein phosphorylation. Annual Review of Biochemistry 44, 831-887. SAMBROOK, J., BOTCHAN,M., GALLIMORE,P., OZANNE, B., PE'I'I'ERSSON,U., WILLIAMS,J. & SHARP. P. A. (1974). Viral D N A sequences in cells transformed by simian virus 40, adenovirus type 12 and adenovirus type 5. Cold Spring Harbor Symposia on Quantitative Biology 39, 615~32. SARNOW, P., HO, Y. S., WILLIAMS,J. & LEVINE, A. J. (1982). Adenovirus Elb-58Kd tumor antigen and SV40 large tumor antigen are physically associated with the same 54Kd cellular protein in transformed cells. Cell 28, 1387 1394. SCHEIDTMANN,K.-H., KAISER,A., CARBONE,A. & WALTER,G. (1981 ). Phosphorylation of threonine in the proline-rich carboxy-terminal region of simian virus 40 large T antigen. Journal o.t Virology 38, 59-69. SCHRIER, P. I., VANDEN ELSEN,P. J., HERTOGLIS,J. J. L. & VANDER EB, A. J. (1979). Characterization of tumor antigens in cells transformed by fragments of adenovirus type 5 DNA. Virology 99, 372-385. VANDER EB, A. J., VANORMONDT,H., SCHRIER,P. I., LUPKER, J. H., JOCHEMSEN,H., VANDEN ELSEN, P. J., DE LEYS,R. J., MAAT, J., VAN BEVEREN,C. P., DIJKEMA,R. & DE WAARD, A. (1979). Structure and function of the transforming genes of human adenoviruses and SV40. Cold Spring Harbor Symposia on Quantitative Biology 44, 383-399. WALTER, G. & FLORY, P. J., JR (1979). Phosphorylation of SV40 large antigen. Cold Spring Harbor Symposia on Quantitative Biology 44, 165-169. (Received 6 October 1982) Downloaded from www.microbiologyresearch.org by IP: 88.99.165.207 On: Fri, 12 May 2017 23:19:43