* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Solid phase peptide synthesis (SPPS), strategies, resins and
Survey
Document related concepts
Point mutation wikipedia , lookup
Citric acid cycle wikipedia , lookup
Butyric acid wikipedia , lookup
Metalloprotein wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Oligonucleotide synthesis wikipedia , lookup
Genetic code wikipedia , lookup
Proteolysis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biochemistry wikipedia , lookup
Biosynthesis wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Transcript
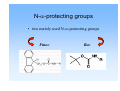
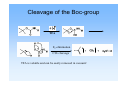
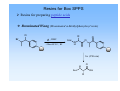
Solid phase peptide synthesis (SPPS), strategies, resins and comparison with Fmoc-strategy General scheme of SPPS attach to linker deprotect amino function couple n times deprotection and coupling cleave N-α-protecting groups • two mainly used N-α-protecting groups Fmoc Boc Boc-protecting group Boc = tert. Butyloxycarbonyl or tert. Butoxycarbonyl stable to bases and nucleophiles unaffected by catalytic hydrogenation deprotection with TFA is rapid Introduction of the Boc-group Di-tert-butyl-dicarbonate = Boc-anhydride = (t-boc)2O 2-(tert. Butoxycarbonyloxyimino)-2-phenylacetonitril = Boc-NO O O O O O O O O N N • • • • both commercially available storage in refrigerator for extended periods (t-boc)2O is more expensive preparation: „Kates S. A., Albericio F. (ed): Solid-Phase Synthesis, A practical guide, Marcel Dekker Inc. 2000, p. 105-107“ N-α-Boc protected amino acids • already N-α-Boc protected amino acids can simply be bought from firms like „Novabiochem“ O e.g. Boc-Ala-OH O N H COOH Cleavage of the Boc-group TFA E1-elimination CO2 cleavage TFA is volatile and can be easily removed in vacuum! Resins for Boc SPPS Resins for preparing peptide acids Merrifield (Chloromethylstyrene-divinylbenzene) - was the standard support for the synthesis of peptide acids by Boc SPPS now only used in the synthesis of small to medium sized peptides, because the benzylic ester resin linkage is not completely stable towards repetitive treatment with TFA , DMF Cl + BocAS-Cs+, boc KI O H N O R - attachment of the C-terminal residue is achieved by heating the resin in DMF with the appropiate amino acid cesium salt in the presence of KI - cleavage is affected by treatment of resin with HF or TFMSA, or by hydrogenolysis - alcohols can be released using reducing agents like DIBALH or LiBH4 - methyl esters can be produced by transesterification with NaOMe Resins for Boc SPPS Resins for preparing peptide acids PAM (4-Hydroxymethylphenylacetamidomethyl) - also a standard support for Boc SPPS stabilizing effect of the phenylacetamidomethyl function on the ester linkage reduction of losses during repetitive TFA acidolysis boc O H N O R O N coupling: H a) first addition of the PAM-linker on to the aminomethyl resin and then coupling of the Boc-protected amino acid b) first coupling of the Boc-protected amino acid to the PAM-linker and then reaction with the aminomethyl resin followed by end-capping of unreacted aminomethyl groups cleavage: - treatment with HF or TFMSA releases the peptide acid Resins for Boc SPPS Resins for preparing peptide acids Brominated Wang (Brominated α-Methylphenylacyl resin) O Br R , DMF + BocAS-Cs+, KI boc O O N H O hν (350 nm) boc O H N OH R Resins for Boc SPPS Resins for preparing peptide amides BHA / MBHA (Benzhydrylamine / 4-Methylbenzhydrylamine) - used for the synthesis of peptide amides by Boc SPPS attachment of the first amino acid with standard methods of amide bond formation cleavage of the carboxamides with HF or TFMSA MBHA is more acid sensitive and the peptide amide can be released with HF or TFMSA under less drastic conditions NH2 NH2 Resins for Boc SPPS Resins for preparing C-terminally modified peptide fragments Brominated PPOA (Brominated [4-Propionylphenoxy]-acetic acid) - versatile resin for the Boc SPPS of peptide acids, esters and hydrazides by photolytic or nucleophilic cleavage O O Br O N H , DMF + BocAS-Cs+, KI boc O O O H N O R O NEt3 in methanol/ dioxane hν or NaOH in dioxane boc O H N O R N H boc NH2NH2/ DMF O H N OMe R boc O H N R N H NH2 Resins for Boc SPPS Resins for preparing C-terminally modified peptide fragments Oxime resin - attachment of the first amino acid with DCC afterwards acetylation of unreacted oxime groups cleavage from the support by various nucleophiles like NaOH, NH3, R1NH2, NaBH4, MeOH, NH2NH2 NO2 NO2 amino acid, DCC HO N R H N O O N General aspects of Boc strategy • cleavage of the N-α-Boc-protection group with TFA (usually 25-50% (v/v) in DCM) • side chain protecting groups must be orthogonal(!), that means: • stable against TFA during N-α-Boc deprotection • removable at the end of peptide synthesis • release of the peptide from the resin by treatment with HF Side chain protecting groups for Boc strategy Arg: Toluolsulfonyl- (Tos) or Mesitylen-2-sulfonyl-group (Mts) cleavage: HF/anisole (Tos) thioanisole (Mts) Ser, Thr, Tyr: Benzyl (Bzl) O cleavage: HF R boc N Asp, Glu: Bzl COOH H O Lys: Fmoc or 2-Chlorobenzyloxycarbonyl (2ClZ) cleavage: piperidine (Fmoc), TFA (2ClZ) O O Cl H N Cys: Acetamidomethyl (Acm) or MeO 4-Methoxybenzyl (MeOBzl) cleavage: Hg2+- or Ag+-salts (Acm), HF (MeOBzl) His: Dinitrophenyl-group (DNP) cleavage: thioles DNP O O H N OH O HN OH HN boc O S OH boc NH boc Advantages and disadvantages of Boc- and Fmoc-strategy Advantages Boc easy to introduce Boc-amino acids are stable at room temp. for extended periods (but storage at 4°C is recommended) deprotection with TFA is rapid successful strategy for many peptide synthesis applications good coupling results Disadvantages temporary and permanent (side chain) protecting groups are both acid labile side chain deprotection during repeated TFA treatment can occur repeated TFA-mediated N-α-deprotection over the course of a long synthesis may lead to modification and/or degradation of sensitive peptide sequences difficulties for fragile peptides that don‘t survive the relatively harsh final HF cleavage Boc-strategy requires the use of “dangerous“ HF and expensive laboratory apparates side reactions are possible: t-Bu+ reacts with nucleophilic side chains like trp, tyr, met, his side chain protecting groups / adding of scavengers (1,2-Ethanedithiole) to the deprotection reagent Advantages and disadvantages of Boc- and Fmoc-strategy Advantages Fmoc orthogonal protection sheme Fmoc-amino acids are easy to prepare in crystalline form in high yield and stable when stored at 4°C milder reaction conditions: milde base (piperidine) for N-α deprotection, TFA only for the final resin cleavage and deprotection progress of each deprotection reaction can be followed by real time spectrophotometric monitoring the release of the cleaved Fmoc-group at 300-320 nm Disadvantages Piperidine: harmful vapor, toxic side reactions: • aspartimide formation at AspX residues like Asp-Gly, -Ser, -Thr, -Asn, -Gln • linker-bound C-terminal Cys undergoes significant racemisation (ca. 0,5%) with each cycle of Piperidinetreatment Literature -Novabiochem 2002/3 Catalog -www.cup.unimuenchen.de/oc/carell/lehre/peptide2.pdf -Kates S. A., Albericio F. (ed): Solid-Phase Synthesis, A practical guide, Marcel Dekker Inc. 2000