* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 4.2 Notes
Introduction to quantum mechanics wikipedia , lookup
ALICE experiment wikipedia , lookup
Standard Model wikipedia , lookup
Identical particles wikipedia , lookup
ATLAS experiment wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Electron scattering wikipedia , lookup
Nuclear structure wikipedia , lookup
Electric charge wikipedia , lookup
Compact Muon Solenoid wikipedia , lookup
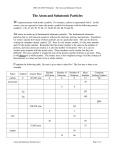
Name: __________________________________________ Date: ___________________ Period: _______ Chapter 4.2 Notes 1. Subatomic particles include __________, _____________, and ________. 2. Protons A. A ______________ is a ___________________ charged subatomic particle that is found in the _______________ of an ______________. B. Each ________________ must contain at least one ____________. C. Each ________________ is assigned a charge of ______________. D. Some _______________ contain more than ___________ protons. 3. Electrons A. An ______________ is a _________________ charged subatomic particle that is found in the space _______________ the nucleus. B. Each electron has a charge of ___________. 4. Neutrons A. A ___________ is a neutral subatomic particle that is found in the ____________of an ______________. B. It has a ___________ almost exactly equal to that of a _________. Particle Properties of Subatomic Particles Symbol Relative Relative Charge mass (Proton = 1) Actual Mass (g) 5. Comparing Subatomic Particles A. ______________, ______________, and _______________ can be distinguished by ________, _________, and ___________ in an atom. B. Protons and ______________ have almost the same ___________. C. According to the relative mass of electrons in the above table; how many electrons would it take to equal the mass of a proton or neutron? _______________ D. ______________ have a charge that is ______________ in size but __________________ the charge of a proton. E. ___________________ have no charge. F. Where are protons and neutrons found? _________________________________________________ G. Where are electrons found? _________________________________________________ Name: __________________________________________ Date: ___________________ Period: _______ H. Everything scientists know about the ____________________ and _______________________ particles is based on how the particles ________________. We are still ______________ to see the inside of an ________. However, we do have microscope that can show how _____________ are arranged on the _____________ of a material. 6. Atomic Number and Mass Number A. The ____________ number of an element equals the number of _______________ in an _______________ of that element. B. The ___________ of any given element always have the ________ number of ___________. C. There is __________ proton in the _________ of every _________ atom. What is the atomic number for hydrogen? ________ D. __________ of different ____________ have different numbers of __________________. E. What other element have an atomic number of 1? _________________________________________________ F. _________ numbers can be used to refer to ____________ since they are _________________ to that element. G. Each ____________ charge in an _____________ is balanced by a ______________ charge because atoms are ________________. H. The atomic number of an element also _______________ the number of __________________ in an atom. 7. Mass Number A. The ____________ number of an _____________ is the sum of the ______________ and ______________ in the ________ of that atom. B. You can find the number of ________________ by subtracting the _________________ number from the _________________ number. C. _____________________________________________________ Fill in the table about the nucleus of these atoms. Element Neutrons Protons Mass # Atomic # Lithium Oxygen Boron Name: __________________________________________ Date: ___________________ Period: _______ Element Sulfur Iron Silver Hydrogen Aluminu m Fill in the table about these atoms. Atomic # Mass # Neutrons Protons Electrons 8. Isotopes A. _______________ are atoms of the __________ element that have different numbers of ______________ and different ______________ numbers. B. Isotopes of an __________ have the same _____________ number but different ______________ numbers because they have different numbers of _______________. Ex. 1) Oxygen has 8 _____________ and 8 _________________ with a mass number of _________________. An _________ of oxygen may have _____ protons and ______ neutrons with a mass number of __________. C. These isotopes are referred to by their _______________ number. Ex. 2) ________________________, _________________________ D. Since ____________-1 has no ____________ and one __________ adding a neutron ________________ its ___________________. D. Water that contains hydrogen-_____ atoms in place of hydrogen-_______ atoms is called _______________ water. Fill in the table about these atoms. Number of Orbitals Energy Levels X self X self X self X self Max # of Electrons X2 X2 X2 X2 9. Energy Levels & Orbitals A. When all of the ___________________ in an ________ have the lowest possible _________________ , the atom is said to be in its _______________ state. B. When an ________ gains energy and moves ____ energy levels it is in an _______________ state.