* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Alcohols
Survey
Document related concepts
Transcript
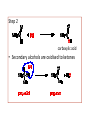
unlimited number of carbon compounds can be formed by the addition of a functional group to a hydrocarbon. A functional group is a reactive portion of a molecule. The combinations of functional groups with hydrocarbons produce a vast number of compounds. Organic molecules containing a hydroxyl group are known as alcohols. Classification of Alcohols Alcohols are classified as primary, secondary, and tertiary Classification is determined by the number of alkyl group to the carbon bonded to the hydroxyl. Primary alcohols In a primary (1°) alcohol, the carbon which carries the -OH group is only attached to one alkyl group. Notice that it doesn't matter how complicated the attached alkyl group is. In each case there is only one linkage to an alkyl group from the CH2 group holding the -OH group. Secondary alcohols In a secondary (2°) alcohol, the carbon with the -OH group attached is joined directly to two alkyl groups, which may be the same or different. Tertiary alcohols In a tertiary (3°) alcohol, the carbon atom holding the -OH group is attached directly to three alkyl groups, which may be any combination of same or different. In phenols the -OH group is directly attached to a carbon that is part of an aromatic ring. Alcohols and phenols are similar in some ways, but there are enough differences so that they are considered different functional groups. One major difference is that phenols are typically about a million times more acidic than alcohols. Physical Properties Boiling Points The chart shows the boiling points of some simple primary alcohols with up to 4 carbon atoms. They are: They are compared with the equivalent alkane (methane to butane) with the same number of carbon atoms. Notice that: • The boiling point of an alcohol is always much higher than that of the alkane with the same number of carbon atoms. • The boiling points of the alcohols increase as the number of carbon atoms increases. • The patterns in boiling point reflect the patterns in intermolecular attractions. Hydrogen bonding • Hydrogen bonding occurs between molecules where you have a hydrogen atom attached to one of the very electronegative elements fluorine, oxygen or nitrogen. • In the case of alcohols, there are hydrogen bonds set up between the slightly positive hydrogen atoms and lone pairs on oxygens in other molecules. • The hydrogen atoms are slightly positive because the bonding electrons are pulled away from them towards the very electronegative oxygen atoms. Solubility of alcohols in water • The small alcohols are completely soluble in water. • Solubility falls as the length of the hydrocarbon chain in the alcohol increases. Chemical Properties 1. Ceric ammonium nitrate test • General test for alcohols • +ve result red coloration indicates the presence of alcohols. 2. Oxidation Reaction • This test is to distinguish Primary and secondary alcohols from tertiary alcohols • Primary alcohols can be oxidised to either aldehydes or carboxylic acids depending on the reaction conditions. Step 1 aldehyde Step 2 carboxylic acid • Secondary alcohols are oxidised to ketones • Tertiary alcohols does not react because there is no free H to loss • +ve result: Purple colorless 3. Ferric Chloride test: • Addition of aqueous FeCl3 to phenol gives a colored solution. • Depending on the structure of phenol, the color can vary from green to purple. Cl Fe Cl O OH + Light yellow FeCl3 + Violet HCl 4. Solubility with NaOH (acidity test) • Phenol is a weak acid therefore reacts with a base • Alcohols are less acidic than Phenols therefore are not neutralized by NaOH O-Na+ OH + NaOH + H2O