* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Mitral Stenosis at Autopsy - JOURNAL of the Louisiana State
Cardiac contractility modulation wikipedia , lookup
Electrocardiography wikipedia , lookup
Cardiovascular disease wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Heart failure wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Infective endocarditis wikipedia , lookup
Coronary artery disease wikipedia , lookup
Aortic stenosis wikipedia , lookup
Myocardial infarction wikipedia , lookup
Artificial heart valve wikipedia , lookup
Jatene procedure wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Atrial fibrillation wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
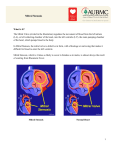
Journal of the Louisiana State Medical Society Pathology Image of the Month Mitral Stenosis at Autopsy Harry Porterfield, DO; Meaghen Ashby, MD; William P. Newman, MD; and Robin McGoey, MD A 52-year-old woman decedent was presented to the hospital autopsy service for a coroner authorized complete autopsy following an admit urine toxicology screen that was positive for cannabinoids. Prior to admission, she was found unresponsive at home after a two month history of increasingly progressive shortness of breath. She was transported to the emergency department and resuscitated after prolonged arrest. She was then admitted to the intensive care unit and subsequently was documented to have significant anoxic brain injury. Care was withdrawn by the family and death was declared on hospital day five. Medical history was reported for type 2 diabetes mellitus, and bipolar schizoaffective disorder with multiple prior psychiatric admissions. Her medical record review revealed a transthoracic echocardiogram two months prior to admission that documented mild mitralregurgitation, moderate mitral valve stenosis and a thickened valvular and subvalvular mitral apparatus with restricted motion of the posterior leaflet. Left atrial enlargement was marked, left ventricular hypertrophy was moderate, and pulmonary hypertension was graded as severe. The ejection fraction was estimated at 70 percent. She was discharged with outpatient follow-up to the cardiology department but was noncompliant with recommendations. At autopsy, the heart exam confirmed a markedly dilated left atrial chamber with a slightly raised, broad fibrous plaque along the atrial endocardial surface. The mitral valve circumference was small at 8.0 cm (8.2-9.1cm) and the leaflets were thickened. The mitral valve chordae tendinae were notably short and thick with evident fusion at multiple sites. A gross photo across the left atrioventricular axis and mitral valve is seen in Figure 1. Additional findings at autopsy included cardiomegaly at 720 gm (234-390 gm), bilateral pulmonary edema containing hemosiderin laden macrophages, congestive hepatosplenomegaly and plexogenic arteriopathy on pulmonary microscopic examination. Figure 1: The left atrioventricular view of the heart through the mitral valve at autopsy showing left atrial dilatation, a left atrial endocardial raised plaque, as well as short and thick chordae with chordal fusion. What is the underlying clinical diagnosis in this case and what is the most likely cause of death? Answer is on p. 145 144 J La State Med Soc VOL 167 May/June 2015 Journal of the Louisiana State Medical Society DIAGNOSIS: Chronic rheumatic mitral valvulitis and heart failure DISCUSSION Rheumatic heart disease (RHD) is the result of valvular damage caused by an abnormal immune response to group A streptococcal (GAS), Streptococcus pyogenes infection, usually during childhood, and typically after pharyngitis.1 Although nearly eradicated in developed nations, RHD continues to be a major health challenge in developing nations where it remains the leading cause of heart failure in children and young adults, accounting for approximately 250,000 deaths annually.2 Despite the potentially significant morbidity and mortality associated with RHD and the predicted rise in the number of people at risk for RHD over the next 20 years, the disease draws little attention from the medical community as evidenced by a paucity of publications on the topic.1 GAS pharyngitis leads to acute rheumatic fever (ARF) in less than 3 percent of cases. However, when it does appear, it occurs within three weeks following the initial infection.3 The incidence worldwide of ARF in children between 5 and 14 years of age is reported to be 350,000 cases annually; though some populations report endemic numbers of cases.1,3 In 1944, Jones described the main clinical features of ARF, which have subsequently been modified, revised and expanded by the World Health Organization (WHO)4 into diagnostic criteria based on a variety of combinations of clinical features, termed major and minor (Table). Cardiac involvement has been documented in up to 50 percent of those diagnosed with ARF. The cardiac hallmark of acute RF is mitral regurgitation of variable severity and, in one series utilizing echocardiographic evaluation of ARF, prolapse of the anterior leaflet.5,6 Pericarditis is also common in ARF and can accompany the valvular pathology. The valvulopathy of ARF can either resolve or can progress into rheumatic heart disease (RHD) which has recently been reported to affect nearly 20 million people worldwide.1 The pathogenesis and risk factors for progression are not widely understood though research does suggest progression occurs more frequently in females.7 RHD may be diagnosed in an individual known to have had prior ARF. Usually RHD is diagnosed in patients who were previously asymptomatic or who cannot recall being affected with ARF or GAS. In the acute phase of RF, cardiac pathology reveals a mixed inflammatory cell pancarditis involving the epi-, myo- and endocardium. A nodular lesion, termed the Aschoff body, may be identifiable microscopically within the myocardium (mainly of the left atrium) or in the pericardium. The Aschoff body, at its pathognomoic phase, is a fusiform nodular aggregate of spindle shaped cells admixed with Table: 2002-3002 WHO criteria for diagnosis of rheumatic fever and rheumatic heart disease (based on revised Jones Criteria)4 Diagnostic Categories Criteria Primary episode of RF 2 Major or 1 Majora and 2 Minorb plus evidence of preceding GAS infectionc Recurrent RF in patient without RHD 2 Majora or 1 Majora and 2 Minorb plus evidence of preceding GAS infectionc Recurrent RF in patient with RHD 2 Minorb plus evidence of preceding GAS infectionc Chronic rheumatic valvulitis (patients presenting for first time with pure mitral stenosis or mixed mitral valve disease and/or aortic valve disease) Do not require any other criteria to be diagnosed as having RHD a Major Criteriaa: carditis, polyarthritis, chorea, erythema marginatum, subcutaneous nodules Minor Criteriab: clinical (fever, polyarthralgia), laboratory (elevated acute phase reactants) Evidence of preceding GAS Infectionc: electrocardiogram (prolonged PR interval), elevated or rising antistreptolysin-O or other streptococcal antibody, or positive throat culture or rapid antigen test for GAS, or recent scarlet fever 145 J La State Med Soc VOL 167 May/June 2015 Journal of the Louisiana State Medical Society chronic inflammatory cells and fibroconnective tissue. Larger mononuclear cells called Anitschkow cells may be seen within the nodule and, on cut section, bear an owl-eye appearance. Aschoff bodies, thought to be manifestations of the inflammatory response to RF, regress and disappear within 3-4 months. Grossly, cardiac pathology in the ARF phase may also reveal a fibrinous pericarditis as well as finely nodular, uniformly sized, verrucous, vegetations along the lines of closure of, primarily, the mitral valve.8 Chronic RHD, either from repeated attacks of RF or from progression of ARF takes on a strikingly different histopathologic form; and mitral valvulopathy is the primary problem. Gross features are characterized by mitral valve stenosis due to commissural fusion, leaflet thickening and subvalvular disease with shortened, thickened, and often fused, chordae tendineae. From the left atrial projection, the valve is said to take on a ‘fish mouth’ appearance. The aortic valve also can show commissure fusion and calcification of the cusps. The tricuspid and pulmonic valves are affected less frequently. Further features can include dilatation of the left atrial chamber and raised map-like plaques (MacCallum patch) along the atrial endocardium due to mitral regurgitation.8-10 The autopsy case presented here of a relatively young female with a several month history of apparent heart failure and recent echo diagnosis of mitral stenosis illustrates the highly characteristic mitral valvulopathy of chronic RHD. Further histopathologic examination of the raised left atrial lesion demonstrated the characteristic features seen in a MacCallum plaque, or mitral regurgitation jet lesion, of bland, loose subendocardial fibrosis.10 The postmortem diagnosis of RHD was, not surprisingly, made in this female individual unbeknownst to have had prior ARF. There is no specific nonsurgical treatment for RHD other than for its complications including heart failure, atrial fibrillation, ischemic embolic events and infective endocarditis. Prevention strategies are postulated to be the most sustainable method for disease control in developing nations.1 Surveillance of RHD patients is most often via echocardiography and clinical exam. The prognosis of RHD largely relates to clinical progression of the valvulopathy into left ventricular systolic dysfunction. Most often, the mitral stenosis is indolent and slowly progressive with a latency period as long as 20-40 years between initial infection and onset of symptoms. However, rheumatic valvulopathy progresses far more rapidly in developed countries such as the US. Mortality in untreated RHD is most often attributable to heart failure in 60-70 percent of patients. It can also be vascular embolism in 20-40%; endocarditis in 1-5%. If pulmonary hypertension is superimposed as evidenced in the current case, the mean survival time falls to less than three years.11 With no evidence of thromboemboli or endocarditis in the current case along with the autopsy findings of congestive splenomegaly and plexogenic arteriopathy in the pulmonary vascular system coupled with antemortem historical evidence of recent shortness of breath, the decedent’s proximate cause of death was presumably 146 J La State Med Soc VOL 167 May/June 2015 heart failure due to chronic rheumatic mitral valvulopathy. REFERENCES 1. Marijon E, Mirabel M, Celermajer DS, et al. Rheumatic heart disease. Lancet. 2012;379:953-964. 2. Carapetis JR, Steer AC, Mulholland ED, et al. The global burden of group A streptococcoal diseases. Lancet Infect Dis. 2005;5:685-694. 3. Essop MR and Peters F. Contemporary issues in rheumatic fever and chronic rheumatic heart disease. Circulation. 2014;130:2181-2188. 4. WHO. Rheumatic Fever and Rheumatic Heart Disease: Report of a WHO Expert Consultation. Geneva 29 Oct-1 Nov 2001. Geneva, Switzerland: WHO; 2004. 5. Kinsley RH, Pocock WA. Rheumatic fever and rheumatic heart disease.In: Barlow JB, ed. perspectives on the mitral valve. Philadelphia, PA: FA Davis Company; 1987:227–245. 6. Marcus RH, Sareli P, Pocock WA, et al. Functional anatomy of severe mitral regurgitation in active rheumatic carditis. Am J Cardiol. 1989;63:577–584. 7. Carapetis JR, Wolff DR, Currie BJ. Acute rheumatic fever and rheumatic heart disease in the top end of Australia’s Northern Territory. Med J Aust. 1996; 164: 146–149. 8. Burke AP. Pathology of rheumatic heart isease. Sep 9, 2013; Available from: Medscape Reference [serial online]. Accessed March 16, 2015. Available at http://emedicine.medscape.com/ article/1962779-overview#a1 9. Chopra P, Gulwani H. Pathology and pathogenesis of rheumatic heart disease. Ind J Pathol Microbiol. 2007;50:685-697. 10. Shivakumarswamy U, Sinhasan SP, Purushotham R, et al. MacCallum Plaque of the Heart: A Medicolegal Case. Heart Views. 2010;11:71-73. 11. Ward C, Hancock BW. Extreme pulmonary hypertension caused by mitral valve disease: natural history and results of surgery. Br Heart Journal.1975;37:74–78. All authors are affiliated with the Department of Pathology at Louisiana State University School of Medicine in New Orleans. Dr. Porterfield is a third year pathology resident, Dr. Ashby is a first year pathology resident, Dr. Newman is a professor of pathology and Dr. McGoey is an associate professor of athology and residency program director. Acknowledgements: The authors would like to gratefully acknowledge the support of the Orleans Parish Coroner’s Office for providing the case material for this report.