* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Naming complexes and isomerisation pdf
Jahn–Teller effect wikipedia , lookup
Hydroformylation wikipedia , lookup
Oxidation state wikipedia , lookup
Metal carbonyl wikipedia , lookup
Cluster chemistry wikipedia , lookup
Spin crossover wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Stability constants of complexes wikipedia , lookup
COORDINATION CHEMISTRY
Modification date: 29 January 2015
The information in this document was obtained from the sources listed at the end of the
documents. This is just a summary to help you and is not meant to replace textbooks. Please
consult textbooks for a more comprehensive overview of the topics. Please inform Dr EL Viljoen
of any correction is required.
Two suggested books to read:
Cotton, F.A., Wilkinson, G. & Gaus, P.L. (1987). Basic inorganic chemistry. Second edition. 157-171.
Housecroft, C.E. & Sharpe, A.G. (2012) Inorganic Chemistry. Fourth edition. p654-656.
McMurry J.E. & Fay, R.C. (2012) Chemistry , six edition. 817-832.
Structure of coordination complexes
Info from Mc Murry and Fay, 2012
A metal complex is a compound in which the central metal ion or atom (yellow sphere, vanadium
ion, in middle in diagram below) is attached to a group of surrounding molecules or ions by
coordinate covalent bonds. An metal complex is indicated with square brackets, for
example [Rh(NH3)5Cl]2-.
The molecules or ions (e.g. oxalate, the red, grey and white spheres in the diagram below) that
surround and bond to the central metal ion or atom are called the ligands. The word ligand is from
the Latin word ligare which means to bind. The ligands coordinate to the metal.
Coordination compounds are compounds that contain metal complexes. An example of such an
coordination compound is [Rh(NH3)5Cl](NO3)2.
The central metal and the ligands bound to it are called the coordination sphere of the complex.
The atoms (the red spheres that represent oxygen) that are directly attached to the metal are called
ligand donor atoms. The ligands act as Lewis basis forming coordinate covalent bonds with the
central metal atom or ion.
The metal centre is the acceptor atom (the yellow sphere in the middle). The central metal or ion
acts as a Lewis acid.
The number of ligand donor atoms (there is 5 red spheres) around one metal ion/atom (yellow
sphere) is the coordination number. There are three ligands in the example below but the
coordination number is 5 since two of the oxalate ligands are bidentate (ligand bonding with two
donor atoms). The coordination number is thus 2+2+1 = 5.
1
The coordination number of the complex below is 5 and the geometry is square pyramidal.
Metal complexes have different coordination geometry (characteristic shapes) which depend on
their coordination number. Common coordination geometries are shown below.
7
2
3
4
6
5
2
https://organometallicchem.wordpress.com/2012/01/10/gee-im-a-tree-predicting-the-geometry-oforganometallic-complexes
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Organometallic_Chemistry/Structural_Fundamen
tals/Predicting_the_Geometry_of_Organometallic_Complexes
Examples of different coordination numbers
Housecroft and Sharpe, 2012
http://en.wikipedia.org/wiki/Coordination_geometry
Cotton et al., 1987
The pink spheres are the metal centres and the white spheres are the ligands
Coordination number two
Linear
This coordination is typical for cations that have an oxidation state of one like Cu, Ag and Au. The
coordination geometry is linear. Examples are [H3N-Ag-NH3]+, [NC-Ag-CN]- and [Cl-Au-Cl]-.
3
Coordination number three
The most common coordination geometries are trigonal plane and the trigonal pyramid. Examples
are for the trigonal planar are HgI3- and [Cu(CN)3]2- and for the trigonal pyramidal is SnCl3-.
Trigonal planar
Trigonal pyramidal
Coordination number four
The most common coordination geometries are either tetrahedral or square-planar. Examples of
tetrahedral complexes are Li(H2O)4+, BeF42-, CoBr42- and Ni(CO)4. Tetrahedral geometry is preferred
for valence electron configurations d0 and d10. Square planar is preferred for d8 metal ions and is
common for complexes of ions Ni2+, Pd2+, Pt2+, Rh+, Ir+ and Au3+. An example for square planar
coordination geometry is [PtCl2(NH3)2] .
Tetrahedral
Square-planar
Tetracarbonylnickel
trans - PtCl2(NH3)2
4
Coordination number five
The most common coordination geometries are the trigonal bipyramid and the square pyramid.
Many five coordinate complexes do not have either structure but a mixture since there is little
difference in energies between the two coordination geometries. For example [Ni(CN)5]3- for a
crystalline salt in which both geometries are present.
Square pyramid
Trigonal bipyramid
Coordination number six
In the octrahedron all six the M-L bonds are equivalent (of the same length). An example is [TiF6]2-,
[Cr(bipy)3]3+ and [Co(H2O)6]2+ . This is typical when all the ligands are of the same type. When
different kinds of ligands are present distortion takes place and all the M-L bonds are not of the
same length any more.
5
Octahedral distortions
(Cotton et al., 1987)
When different kinds of ligands are present distortion takes place and all the M-L bonds are not of
the same length any more. In some cases where all the ligands are chemically the same, octahedron
is also distorted due to inherent electronic effects in the metal or by forces in the surroundings.
A compression or elongation in one of the L-M-L axis relative to the other two is named tetragonal
distortion (a≠b 3 a-lengths are the same and three b-lengths are the same).
When L-M bonds differs the coordination geometry is named a rhomic structure (a≠b≠c)
6
If the octahedron is compressed or elongated on an axis connecting the centres of the two opposite
triangular faces the coordination geometry is named trigonal.
The trigonal prism coordination geometry is only rarely found. Examples are hexamethyl complexes
with d0 configuration, such as hexamethyltungsten and [Zr(CH3)6]2-.
[Zr(CH3)6]2-
7
TYPES OF LIGANDS
(Cotton et al., 1987)
Monodentate and polydentate ligands
Monodentate ligands donate one electron pair. Examples of monodentate ligands are Cl-, CN-, NH3
and H2O.
Polydentate ligands also called multidentate are ligands that contain two or more atoms that
simultaneously form a two-electron donor bond to the same metal ion. Polidentate ligands are also
called chelating agents. A complex that contains polydentate ligands, a chelated complex is
thermodynamically more stable that a complex than has similar monodentate ligands. The
increased thermodynamically stability as a result of chelating agents are called the chelate effect.
Polydentate ligands can further be classified according to the number of two electron donor bond
made to the same metal ion.
Bidentate ligands have two possible points of attachment to the metal ion. A bidentate ligand has
therefore two donor atoms.
4 membered ring: carboxylate; nitrate; dithiocarbate; sulphate
5 membered ring: Ethylenediamine, en H2NCH2CH2NH2
8
5 membered ring: Oxalate, C2O42-
6 membered ring: Bipyridyl, bipy
6 membered ring: Acetylacetonate, acac
Tridentate ligands have three possible points of attachment to the metal ion. A tridentate ligand
therefore has three donor atoms. In the case of the diethylene triamine (dien) the three nitrogen
atoms are the donor atoms, since each of them can donate a electron pair to the metal atom/ion
center.
Diethylene triamine, dien
9
Tetradentate ligands have four possible points of attachment to the metal ion.
Porphyrin
Hexadentate ligands have six possible points of attachment to the metal ion.
Ethelenediaminetretaacetate ion, EDTA4-.
10
The table below shows some examples ligands with different denticities. The donor atoms are
shown in pink.
Info from Housecroft and Sharpe, 2012
11
Ambidentate ligand
An ambidentate ligand is a ligand that can bond in two ways.
Examples:
In thiocyanate, SCN-, either (not both simultaneously to the same metal ion) the S or the N can be
the ligand donor atom. The name depends on which atoms is the donor atom. When S is the donor
atom the ligand name is thiocyanato and when the N is the donor atom the ligand name is
isothiocyanato.
In nitrogen dioxide, nitrite, NO2-, either (not at the same time to the same metal ion) the N or the O
can be the ligand donor atom. In the case where the N is the donor atom the name of the ligand is
nitro and where the O is the donor atom the ligand name is nitrito.
In cyanide, CN- either the C or the N can be the ligand donor atom. When the carbon is the ligand
donor atom the name is cyano and when the N is the donor atom the name is isocyano.
Bridging ligands
A bridging ligand is a ligand that is bonded to two metals ions at the same time.
[NH3)5Co-NH2-Co(NH3)4(H2O)]Cl5
Pentraamminecobalt(III)-µ-amidotetraammineaquacobalt(III)chloride
Tetraamminecobalt(III)- µ-amido-µ-peroxotetraamminecobalt(III)
Di-µ-cholobis[terachloroniobium(IV)]
[NH3)5Cr-OH-Cr(NH3)5]Br5
µ-Hydroxobis[pentaamminechromium(III)] bromide
12
OXIDATION NUMBERS
McMurry J.E. & Fay, R.C. (2012) Chemistry , six edition. 817-832
Determining the oxidation number of transition metals in a complex.
• Alkali metal atoms ions (group 1) have an oxidation number of +1 and alkali earth metals
ions (group 2) have an oxidation number of +2.
• The oxidation number of oxygen is usually -2 unless it is part of a peroxide component when
oxygen has an oxidation number of -1.
• The oxidation number for hydrogen is +1 when bonded to non-metals and -1 when bonded
to metals.
• The oxidation number of halogens (group 7) are -1 when bonded to another element.
• The charge of the ligand is used as its oxidation state as a unit. See table 3 for charges.
• The sum of the oxidation numbers of all times in a neutral compound is zero. The sum of
the oxidation numbers of all atoms in a polyatomic ion equals the charge of the ion.
Examples:
X + 4(0) = +2
Oxidation number of Cu:
[Cu(NH3)4]2+
The oxidation number of copper in the complex is thus +2.
Oxidation number of Rh:
[Rh(NH3)5Cl](NO3)2
The oxidation number for rhodium, Rh, is thus +3.
X+ 5(0) + (-1) + 2(-1) = 0
Oxidation number of Co:
[Co(NH3)6]3+
The oxidation number for cobalt is thus +3.
X+6(0) = +3
Oxidation number of Ni:
[Ni(H2O)6]2+
The oxidation number for nickel is thus +2.
X + (6(0) = +2
Oxidation number of Co:
[Co(NH3)5Cl]Cl2
The oxidation number for cobalt is thus +3.
Oxidation number of Mo:
Na2[MoOCl4]
The oxidation number for molybdenum is thus +4.
Oxidation number of Fe:
[Fe(CO)4Cl2]
The oxidation number for iron is thus +2.
X = +2
X + 5(0) + (-1) + 2(-1) = 0
2(1) + X + (-2) + 4(-1) = 0
X + 4(0) + 2(-1) = 0
13
NAMING OF COMPLEXES
Cotton et al., 1987
McMurry J.E. & Fay, R.C. (2012) Chemistry , six edition. 817-832.
IUPAC rules:
• If the compound is a salt, name the cation (+) first and then the anion (-).
For example K4[Fe(CN)6] is named as potassiumhexacyanoferrate(II).
• If the complex is an ion or neutral, name the ligands first and then the metal.
For example Fe(CN)64- is named hexacyanoferrate(II) ion.
• If the complex is an anion (negatively charged), the metal name ends with ate and the Latin
form is used. Please consult table 1 below for examples.
Table 1: Metallate names
Metal
Anion name
aluminium
aluminate
chromium
chromate
cobalt
cobaltate
copper
cuprate
gold
aurate
lead
plumbate
molybdenum
molybdate
•
•
•
•
Anion name
ferrate
manganite
nickelate
platinate
zincate
argentate
tungstate
Roman numerals in parentheses are used to indicate the oxidation state of the metal. It is
placed after the metal name.
If a complex contain more than one ligand the prefixes are used as shown in the table below.
If the name of the ligand itself contains a prefix for example ethylenediamine, put the ligand
name in parentheses (brackets) and use the alternative prefix which is shown in the brackets
in the table below.
Table 2: Prefix names
number of ligand
prefix (alternative)
1
2
3
4
5
6
Metal
iron
manganese
nickel
platinum
zinc
silver
tungsten (W)
---di(bis)
tri(tris)
tetra (tetrakis)
penta (pentakis)
hexa (hexakis)
number of
ligand
7
8
9
10
11
12
prefix
hepta
octa
nona (ennea)
deca
undeca
dodeca
Ligands are usually listed alphabetic order ignoring the prefixes that designate the number
of each type of ligand. See table 3 for commonly used ligand names.
Indicate isomers with an italic prefix followed by a hyphen. For example cis-, trans-, fac-,
mer-. The abbreviations fac-, mer- stands for factial and meridional respectively and are
used in the naming of octrahedral complexes.
14
•
Indicate bridging ligands by the prefix µ. If there is more than one bridging component of
the same type then the prefix µ is combined with prefix names from table 2. For example if
there are two bridging components of the same type then the prefix will be di-µ.
Table 3: Ligand names
Anionic
bromide, Brfloride, Fchloride, Clhydride, Hhydroxide, OHacetate, CH3CO2carbonate, CO32oxide, O2peroxide, O22oxalate, C2O42sulfate, SO42nitrite NO21nitrite ONO
cyanide, CNcyanide, NCthiocyanate, SCNthiocyanate, NCS-
Ligand name
bromo
fluoro
chloro
hydrido
hydroxo
acetato
carbonato
oxo
peroxo
oxalato
sulfato
nitro
nitrito
cyano
isocyanto
thiocyanato
isothiocyanato
Neutral
ammonia, NH3
water, H2O
carbon monoxide, CO
nitrogen oxide, NO
ethylenediamine, en
pyridine, C5H4N
(C6H5)3P
(C2H5)3P
Ligand name
ammine
aqua
carbonyl
nitrosyl
ethylenediamine
pyridine
triphenylphosphine
trietthylphosphine
Anions with names ending in ate have ligand names ending in ato for example SO42- which anion
name is sulfate will have the ligand name sulfato.
Anions with names ending in ide have ligand names ending in o for example Cl- which anion name is
chloride will have the ligand name choro.
Examples:
K3[Fe(CN)6]
Potassium hexacyanoferrate(III) (ferrate because metal complex is an anion)
Diamminedichloroplatinum(II)
Pt(NH3)2Cl2
(platinum because metal complex is not an anion)
Tetraaquadichlorochromium(III) chloride
[Cr(H2O)4Cl2]Cl
(Chloro for ligand and chloride for anion)
[Co(NH3)6]Cl3
Hexaamminecobalt(III) chloride
[Fe(C2O4)3]3-
Trioxalatoferrate(III) ion
[Co (en)2 (H2O)Cl] Cl2
Aquachlorobis(ethylenediamine)cobalt(III) chloride
[NH3)5Co-NH2-Co(NH3)4(H2O)]Cl5
Pentraamminecobalt(III)-µ-amidotetraammineaquacobalt(III)chloride
15
mer-Tris(trietthylphosphine)trihydridoruthenium(III)
mer-[Ru{(C2H5)3P}3H3
(curly brackets are sometimes used to avoid confusion)
ISOMERISM
Info from McMurry and Fay, 2012 p824-829 and Housecroft and Sharpe, 2012, p654-661
Isomers are compounds with the same molecular formula but with different arrangements of the
atoms. These different arrangements of atoms leads to different properties like for example
different colours, solubility, melting points and chemical reactivity.
There are two main groups of isomerism which are structural isomerism and stereoisomerism.
Structural isomerisation (also called constitutional isomers): Compound with different metal-ligand
connections. It means that the donor atoms are different.
Structural isomerism group can be further subdivided into ionisation isomers, hydrate isomers,
coordination isomers and linkage isomers. It means that all the donor atoms are the same.
Stereoisomerism: Stereoisomers has different special arrangements and with the same metalligand connections.
Stereoisomerism can be further subdivided into diastereoisomers and enantiomers.
16
Isomers
Compounds with same
formula but different
arragement of atoms
Stereoisomers
Structural
isomers
Compounds with same
connections but
different spacial
arrangements of atoms
Compounds
with different
connections
between atoms
Linkage
isomers
Isomers with
different
metal-ligand
bonds
Coordination
isomers
one metal
complex is the
cation and the
other metal is the
anion. Ligands
swopped around
between metal
centres.
Hydrate
isomers
Ionisation
isomers
Isomers
that yield
different
ions in
solution
placement
of the
water
molecules
differs in
isomers
Diastereoisomers
Geometrical
isomers
Non-mirror-image
isomers
Enantiomers
Optical
isomers
Mirror-image
isomers
17
Structural isomerisation (also called constitutional isomers)
Compound with different metal-ligand connections.
Ionisation isomers
Ionisation isomers are isomers of a complex that differ in the anion that is coordinated to the metal
atom. The square brackets are used to differentiate between ligands and anions. Ligands are placed
inside the square brackets and anions are placed outside the square brackets.
[Co(NH3)5(SO4)]Br
Red colour, SO42- is a ligand and Br- is the anion in the salt
Donor atoms: N and O
[Co(NH3)5Br]SO4
Violet colour, Br- is a ligand and SO42- is the anion in the salt
Donor atoms: N and Br
Take note that these two components have exactly the same molecular formula yet their colours are
very different. Different arrangement of atoms can thus lead to components having different
properties.
The donor atoms of these two complexes are different and therefore they are called structural
isomers.
Think of what will happen when these isomers are placed in a solution. They will give different ions.
Because the give different ions in solution, they are called ionisation isomers.
[Co(NH3)5(SO4)]Br will give the following ions: [Co(NH3)5(SO4)]+ and Br[Co(NH3)5Br]SO4 will give [Co(NH3)5Br]2+ and SO42-
Hydrate isomers
In hydrate isomers the placement of the water molecules differs. The water can either be a ligand,
thus coordinated to the metal or a crystal water which is not directly attached to the metal.
[Cr(H2O)6]Cl3
violet colour Donor atoms: six oxygens
[Cr(H2O)5Cl]Cl2•H2O
light green colour Donor atoms: five oxygens and one Cl
[Cr(H2O)4 Cl2]Cl•2H2O dark green colour Donor atoms: four oxygens and two Cl
The donor atoms of these three complexes are different and therefore they are called structural
isomers.
The water can either be a ligand or it can be a crystal water and therefore these isomers are called
hydrate isomers.
18
Coordination isomers
This type of isomer is typically present when there are two metal ions present. The one metal is part
on a complex to form the cation and the other metal is part of a complex forming the anion. The
two metal ions can be the same type of they can be different.
[Cu(NH3)4][PtCl4]
Cu complex is the cation and the Pt complex is the anion. The NH3 ligands
are bonded to the Cu ion. The Cl- ligands are bonded to the Pt ion
Donor atoms for Cu: four N
Donor atoms for Pt: four Cl
[Pt(NH3)4][CuCl4] Pt complex is the cation and the Cu complex is the anion. The NH3 ligands are
bonded to the Pt ion. The Cl- ligands are bonded to the Cu ion.
Donor atoms for Pt: four N
Donor atoms for Cu: four Cl
The donor atoms of these isomers are different and therefore they are called structural isomers.
The ligands are coordinated to different metal ion centres and therefore they are
called coordination isomers.
19
Linkage isomers
Linkage isomers are isomers were the ligand can bond in different ways to the metal ion. The ligands
can use different donor atoms to link to the metal centre. The ligands are ambidentate ligands.
Please see above a description of ambidentate ligands and how they are named.
Donor atoms for the left hand side complex (nitrito isomer) are: five nitrogens and one oxygen
Donor atoms for the right hand side complex (nitro isomer) are: six nitrogens and no oxygen
The donor atoms of these isomers are different and therefore they are called structural isomers.
Since the ligands are linked differently to the metal ion center, they are called linkage isomers
Stereoisomerism
Stereoisomers has different special arrangements and with the same metal-ligand connections.
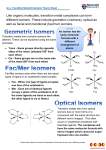
Diasterioisomers (also called geometrical isomers)
Geometrical isomers are non-mirror-image stereoisomers.
Tetrahedral complex has no geometric isomers. If the tretrahedral complex below is rotated by 90°,
it is clear that the two structures are identical.
Square planar complexes and octahedral complexes and have geometric isomers/ diasterioisomers.
A square planar complex Pt(NH3)2Cl2, in the cis isomer the two Pt-Cl bonds will be next to each other
20
orientated at a 90° angle and in the trans isomer the two Pt-Cl bonds will be opposite each other at
a 180° angle. In Latin the word cis means next to and trans means across.
Cis-trans isomers is possible for complexes of the type MA2B4 where the A ligands can either be next
to each other (cis) or opposite each other (trans). Meridional (mer) and facial (fac) isomers are
possible for complexes of the type MA3B3. In the facial isomer, ligands of one type form an
equilateral triangle on one of the octahedron faces.
Cis-trans isomers and mer-fac isomers are examples of geometric isomers also called
diasterioisomers.
In the examples below, the donor atoms of these isomers are the same and therefore they are
called stereoisomers isomers.
21
(Housecroft and Sharpe)
22
Enantiomers (also called optical isomers)
Enantiomers are nonsuperimposable (cannot be placed on top of each other) mirror image of each
other. Enatiomers are also called chiral components. They are like one’s left and right feet, which
are mirror images from each other but they are nonsuperimposable therefore you need a left foot
shoe and a right foot shoe. The same is true for ones hands, eyes and ears.
Enantiomers have identical physical properties like identical melting points, solubilities and colours.
Enantiomers are only different how they interact with plane polarised light.
23
A complex whose solution rotates the plane of polarised light to the right is named dextrorotary and
a prefix d- is used for this complex. A complex whose solution rotates the plane of the polarised
light to the left is named levorotary and a prefix l- is used for this complex.
In the examples below, the donor atoms of these isomers are the same and therefore they are
called stereoisomers isomers.
An equal mixture of enantiomers is called a racemic mixture. Such a mixture has no effect on
polarised light.
Sources consulted
Brown, T.L. LeMay, H.E., Bursten, B.E., Murphy, C.J., Landford, S.J. & Sagatys, D. Chmistry. The
central science: a broad approach. 103-104; 703-716.
Cotton, F.A., Wilkinson, G. & Gaus, P.L. (1987). Basic inorganic chemistry. Second edition. 157-171.
Ebbing, D.D. (1996) General Chemistry. Fifth edition. 993-995.
Housecroft, C.E. & Sharpe, A.G. (2012) Inorganic Chemistry. Fourth edition. p654-656.
Huheey, J. (before 1983) Inorganic chemistry Principles of structure and reactivity. SI Units edition.
p371-416.
McMurry J.E. & Fay, R.C. (2012) Chemistry , six edition. 817-832.
Diagrams and picture obtained from the following sites. These sites shown should not be consulted
for studying purposes since these sources may not be reliable.
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Coordination_Chemistry/Ligands
http://www.chemicool.com/definition/ligand.html
24
http://en.wikipedia.org/wiki/Porphyrin
http://en.wikipedia.org/wiki/Bipyridine
http://classes.uleth.ca/200203/chem38201/Expt2-3820.pdf (good exp)
http://www.mpilkington.com/Lecture_3.pdf
http://www.chem.tamu.edu/rgroup/dunbar/Teaching/CHEM362/Lecture_15-16.pdf
http://en.wikipedia.org/wiki/Bridging_ligand
http://www.sciencedirect.com/science/article/pii/S0079670008000981
http://organometallicchem.wordpress.com/2012/01/10/gee-im-a-tree-predicting-the-geometry-oforganometallic-complexes/
http://cnx.org/content/m34480/latest/
http://en.wikipedia.org/wiki/Coordination_geometry
https://www.boundless.com/chemistry/transition-metals/bonding-in-coordination-compoundscrystal-field-theory/tetrahedral-and-square-planar-complexes/
http://www.3dchem.com/inorganicmolecule.asp?id=579
http://en.wikipedia.org/wiki/Trigonal_bipyramidal_molecular_geometry
http://www.d.umn.edu/~pkiprof/ChemWebV2/Coordination/CN6.html
Questions:
Please see study guide for more questions
1. Cobalt (III) has a coordination number of 6 in all its complexes. Is the carbonate ion acting as
a monodentate or as a bidentate ligand in [Co(NH3)4CO3]+ ion? What is the oxidation state of
the cobalt?
2. Explain what a linkage isomer is and give an example.
3. What is the denticity of the following ligands?
a.
b.
c.
4. What is a chelating agent? Give an example of one.
5. Study the complexes shown below and answer the questions:
25
a.
b.
c.
d.
e.
f.
g.
What is the complex’s coordination number?
What is the name of the complex?
What type of isomerisation is present in the two complexes?
Which complex is the cis-complex?
What is the oxidation number of Pt?
Identify a Lewis base in the complex.
Name a donor atom.
6. You have a complex whose solution rotates the plane of polarised light to the right. What
type of isomerisation is present?
7. What is the difference between stereoisomers and structural isomers?
26