* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chapter 23 – Transition Metals and Coordination Chemistry
Hydroformylation wikipedia , lookup
Cluster chemistry wikipedia , lookup
Metal carbonyl wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Jahn–Teller effect wikipedia , lookup
Metalloprotein wikipedia , lookup
Stability constants of complexes wikipedia , lookup
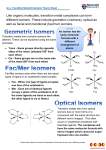
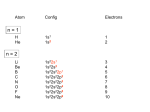
Chapter 23 – Transition Metals and Coordination Chemistry Many compounds of transition metals are colored (used in paints and to stain glass; produce color in gemstones). 23.1 Classification of Solids Most metals, including transition metals, are found in solid inorganic compounds known as minerals. Minerals are named by common, not chemical, names. The properties of the transition metals are similar to each other (and very different to the properties of the main group metals). The similarities in properties come from similarities in valence electron configuration. Electron Configuration For 1st & 2nd transition series = ns2(n−1)dx 1st = [Ar]4s23dx; 2nd = [Kr]5s24dx For 3rd & 4th transition series = ns2(n−2)f14(n−1)dx Some individuals deviate from the general pattern by “promoting” one or more s electrons into the underlying d to complete the subshell – Group 1B Form ions by losing the ns electrons first, then the (n – 1)d Irregular Electron Configurations Due to sublevel splitting, the 4s sublevel is lower in energy than the 3d; and therefore the 4s fills before the 3d. But the difference in energy is not large. Some of the transition metals have irregular electron configurations in which the ns only partially fills before the (n−1)d or doesn’t fill at all; therefore, their electron configuration must be found experimentally. Expected Cr = [Ar]4s23d4 Cu = [Ar]4s23d9 Mo = [Kr]5s24d4 Ru = [Kr]5s24d6 Pd = [Kr]5s24d8 Found Experimentally Cr = [Ar]4s13d5 Cu = [Ar]4s13d10 Mo = [Kr]5s14d5 Ru = [Kr]5s14d7 Pd = [Kr]5s04d10 This leads to some exceptions on the overall trend. Half-filled and filled d subshells are specially stable. Transition metal ions lose e-‘s from s orbital first: Fe: [Ar]4s23d6 Fe2+: [Ar]3d6 Fe3+: [Ar]3d5 1 Fossum-Reyes Atomic Radii Why this trend? 1. Increasing nuclear charge tends to make atoms smaller. 2. However, the strongest (and, therefore, shortest) metallic bonds are found in the center of the transition metals. First these two factors cooperate, but then they counteract each other. Traits of Transition Metals Because most transition metals have only partially occupied d subshells, the metals and/or their compounds often: exhibit multiple oxidation states (vary by 1) Highest oxidation state is the same as the group number for Groups 3B to 7B. Because most transition metals have only partially occupied d subshells, the metals and/or their compounds often: exhibit multiple oxidation states (vary by 1). Are pigmented. Because most transition metals have only partially occupied d subshells, the metals and/or their compounds often: exhibit multiple oxidation states (vary by 1). Are pigmented. Have magnetic properties. Magnetism Diamagnetic substance – all Paramagnetic – at least one are paired (weakly repelled from a magnetic field). is unpaired in the electronic structure (attracted to a magnetic field). 23.2 Transition-Metal Complexes When a monatomic cation combines with multiple monatomic anions or neutral molecules it makes a complex ion. The attached anions or neutral molecules are called ligands. The charge on the complex ion can then be positive or negative, depending on the numbers and types of ligands attached. When a complex ion combines with counter-ions to make a neutral compound it is called a coordination compound. 2 Fossum-Reyes The primary valence is the oxidation number of the metal. The secondary valence is the number of ligands bonded to the metal: – coordination number Coordination numbers range from 2 to 12, with the most common being 6 and 4: [ ( ) ] Swiss chemist Alfred Werner (1893): Some molecules/ions are bonded directly to the metal and are part of the “coordination sphere”. The rest are just counterions (present to balance the charge). Assume a coordination number of 6 for . Table 23.3 The first compound splits into 4 ions: 3 free (not bonded to the metal). The second compound: 2 free . - Test conductivity (# ions it splits into). - Add a ppt reagent to test for # chloride ions (like Compounds 3 and 4 have 1 free ). : different arrangements. Q. Pd tends to form complexes with coordination number 4. A compound has the composition a) What is the correct formula? b) if excess ( ) will ppt per formula unit? ( ) is added, how many moles of . The Metal–Ligand Bond Reaction between a Lewis acid (the metal) and Lewis base (the ligand). The new complex has distinct physical and chemical properties. 3 Fossum-Reyes Metal ions in water are attached to several water ligands: ( ( ) ) ( ) The coordination number depends on the metal and the ligands. Q. What is the oxidation number of the metal in: [ ( Q. A complex contains: ) ]( ) . Formula? Common Geometries Coordination Number Shape Model Example [𝐴𝑔(𝑁𝐻 ) ] 2 Linear 4 Square Planar 4 Tetrahedral [𝑍𝑛(𝑁𝐻 )4 ] 6 Octahedral [𝐹𝑒(𝐻 𝑂) ] [𝑃𝑑𝐶𝑙4 ] 23.3 Common Ligands in Coordination Chemistry Some ligands are polydentate (i.e. take up 2 or more coordination sites on the metal). They are also known as chelating agents. (Table 23.4) Complexes containing polydentate ligands are especially stable. Their formation is entropically favorable: 4 ( ) ( ) ( ) 2 particles 7 particles more disorder 4 can be used to “tie up” free metal ions in solution. (Added to some canned foods salad dressings, etc. Treatment for lead poisoning.) 4 Fossum-Reyes Many transition metals (V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Mo) are components of “metalloproteins” in the body. In the metalloprotein, the metal forms a complex with groups in the protein acting as ligands. Chelates in Biological Systems Common ligand: porphine. Complex of porphine and metal is known as porphyrin. Variations possess diff. metals, diff. groups attached to porphine. This type of complex is a component of myoglobin (stores oxygen), hemoglobin (transports oxygen in blood) and chlorophyl (needed for photosynthesis in plants). The iron in hemoglobin carries O2 and CO2 through the blood. Carbon monoxide and cyanide are poisonous because they will bind more tightly to the iron than will oxygen. Read “The Battle for Iron in Living Systems” (p. 987) 23.4 Nomenclature and Isomerism in Coordination Chemistry 1. Name the cation and then the anion. 2. The name of the complex is one single word. Ligands first (in alphabetical order), and the metal is last. Prefixes are not considered for the alphabetical order. 3. Anionic ligands end in the letter o, but electrically neutral ligands ordinarily bear the name of the molecules; exceptions are: (Table 23.5) For example: [ ( ) ( ) ( ) ] diamminediaquadicyanoiron(III) ion 5 Fossum-Reyes 4. Greek prefixes (di-, tri-, tetra-, etc.) are used to indicate the number of each kind of ligand when more than one is present. If the ligand contains a Greek prefix or is polydentate, the prefixes bis-, tris-, tetrakis-, etc. are used and the ligand name is placed in parentheses. [ ( ) ] tris(ethylenediamine)–cobalt(III) bromide 5. If the complex is an anion, its name ends in -ate. Some metals use their Latin names: Fe ferrate Sn stannate Ag argenatate Cu cuprate Pb plumbate Au aurate [ 4] tetrachlorocobalate(II) ( ) ] 4[ Potassium hexacyanoferrate(II) 6. The oxidation number of the metal is given in parentheses in Roman numerals following the name of the metal. [ ( ) ] Hexaamminenickel(II) bromide [ ( ) ( )( )] Aquacyanobis(ethylendiamine)cobalt(III) chloride [ 4] Sodium tetrachlorooxomolybdate(IV) More Examples [ ( ) ] 4 Hexaaquanickel(II) sulfate [ ( ) ( ) ] Dicyanobis(ethylenediamine)cromium(III) chloride [ ( ) ] Potassium amminetrichloroplatinate(II) [ ] Sodium ethylendiaminetetraacetatoferrate(III) [ ( ) ] Potassium dicyanoargentate(I) 6 Fossum-Reyes Q. Name or give the formula of: )4 ] 1. [ ( 2. 3. [ [ 4] ( ) ( ) ] 4. Lithium tetrahydroxostannate (II) 5. Sodium diaquedioxalatoaurate (III) Isomerism Isomers: Compounds with the same molecular formula, but different structures. 1. Structural isomers: differ in connectivity of atoms. for complexes, 2 subcategories: A. Linkage isomers – the ligand is bound to the metal by a different atom. B. Coordiantion-sphere isomers differ in what ligands are bound to the metal and which fall outside the coordination sphere. ( ) Coordination-Sphere Isomers [ [ [ ( ( ( ) ] ) ] )4 ] 2. Stereoisomers – same order of connections, but different arrangement in space. A. Geometric isomers - the ligands have a different spatial relationship. More polar, more soluble in . Anticancer drug. No anticancer activity. No cis-trans isomers for tetrahedral complexes. 7 Fossum-Reyes Facial – three identical ligands facing each other. Meridional – three identical ligands form an arc around the center. B. Optical isomers: Enantiomers – nonsuperimposable mirror images (like our hands). A molecule that is nonsuperimposable on its mirror image is chiral (a chiral molecule would be superimposable). Common type of chiral molecule: Tetrahedral geometry with 4 different substituents. Square planar compounds have no optical isomerism (since they are flat). Optical isomers rotate polarized light in opposite directions (dextro or levorotatory). 8 Fossum-Reyes Q. What type of isomerism is possible? Draw the isomers. ) ] a. [ ( 4 ) ] b. [ ( 4 ] (square planar) c. [ ( ) 23.5 Color in Coordination Chemistry Transition metal ions show many intense colors in host crystals or solution. The color of light absorbed by the complexed ion is related to electronic energy changes in the structure of the complex: the electron “jumping” from a lower energy state to a higher energy state. The observed color is the complementary color of the one that is absorbed. If something absorbs all ’s of visible light, it looks black. If something absorbs no ’s of visible light, it looks colorless or white. Q. The complex [Ti(H2O)6]3+ has an absorbance maximum at 500 nm, which corresponds to green light. What color is this complex? Aside: Review of orbital shapes and orientations 9 Fossum-Reyes 23.6 Crystal-Field Theory Ligands are assumed to behave like point charges. The ligands exert an electric field, and the energies of the d orbitals on the metal are altered (due to repulsions). In an octahedral field : 6 ligands on axes. d orbitals are degenerate (same E) on the free metal ion. No longer degenerate on the octahedral field. Why does the particular split? Because of repulsions between ligands ( ). charges, d electrons on metal are more stable in orbitals pointing between Less stable in orbitals pointing directly toward ligands ( ). The size of the crystal field splitting energy ( between d levels ), depends on the kinds of ligands and their relative positions on the complex ion, as well as the kind of metal ion and its oxidation state: Strong ligand large splitting (big include CN─ > NO2─ > en > NH3 ) Weak ligand small splitting (small ) ─ ─ ─ ─ include H2O > OH > F > Cl > Br > I─ The colors of complex ions are due to electronic transitions between the split d sublevel orbitals. The wavelength of maximum absorbance can be used to determine the size of the energy gap between the split d sublevel orbitals: The spectrochemical series ranks ligands in order of their ability to increase the energy gap between d orbitals. weak Increasing strong I─< Br─ < Cl─ < F─ < OH─ < H2O < NH3 < en < NO2─ < CN─ < CO CO is a very strong ligand. Poisonous! It binds to cannot get oxygen around). in hemoglobin much more tightly than does (blood The stronger the crystal-field strength of the ligand, the larger the energy gap between d orbitals, and the shorter the wavelength of light absorbed by the complex. 10 Fossum-Reyes Electron Configurations in Octahedral Complexes Recall that when transition metals form cations, s electrons are lost first. Thus, and is a Ion. is a ion, is a ion, If one to three electrons add to the d orbitals in an octahedral complex ion, Hund’s rule applies. (The first three electrons go into different d orbitals with their spins parallel.) We have a choice for the placement of the fourth electron: If it goes into a higher-energy orbital, then there is an energy cost (Δ). If it goes into a lower-energy orbital, there is a different energy cost (called the spin-pairing energy “P” due to pairing with the electron already present). Weak-field ligands tend to favor adding electrons to the higher-energy orbitals (high-spin complexes) because Δ is less than the spin-pairing energy. Strong-field ligands tend to favor adding electrons to lower-energy orbitals (low-spin complexes) because Δ is greater than the spin-pairing energy. For For 4 ions (octahedral field), both high and low spin arrangements are possible. and , only one possible way of arranging in each. Can determine # unpaired experimentally. Paramagnetic – at least 1 unpaired . Diamagnetic – all ’s paired. Q. Out of the following complex ions, one is high spin and the other is low spin: ( ) Which is which? Draw the splitting diagram for each. How many unpaired in each? 11 Fossum-Reyes Another factor that influences crystal-field splitting is the size of d orbitals. Larger orbitals lead to more effective overlap and stronger interactions so 4d and 5d complexes are always low– spin. Tetrahedral Complexes By using the same arguments as for the octahedral case, we can derive the relative orbital energies for d orbitals in a tetrahedral field. Since the ligands interact more strongly with the planar orbitals in the tetrahedral geometry, their energies are raised. This reverses the order of energies compared to the octahedral geometry. All tetrahedral complexes are high spin because of reduced metal orbital – ligand interaction. Square Planar Geometry and Crystal Field Splitting The most complex splitting pattern. Square-planar complexes can be thought of as follows: Start with an octahedral complex and remove two ligands along the z-axis. As a consequence the four planar ligands are drawn in towards the metal. Most d8 metal ions form square-planar complexes ( ). The majority of complexes are low-spin (i.e., diamagnetic). Q. ( )4 is paramagnetic, while ( )4 is diamagnetic. Draw the splitting diagram for each complex. 12 Fossum-Reyes ) ( ) is light blue-green. Would you expect the Q. ( low-spin configuration? ion in this complex to have a high-spin or Q. An octahedral complex of with unknown ligand A looks bluish-green. A complex of with unknown ligand B looks purple. Which is the stronger ligand, A or B? Explain. Draw splitting diagrams. Q. compounds are colorless. Explain why. 13 Fossum-Reyes