* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Energy - Doctor Jade Main
Metalloprotein wikipedia , lookup
Basal metabolic rate wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Electron transport chain wikipedia , lookup
Photosynthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
Biochemistry wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
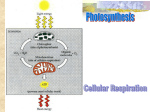
Energy Energy is the capacity to perform work. The First Law of Thermodynamics tells us that energy cannot be destroyed nor created but can be converted from one form into another. Energy flows into the ecosystem as sunlight. This kinetic energy is transformed into chemical energy, in the form of food and fuels by photosynthesis. Animals consume the food products to provide ATP or energy for cells to perform work. Cells release energy by cellular respiration. In this process oxygen is used to convert the chemical energy stored in fuel molecules (glucose) to chemical energy (ATP) the cell can use to carry on its processes. ATP is the molecule that provides energy for cellular work. ATP consists of adenine, a nitrogenous base, ribose, a five carbon sugar and three PO4 groups. Phosphate bonds are unstable in this molecule and can be easily broken by hydrolysis. Each PO4 released from ATP yields 7Kcal of energy. When one phosphate group is removed ATPADP + pi + 7Kcal of energy. When another phosphate is removedAMP + pi + 7Kcal of energy. PO4 groups don’t just fly off; they are transferred to other molecules. This transfer is called phosphorylation. Photosynthesis Photosynthesis is the process by which green plants and some organisms use light energy to convert CO2 + water glucose. Glucose is the basic energy source for virtually all organisms. Plants use much of this glucose as energy to build leaves, flowers, fruits, and seeds. They are referred to as autotrophs or self feeders. Most plants produce more glucose than they can use and store it as starch and other carbohydrates. Humans and other animals depend on glucose as an energy source. They cannot produce it. All living things that do not carry out photosynthesis are consumers or heterotrophs meaning other feeders. Energy is obtained by eating plants or animals that have eaten plants. An important byproduct of photosynthesis is O2. Humans and other animals breathe in oxygen and use it in cellular respiration. Humans are also dependent on ancient products of photosynthesis, the fossil fuels of natural gas, coal, and petroleum for modern industrial energy. Virtually all life on Earth, directly or indirectly, depends on photosynthesis as a source of food, energy, and O2. Photosynthesis requires, CO2 and water. CO2 enters plants via pores called stomata in the leaves. Water is absorbed by roots from the soil and sent to the leaves via a series of veins. Membranes in the chloroplasts (chlorophyll containing organelles) provide the sites for the reactions of photosynthesis. Photosynthesis is a redox process. The equation is: 6CO2 + 12H2O C6H12O6 + 6O2 +6H2O in the presence of light. In a redox reaction there must be both an oxidation and a reduction. In photosynthetic reactions water is oxidized, that is it loses electrons and hydrogen ions while carbon dioxide is reduced to sugar as it gains electrons and hydrogens. These reactions proceed in an uphill fashion. As water is oxidized and CO2 is reduced electrons gain energy by being boosted up an energy hill. Light energy causes electrons in chlorophyll and other light-trapping pigments to boost electrons up and out of their orbit. Hydrogens along with electrons are transferred to CO2sugar and require that H2O is split into H and O2. O2 escapes to the air. Light drives electrons from H2O to NADP+ which is oxidized NADPH which is reduced. There are 2 stages to photosynthesis. Light-dependent reactions, in which chloroplasts trap light energy and convert it to chemical energy, contained in nicotinamide adenine dinucleotide phosphate (NADPH) and ATP. Both of which are used in the second stage of photosynthesis, the light-independent reactions or the Calvin cycle. Light Energy for Photosynthesis The energy of sunlight is called radiation or electromagnetic energy. This type of energy travels as waves. The distance between 2 waves is the wavelength. Light contains many colors, each with a defined range of wavelengths measured in nanometers. This range of wavelengths is called the electromagnetic spectrum. A part of this spectrum can be seen by humans and is called visible light. Light absorbing molecules or pigments are built into plant thylakoid membranes. These pigments absorb some wavelengths of light and reflect others. Plants appear green because the main photosynthetic pigment they contain-chlorophyll, does not absorb green light. It is reflected back. As light is absorbedenergy is absorbed. Chlorophyll A absorbs blueviolet and red light and reflects green. It participates in light reactions. Chlorophyll B absorbs blue and orange light and reflects yellow-green wavelengths and does not directly participate in light reactions. It serves to broaden the range of light a plant can use by sending its absorbed energy to chlorophyll A. Carotenoids are yellow-orange pigments that absorb bluegreen wavelengths and reflect yellow-orange. Some pass their absorbed energy to chlorophyll A and others have a protective function. When a pigment absorbs a photon of light one can say the pigment’s electrons have gained energy. The electrons are excited which is an unstable state. Electrons do not stay in this state. They rapidly fall back to their original orbits. Pigments release absorbed energy from excited electrons to neighboring molecules as electrons fall back to their ground state after being excited by a photon of light. Chlorophyll and other pigments are found clustered next to one another in a photosystem. Energy passes rapidly from one chlorophyll pigment molecule to another. Energy jumps from pigment to pigment until it arrives at the reaction center of the photosystem. The reaction center contains a chlorophyll a molecule sitting next to a primary electron acceptor. The electron acceptor traps a light excited electron from the reaction center chlorophyll and passes it to an electron transport chain which uses trapped energy to make ATP & NADPH. Light-Independent Reactions-Calvin Cycle The energy of ATP and the electrons and hydrogen ions of NADPH made in the light dependent reactions are used in the next stage of photosynthesisthe Calvin cycle. The Calvin cycle takes place in the stroma of the chloroplast. Each step is controlled by a different enzyme. It is a cycle of reactions that makes sugar molecules from CO2 and energy-containing products of the light reaction. Steps of the Calvin Cycle The starting material for the light independent reactions is ribulose bisphosphate (RuBP). The first step is carbon fixation. In this step rubisco (an enzyme) attaches CO2 to RuBP. Next a reduction reaction takes place. NADPH reduces 3-phosphoglyceric acid (3-PGA) to glyceraldehyde 3phosphate (G3P) with the assistance of ATP. To do this the cycle must use carbons from three CO2 molecules. In order to complete the cycle it must regenerate the beginning component-RuBP. For every three molecules of CO2 fixed, one G3P molecule leaves the cycle as the product of the cycle and the remaining five G3P molecules are rearranged using ATP to make three RuBP molecules. The regenerated RuBP is used to start the Calvin cycle again. The process occurs repeatedly in each chloroplast as long as CO2, ATP, and NADPH are available. The thousands of glucose molecules produced in this reaction are processed by plants to produce energy in the process known as aerobic respiration, used as structural materials, or stored. Cellular Respiration The products of photosynthesis are used in cellular respiration where oxygen is consumed (an aerobic process) as glucose is broken into CO2 and H2O. Respiration means breathing but that is not it’s meaning in this context although the processes are related. Cellular respiration refers to an exchange of gases. In cellular respiration O2 is taken from the environment and CO2 is released. The purpose of cellular respiration is to provide ATP for cellular work. The process is an oxidation. It requires oxidization of food molecules, like glucose, to CO2 and water. The overall equation is 6C6H12O2 + 6O2 6CO2 + 6H2O + ATP. Energy is released trapped in the form of ATP to be used for all energy-consuming activities of cells. During this process electrons are transferred from sugar molecules to O2 making H2O. You do not see any electrons transferred in the equation above. But you can see changes in H ions. Glucose molecules lose hydrogen atoms as it is converted to CO2 while O2 gains hydrogen atoms to form water. These hydrogen movements represent electron transfers because each hydrogen atom consists of one electron and one proton. Electrons move along with hydrogens from glucose to O2. Energy is released in the process. Oxidation-Reduction Reactions/Role of Coenzymes Chemical reactions involved in the transfer of hydrogens are oxidationreduction reactions or Redox reactions because for one thing to be oxidized another must be reduced. Oxidation refers to the combing of O2 with other elements. O2 combines with CCO2 +H20 + energy in the form of heat and light. Oxidation also occurs when H+ atoms are removed from compounds. Oxidized things lose electrons. They move to substances that more strongly attract them. The loss of electron is called oxidation. Glucose is oxidized. It loses electrons to O2. One cannot have an oxidation reaction without also having a reduction reaction. O2 is reduced. It gains electrons from glucose. When electrons change partners energy is released. When an electron is lost or a substance is oxidized, it looses energy. When an electron is gained a compound is reduced and it gains energy. As food fuels are oxidized, they loose energy. That energy is transferred to other moleculesATP. These reactions are catalyzed by enzymes which require coenzymes. Enzymes cannot accept H atoms therefore coenzymes act as reversible hydrogen or electron acceptors and are reduced each time a substrate is oxidized. NAD+ or nicotinamide adenine dinucleotide and FAD or flavin adenine dinucleotide are the major coenzymes used in cellular respiration. Oxidation of food energy in the body must be done slowly. There is a step by step removal of pairs of H+ s or pairs of electrons from substrate molecules. Phases of Cellular Respiration Cellular respiration consists of three stages: glycolysis, Kreb’s cycle and electron transport chain. These three are related and occur in the order shown above. Glycolysis begins respiration by breaking down glucose into two molecules of pyruvic acid; a process that takes place in the cytosol of a cell and may or may not use oxygen. The Kreb’s Cycle or citric acid cycle is the complete oxidation of pyruvic acid to CO2 and water and occurs in mitochondria. The electron transport chain or oxidative phosphorylation also takes place in mitochondria. NADH and FADH2 made in the citric acid cycle shuttle their electrons to the electron transport chain where ATP is produced by oxidative phosphorylation. Anaerobic Respiration-Glycolysis The first step of cellular respiration is glycolysis when the six carbon glucose is split into two 3-carbon molecules of pyruvic acid. Glycolysis is sometimes termed anaerobic respiration because it does not need oxygen. The process occurs in all living organisms. Stages of Glycolysis The entire process of glycolysis can be broken into several stages each with several reactions. Stage 1 is a preparatory phase. It consumes energy. In this stage ATP is used to energize a molecule of glucose which is then split into two small sugars that are primed to release energy. This is sometimes termed the sugar activation or the energy investment phase. It uses 2 ATPs and provides activation energy needed to prime later states. Glucose diffuses into cells and is phosphorlyated + ATP catalyzed by hexokinase Glu6PO4 + ADP. In this reaction a PO4 group is added. Glucose 6 Phosphate is isomerizedfructose 6 Phosphate. Fru6P + ATP fructose-1,6-disphosphate (Fru 1,6diP). Fru 1,6 diP + Aldolase (enzyme)2, 3-carbon products glyceraldehyde-3-PO4-. Glyceraldehyde-3 phosphate enters the second stage of the process which yields an energy payoff. Glyceraldehyde-3-P dehydrogenase catalyzes the NAD+ dependent oxidation of glyceraldehyde 3P 1,3 diphosphoglycerate + 2 NADH. In this reaction an H+ is removed and picked up by NAD+ NADH + H+. The NADH will continue on to the electron transport chain. In the third and last stage of glycolysis ATP and pyruvate are produced. 1,3 diphosphoglycerate + ADP3 phosphoglycerate + 2 ATPs. 3 phosphoglycerate2 phosphoglycerate. 2 phosphoglycerate splits off water phosphoenolpyruvate. Phosophoenolpyruvate + ADP2 pyruvate + 2ATPs. The final products of glycolysis: 2 pyruvic acids-C3H4O3 + 4 ATP + 2 NADH + H+ + 2H2O. In total 4 ATPs are made. Remember the net ATP produced is two because for each molecule of glucose two ATPs are consumed. Pyruvate is an important branch point in glucose metabolism. Its fate depends on oxygen availability. Glycolysis requires NAD+ which must be regenerated from NADH to maintain the pathway. During aerobic glycolysis when oxygen is present electrons of cytoplasmic NADH are transferred to mitochondrial carriers of the oxidative phosphorylation pathway generating a continuous pool of cytoplasmic NAD+ and glycolysis continues. When there is not enough oxygen, NAD+ is regenerated by converting pyruvate to lactic acid via lactate dehydrogenase. When O2 is available lactic acidpyruvic acid oxidationenters aerobic pathways-Krebs cycle & electron transport chain. Anaerobic metabolism is limited by the buildup of lactic acid which begins in minutes. A build up of lactic acid disrupts the acid base balance in the body. It degrades athletic performance by impairing muscle cell contraction and produces physical discomfort. Glycolysis is used only for short bursts of high level activity lasting several minutes at most. This process cannot supply ATP for longer, endurance activities. Citric Acid Cycle A specific mechanism transports pyruvate into mitochondria where aerobic processes occur. Inside the mitochondrion, a multi-enzyme complexpyruvate dehydrogenase converts pyruvateacetyl CoA, a two carbon metabolite. This is a major branch point in metabolism. Acetyl CoA can be converted into fatty acids, amino acids or made into energy. The Krebs cycle was named for its discoverer Hans Krebs. It is often called the citric acid or the tricarboxylic acid cycle. The process requires oxygen and therefore is termed aerobic and occurs in mitochondria. The cycle begins and ends with oxaloacetate. The two carbons of acetyl CoA’s condense with the four carbons of oxaloacetate forming a six carbon molecule called citrate. This is the first substrate of the citric acid cycle. The cycle continues around several successive steps in which atoms of citric acid are rearranged producing different intermediates called keto acids. In summary there are two decarboxylations & four oxidations per turn of the cycle. Two CO2, 3 NADH, 1 FADH2, and 1 GTP are produced for each acetyl CoA that enters the cycle. NADH & FADH2 are used to carry electrons to the electron transport chain, where more energy can be harvested from them via further oxidization reactions. Oxidative Phosphorylation/Electron Transport Chain The final stage of cellular respiration is oxidative phosphorylation which involves the electron transport chain, chemiosmosis and takes place on the inner mitochondrial membrane. Oxygen is required. Unlike anaerobic ATP production oxidative phosphorylation produces a tremendous amount of energy. It is the primary method of energy production. The electron transport chain transfers pairs of electrons from entering substrates to oxygen. Oxidative phosphorylation captures free energy released during electron transport and couples it to the rephosphorylation of ADP to make ATP. Electron Transport Chain The electron transport chain consists of a system of electron carriers embedded or built into the inner membrane of the mitochondria. Many compounds making up the electron transport chain belong to a special group of chemicals called cytochromes. Electrons captured in NADH and FADH2 are transported or passed from one compound to the next in the electron transfer chain. As electrons are transferred energy is harvested and stored by forming ATP. Carriers pass electrons from one compound to the next via a series of redox reactions. First the membrane accepts electrons and donates them to the next acceptor with each transfer resulting in the release of a great deal of energy. Pumping of electrons (H+) back and forth across the mitochondrial membrane is key to this process. Hydrogen ions are pumped from where they are less concentrated to where they are more concentrated. This hydrogen ion gradient stores potential energy. ATP synthases built into the inner mitochondrial membrane make ATP as hydrogen ions are driven back across the membrane by the energy of the concentration gradient. Since the membrane is not permeable to hydrogen ions they can only cross via a channel in the membrane. This channel is through the ATP synthase molecule. This process is called chemioosmosis. The synthase attaches phosphate groups to ADPs producing ATP. The last step in the electron transport chain occurs when used up electrons, along with spare hydrogen ions combine with O2, the final acceptor, to form water. The transfer of electrons from NADH + H+ to O2 releases a large amount of energy. It is an exergonic reaction. Energy must be released in small steps so not to loose all energy as heat. Energy Yield All of the steps of glucose oxidation are interrelated. Glycolysis makes pyruvic acid which feeds into the citric acid cycle which makes reducing equivalents (NADH & FADH2) that enter the electron transport chain. Electrons captured in NADH and FADH2 are transported or passed from one compound to the next in the electron transport chain and energy is harvested and stored by forming ATP. For each molecule of NADH approximately 3 molecules of ATP formed, and for each molecule of FADH2, about 2 molecules of ATP are formed. Glycolysis produces 4 ATPs, 2 of which are used in the reaction for a net of 2 ATP. It makes 2 NADH which in the electron transport chain yield 6 ATPs. The citric acid cycle produces 2 GTP (ATP equivalents) and 8 NADH24 ATPs and 2 FADH24 ATPs resulting in 38 ATPs. The complete oxidation of 1 mole of glucose to CO2 and H2O yields a total of 38 ATP molecules. Fermentation Another way certain cells harvest energy from food is fermentation. During fermentation pyruvic acid made by glycolysis is turned into a waste product and two ATP molecules. Formation of the waste product supplies NAD+ needed to carry out glycolysis. Two of the most common types of fermentation are lactic acid fermentation and alcohol fermentation. They are named for the waste products they produce that is either lactic acid or ethanol and CO2.