* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PPT File
Survey
Document related concepts
Transcript
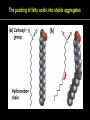
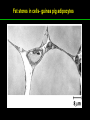
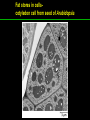
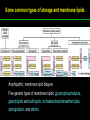
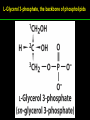
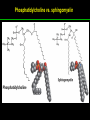
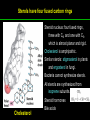
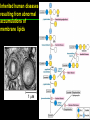
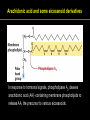
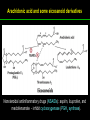
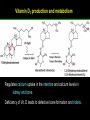
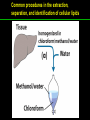
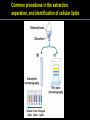
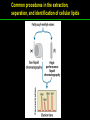
高等生化學 Advanced Biochemistry Lipids 陳威戎 Lipids Lipids 1. Storage Lipids 2. Structural Lipids in Membranes 3. Lipids as Signals, Cofactors, and Pigments 4. Working with Lipids Storage Lipids 1. Fatty acids are hydrocarbon derivatives. 2. Triacylglycerols are fatty acid esters of glycerol. 3. Triacylglycerols provide stored energy and insulation. 4. Many foods contain triacylglycerols. 5. Waxes serve as energy stores and water repellents. Fatty acids are hydrocarbon derivatives Carboxylic acids with hydrocarbon chains ranging from 4 to 36 carbons long (C4 to C36). Unbranched and fully saturated; one or more double bonds Nomenclature: chain length; number and position of double bonds. The most commonly occurring fatty acids: even numbers of carbon atoms in an unbranched chain of 12~24 carbons. The double bonds of polyunsaturated fatty acids are almost never conjugated, and are in the cis configuration. The longer to fatty acyl chain and the fewer the double bonds, the lower is the solubility. In unsaturated FA, a cis double bond forces a kink in the hydrocarbon chain. FA with such kinks cannot pack tightly, and their interactions with each other are weaker, thus having markedly lower melting points. Polyunsaturated fatty acids (PUFAs) The packing of fatty acids into stable aggregates The packing of fatty acids into stable aggregates Triacylglycerols are fatty acid esters of glycerol Simple triacylglycerols: same kind of FA in all three positions. - name after the FA. ex: tristearin, tripalmitin, and triolein. Mixed triacylglycerols: two or more different FAs - specify the name and position of each FA Nonpolar, hydrophobic, essentially insoluble in water. Lipids have lower specific gravities than water. Glycerol and triacylglycerol Triacylglycerol: triglycerides, fats, neutral fats. Triacylglycerols provide stored energy and insulation Oily droplets in the aqueous cytosol of most eukaryotic cells serve as depots of metabolic fuel. - vertebrates: adipocytes (fat cells) - plants: germinating seeds Advantages to using TG as stored fuels: - carbon atoms are more reduced - TGs are hydrophobic and unhydrated In some animals, TGs stored under the skin serve not only as energy stores but as insulation against low temperature. Fat stores in cells- guinea pig adipocytes Fat stores in cellscotyledon cell from seed of Arabidopsis Many foods contain triacylglycerols Most natural fats are complex mixtures of simple and mixed TGs, containing a variety of FAs differing in chain length and degree of saturation. - Vegetable oils: unsaturated FAs, liquid at RT - Catalytic hydrogenation - Animal fats: saturated FAs, greasy solids at RT Lipid-rich foods exposed too long to the oxygen in air: - become spoiled and rancid - oxidative cleavage of double bonds in FAs Fatty acid composition of three food fats Waxes serve as energy stores and water repellents Biological waxes are esters of long-chain (C14 to C36) FAs with long-chain (C16 to C30) alcohols. Melting points (60-100 oC) are generally higher than those of TGs. Chief storage form of metabolic fuel for some free-floating algae. Waxes serve a diversity of other functions related to their waterrepellent properties and their firm consistency. Biological waxes also find a variety of applications in the pharmaceutical, cosmetic, and other industries. Lanolin, beeswax, and wax extracted from spermaceti oil are widely used in the manufacture of lotions, ointments, and polishes. Biological wax Lipids 1. Storage Lipids 2. Structural Lipids in Membranes 3. Lipids as Signals, Cofactors, and Pigments 4. Working with Lipids Structural lipids in membranes 1. Glycerophospholipids are derivatives of phosphatidic acid. 2. Some phospholipids have ether-linked fatty acids. 3. Chloroplasts contain galactolipids and sulfolipids. 4. Archaebacteria contain unique membrane lipids. 5. Sphingolipids are derivatives of sphingosine. 6. Sphingolipids at cell surfaces are sites of biological recognition. 7. Phospholipids and sphingolipids are degraded in lysosomes. 8. Sterols have four fused carbon rings. Some common types of storage and membrane lipids Amphipathic; membrane lipid bilayers Five general types of membrane lipids: glycerophospholipids, galactolipids and sulfolipids, archaebacterial tetraether lipids, sphingolipids, and sterols. L-Glycerol 3-phosphate, the backbone of phospholipids Glycerophospholipids (Phosphoglycerides) Glycerophospholipids (Phosphoglycerides) Ether lipids About half of the heart phospholipids are plasmalogens. Ether lipids A potent molecular signal released from basophils which stimulates platelet aggregation and the release of serotonin from platelets, also exerts effects on liver, smooth muscle, heart, uterine, and lung, and plays an important role in inflammation and the allergic response. Three glycolipids of chloroplast membranes Galactolipids are localized in the thylakoid membranes (inner membranes) of chloroplasts; they make up 70% to 80% of the total membrane lipids of a vascular plant. Three glycolipids of chloroplast membranes Phosphate is often the limiting plant nutrient in soil, and perhaps the evolutionary pressure to conserve phosphate for more critical roles favored plant that made phosphatefree lipids. Three glycolipids of chloroplast membranes Sulfolipids contain a sulfonated glucose residue and bear a fixed negative charge like that of the phosphate group in phospholipids. Archaebacteria contain unique membrane lipids Long-chain (C32) branched hydrocarbons Twice the length of phospholipids and sphingolipids Glycerol dialkyl glycerol tetraethers (GDGTs) Sphingolipids are derivatives of sphingosine Sphingosine (4-sphingenine) Ceramide: structural parent of all sphingolipids Three subclasses of sphingolipids: 1. Sphingomyelins 2. Glycosphingolipids: - cerebrosides and globosides (neutral glycolipids) 3. Gangliosides Sphingolipids Sphingolipids Sphingolipids at cell surfaces are sites of biological recognition Johann Thudichum discovered sphingolipids a century ago and named them after the Sphinx. In human, at least 60 different sphingolipids have been identified in cellular membranes. The carbohydrate moieties of certain sphingolipids define human blood groups. Gangliosides are concentrated in the outer surface of cells, where they present points of recognition for extracellular molecules. Phosphatidylcholine vs. sphingomyelin Glycosphingolipids as determinants of blood groups Phospholipids and Sphingolipids are degraded in lysosomes Phospholipase A series remove one of the two FAs, producing a lysophospholipid. Gangliosides are degraded by a set of lysosomal enzymes that catalyze the stepwise removal of sugar units, finally yielding ceramide. The specificities of phospholipases Sterols have four fused carbon rings Steroid nucleus: four fused rings, three with C6, and one with C5, which is almost planar and rigid. Cholesterol is amphipathic. Similar sterols: stigmasterol in plants and ergosterol in fungi. Bacteria cannot synthesize sterols. All sterols are synthesized from isoprene subunits. Steroid hormones Cholesterol Bile acids Inherited human diseases resulting from abnormal accumulations of membrane lipids Lipids 1. Storage Lipids 2. Structural Lipids in Membranes 3. Lipids as Signals, Cofactors, and Pigments 4. Working with Lipids Lipids as signals, cofactors, and pigments 1. Phosphatidylinositols and sphingosine derivatives act as intracellular signals. 2. Eicosanoids carry messages to nearby cells. 3. Steroid hormones carry messages between tissues. 4. Plants use phosphatidylinositols, steroids, and eicosanoid-like compounds in signaling. 5. Vitamins E and K and the lipid quinones are oxidationreduction cofactors. 6. Dolichols activate sugar precursors for biosynthesis. Phosphatidylinositols in cellular regulation Eicosanoids carry messages to nearby cells Eisosanoids: paracrine hormones Involved in reproductive function; inflammation, fever, pain, blood clotting, regulation of blood pressure, gastric acid secretion, and in a variety of other processes important to human health. All eicosanoids are derived from arachidonic acid, the 20-C PUFA. Three classes of eicosanoids: - Prostaglandins (PG) - Thromboxanes (Tx) - Leukotrienes (LT) Arachidonic acid and some eicosanoid derivatives In response to hormonal signals, phospholipase A2 cleaves arachidonic acid (AA) -containing membrane phospholipids to release AA, the precursor to various eicosanoids. Arachidonic acid and some eicosanoid derivatives Nonsteroidal antiinflammatory drugs (NSAIDs): aspirin, ibuprofen, and meclofenamate - inhibit cyclooxygenase (PGH2 synthase). Prostaglandins (PG) Two groups originally defined: PGE (ether-soluble) PGF (phosphate buffer-soluble) Affect a wide range of cellular functions by regulating the synthesis of the intracellular messenger: 3’, 5’-cyclic AMP (cAMP). Stimulate contraction of the smooth muscle of the uterus. Affect blood flow to specific organs, the wake-sleep cycle, and the responsiveness of certain tissues to hormones such as epinephrine and glucagon. Elevate body temperature (producing fever), cause inflammation and pain. Thromboxanes and Leukotrienes Thromoboxanes (Tx) - produced by platelets (thrombocytes) - act in blood clotting and the reduction of blood flow to the site of a clot. Leukotrienes (LT) - first found in leukocytes - three conjugated double bonds - powerful biological signals - induces contraction of the muscle lining the respiratory tract - overproduction causes asthmatic attacks Steroid hormones carry messages between tissues Steroids: oxidized derivative of sterols - more polar than cholesterol Steroid hormones: - bind to highly specific receptors - trigger changes in gene expression and metabolism Major groups: - male and female sex hormones - produced by the adrenal cortex - antiinflammatory drugs for asthma and rheumatoid arthritis Fat-soluble vitamins Four vitamin groups: A, D, E, K All are isoprenoid compounds synthesized by the condensation of multiple isoprene units. Vitamins A (retinol) and D (cholecalciferol) are hormone precursors. Vitamins E (tocopherol) and K (phylloquinone and metaquinone) and the lipid quinones are oxidation-reduction cofactors. Vitamin D3 production and metabolism Regulates calcium uptake in the intestine and calcium levels in kidney and bone. Deficiency of Vit. D leads to defective bone formation and rickets. Vitamin D3 production and metabolism Vitamin A and its various forms Function as a hormone and the visual pigment of the vertebrate eye. Retinoic acid: - regulates gene expression in the development of epithelial tissue - active ingredient in the drug tretinoin (Retin-A) Retinal: - pigment that initiates the response of rod and cone cells to light - producing a neuronal signal to the brain Retinol: - good dietary sources: fish liver oil, liver, eggs, whole milk, butter - converted from b-carotene Deficiency in Vit. A leads to dryness of the skin, eyes, and mucous membranes; retarded development and growth; night blindness. Vitamin A1 and its precursor and derivatives Vitamins E and K and the lipid quinones are oxidation-reduction cofactors Vitamin E (tocopherols): - substituted aromatic ring with a long isoprenoid side chain - hydrophobic and associates with membrane and lipoproteins - biological antioxidants (the aromatic ring destroys ROSs) - eggs, vegetable oils and wheat germ - laboratory animals fed diets depleted of Vit. E develop scaly skin, muscular weakness and wasting, and sterility - Vit. E deficiency in human is very rare Vitamins E and K and the lipid quinones are oxidation-reduction cofactors Vitamin K: - aromatic ring undergoes a cycle of oxidation and reduction during the formation of active prothrombin - prothrombin: protease, convert fibrinogen to fibrin - deficiency in Vit. K slows blood clotting, rare in human - Vit. K1 (phylloquinone): green plant leaves - Vit. K2 (menaquinone): bacteria in small intestine Vitamins E and K and the lipid quinones are oxidation-reduction cofactors Warfarin: - a synthetic compound that inhibits the formation of active prothrombin - a potent rodenticide - also an anticoagulant drug for surgical patients with coronary thrombosis Ubiquinone (coenzyme Q) and plastoquinone: - electron carriers in the oxidation-reduction reactions that drive ATP synthesis in mitochondria and chloroplast Dolichols: - activate sugar precursors for biosynthesis - anchor sugars to the membrane via hydrophobic interactions Some other biologically active isoprenoid compounds or derivatives Some other biologically active isoprenoid compounds or derivatives Lipids 1. Storage Lipids 2. Structural Lipids in Membranes 3. Lipids as Signals, Cofactors, and Pigments 4. Working with Lipids Working with Lipids 1. Lipid extraction requires organic solvents. 2. Adsorption chromatography separates lipids of different polarity. 3. Gas-liquid chromatography resolves mixtures of volatile lipid derivatives. 4. Specific hydrolysis aids in determination of lipid structure. 5. Mass spectrometry reveals complete lipid structure. Common procedures in the extraction, separation, and identification of cellular lipids Common procedures in the extraction, separation, and identification of cellular lipids Common procedures in the extraction, separation, and identification of cellular lipids Common procedures in the extraction, separation, and identification of cellular lipids Determination of the structure of a fatty acid by mass spectrometry