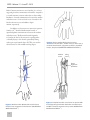
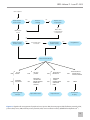
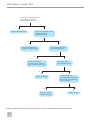
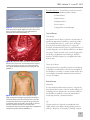
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
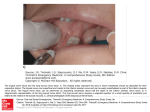
Download Volume 11 Issue R7 Hand:Peripheral Nerves and Upper Extremity
Survey
Document related concepts
Transcript
Volume 11 • Issue R7 HAND: PERIPHERAL NERVES AND UPPER EXTREMITY FUNCTIONAL RECONSTRUCTION Justin S. Chatterjee Reconstructive www.SRPS.org Editor-in-Chief Jeffrey M. Kenkel, MD Editor Emeritus F. E. Barton, Jr, MD Contributing Editors R. S. Ambay, MD R. G. Anderson, MD S. J. Beran, MD S. M. Bidic, MD G. Broughton II, MD, PhD J. L. Burns, MD J. J. Cheng, MD C. P. Clark III, MD D. L. Gonyon, Jr, MD A. A. Gosman, MD K. A. Gutowski, MD J. R. Griffin, MD R. Y. Ha, MD F. Hackney, MD, DDS L. H. Hollier, MD R. E. Hoxworth, MD J. E. Janis, MD R. K. Khosla, MD J. E. Leedy, MD J. A. Lemmon, MD A. H. Lipschitz, MD R. A. Meade, MD D. L. Mount, MD J. C. O’Brien, MD J. K. Potter, MD, DDS R. J. Rohrich, MD M. Saint-Cyr, MD M. Schaverien, MRCS M. C. Snyder, MD M. Swelstad, MD A. P. Trussler, MD R. I. S. Zbar, MD Senior Manuscript Editor Dori Kelly Business Manager Becky Sheldon Corporate Sponsorship Barbara Williams 30 Topics Grafts and Flaps Wound Healing, Scars, and Burns Skin Tumors: Basal Cell Carcinoma, Squamous Cell Carcinoma, and Melanoma Implantation and Local Anesthetics Head and Neck Tumors and Reconstruction Microsurgery and Lower Extremity Reconstruction Nasal and Eyelid Reconstruction Lip, Cheek, and Scalp Reconstruction Ear Reconstruction and Otoplasty Facial Fractures Blepharoplasty and Brow Lift Rhinoplasty Rhytidectomy Injectables Lasers Facial Nerve Disorders Cleft Lip and Palate and Velopharyngeal Insufficiency Craniofacial I: Cephalometrics and Orthognathic Surgery Craniofacial II: Syndromes and Surgery Vascular Anomalies Breast Augmentation Breast Reduction and Mastopexy Breast Reconstruction Body Contouring and Liposuction Trunk Reconstruction Hand: Soft Tissues Hand: Peripheral Nerves Hand: Flexor Tendons Hand: Extensor Tendons Hand: Fractures and Dislocations, the Wrist, and Congenital Anomalies Selected Readings in Plastic Surgery (ISSN 0739-5523) is a series of monographs published by Selected Readings in Plastic Surgery, Inc. For subscription information, please visit our web site: www.SRPS.org. This blank page is included for proper presentation in two-page view. SRPS • Volume 11 • Issue R7 • 2015 HAND: PERIPHERAL NERVES AND UPPER EXTREMITY FUNCTIONAL RECONSTRUCTION Justin S. Chatterjee, MBChB, MSc, EBOPRAS, FRCSGlasg(Plast), FRCSEd(Plast), EurDiplHandSurg(FESSH), DipHandSurg(BSSH), Birmingham Hand Centre, Birmingham, United Kingdom PERIPHERAL NERVES Anatomy A nerve cell is called a neuron. A neuron consists of a plasma membrane surrounding a cell body with characteristic cytoplasmic extensions, the axon (surrounded by a Schwann sheath), and the dendrite. The axon of one neuron communicates with the dendrite or cell body of another neuron through synapses. The axons of peripheral nerves are surrounded by endoneurium and are arranged in groups called fascicles or funiculi, each of which is surrounded by a distinct perineurium (Fig. 1).1 Endoneurium The perineurium is surrounded by a loose areolar intraneural epineurium containing multiple vascular channels. Bundles of fascicles surrounded by intraneural epineurium are enclosed by an outer circumferential epineurium encircling the entire peripheral nerve with its fascicles (Fig. 2).1,2 Circumferential epineurium Perineurium Fascicle Intraneural epineurium A Perineurium around one fascicle Fascicular bundle Perineurium Intraneural epineurium Circumferential epineurium Axon B Figure 1. Nerve fascicle surrounded by perineurium. (Modified from Gutowski.1) Figure 2. Human peripheral nerve. A, cross-section; B, alternate view shows fascicular bundle. (Modified from Gutowski.1) 1 SRPS • Volume 11 • Issue R7 • 2015 The neuroanatomy pertinent to surgical repair includes the following: •• Outer epineurium sutured in epineurial repair •• Inner epineurium sutured in fascicular bundle and perineurial repair •• Perineurium sutured in fascicular repair The vascular supply of peripheral nerves is via arteriae nervorum entering the nerve segmentally and dividing into longitudinal superficial and interfascicular arterioles. The arterioles communicate with each other in a vascular plexus. Extrinsic vessels supply intrinsic longitudinal vessels found in all layers of the epineurium, and the intrinsic longitudinal vessels in turn communicate with extensive capillary plexuses and deep epineurial vessels (Fig. 3).1,3 The longitudinal capillary plexus pattern is duplicated within each nerve fascicle, creating a longitudinal epineurial and a separate longitudinal perineurial vascular system, each with its respective capillary network. Because of the duplication of longitudinal vascular systems at the epineurial and perineurial levels, intraneural dissection for fascicular repair need not devascularize the perineurial longitudinal blood supply. Perineurial plexus Epineurial vessels Although connections do exist, they are not numerous enough to preclude intraneural neurolysis, fascicular repair, or interfascicular grafting. Williams and Jabaley6 emphasized the importance of the internal neural anatomy regarding nerve repair in the hand. Capillary within fascicle Deep epineurial plexus Figure 3. Microvasculature of peripheral nerve. (Modified from Gutowski.1) 2 Peripheral nerves exhibit branching and fascicular plexus formation. In the musculocutaneous nerve of the arm, Sunderland,4 in 1945, identified fascicular branching every few millimeters (Fig. 4, left).1 Many surgeons assumed that pattern occurred at all levels in the forearm and were consequently pessimistic about the chances of successful intraneural dissection and nerve grafting in the upper extremity. In 1980, however, Jabaley et al.5 dissected the median, ulnar, and radial nerves in the forearm and found discrete branches and bundles within the main nerve trunk that could be traced for considerable distances without disturbing adjacent fibers (Fig. 4, right).1 Figure 4. Peripheral nerve fascicular branching pattern according to Sunderland4 (left) and according to Jabaley et al.5 (right). (Modified from Gutowski.1) SRPS • Volume 11 • Issue R7 • 2015 Physiology Signals in peripheral nerves are conducted either by localized potentials or action potentials. Localized potentials occur over short distances, decrease over distance, and are most important in intercellular junctions or sensory nerve endings. Action potentials are conducted impulses that do not decrease over distance. A compound action potential is the net algebraic sum of individual fiber action potentials in a multifiber nerve trunk. Action potentials in unmyelinated fibers progressively excite adjacent inactive areas, and the rate of conduction is directly proportional to the crosssectional area of the axon. In contrast, current flow along myelinated fibers is limited to sites where the myelin sheath is interrupted (i.e., the nodes of Ranvier) (Fig. 5).1 The rapid conduction speeds in myelinated nerves are the result of impulses jumping from one node of Ranvier to the next, a process known as saltatory conduction. Schwann cells exist in two separate groups, one primarily involved with degeneration of myelinated fibers after nerve transection and one responsible for protection and remyelination of regenerating neurites. Schwann cells within the endoneurial space produce new endoneurial tubules or basement membranes,7 whereas those proliferating in the distal stump most likely produce an unidentified trophic substance to the regenerating axonal buds.8 The speed of conduction of an impulse through a nerve depends on the diameter of the nerve fiber and its membrane properties (Table 1).9 The clinical interpretation of electrophysiological recordings requires an understanding of the evoked response, if any; the conduction velocity of the nerve impulse; and the amplitude and shape of the wave form, which can be altered experimentally.9 Terzis and Publicover10 and Van Beek et al.11 reviewed techniques of nerve stimulation and recording in the clinical setting. Wilbourn12 provided a clinical review of electrodiagnostic examination of peripheral nerve injuries, which should be reviewed by surgeons for further information. thoracic outlet known as the brachial plexus (Fig. 6).13 The brachial plexus consists of spinal nerve roots (C5-T1), trunks, divisions, cords, and terminal branches. Several branches are given off at various points in the plexus, and knowledge of their anatomic location is essential in clinical assessment of a patient with brachial plexus abnormality. The terminal branches include the ulnar, musculocutaneous, median, radial, and axillary nerves. The median nerve arises from the lateral and medial cords (C5-T1). In the arm, the median nerve remains in the anterior compartment and passes from lateral to medial over the brachial artery. The ulnar nerve arises from the medial cord (C8-T1). The ulnar nerve commences in the anterior compartment of the arm and passes to the posterior compartment through the medial intermuscular septum. The radial nerve arises from the posterior cord (C5-T1). The radial nerve commences in the posterior compartment of the arm and passes to the anterior compartment through the lateral intermuscular septum. Matloub and Yousif14 reviewed the innervation patterns in the forearm and hand. Unmyelinated axons Myelin Axon neurofibrils Schwann cells Node of Ranvier Unmyelinated fiber Myelinated fiber Nerves of the Upper Limb Brachial Plexus Forearm and Hand The spinal nerves of the upper limb undergo series of unions and divisions between the spinal column and the Figure 5. Unmyelinated nerve fiber compared with myelinated nerve fiber. (Modified from Gutowski.1) 3 SRPS • Volume 11 • Issue R7 • 2015 Table 1 Types of Nerve Fibers9 Fiber Type A Fiber Diameter (mm) Conduction Velocity (m/s) a Proprioception, somatic motor sense 12−20 70−120 b Touch, pressure 5−12 30−70 g Motor to muscle spindles 3−6 15−30 d Pain, temperature, touch 2−5 12−30 < 3−15 B C Function Preganglionic sympathetics dr* Pain, reflex responses 0.4−1.2 0.5−2 S* Postganglionic sympathetics 0.3−1.3 0.7−2.3 *dr, dorsal root; S, sympathetic. Median Nerve In the forearm, the superficial trunk of the median nerve innervates the pronator teres (PT), flexor carpi radialis (FCR), palmaris longus (PL), and flexor digitorum superficialis (FDS) of the index finger. The deep trunk innervates the flexor digitorum profundus (FDP) to the index and middle fingers, flexor pollicis longus (FPL), and pronator quadratus and provides sensation to the radiocarpal joint through the anterior interosseous nerve. The palmar cutaneous branch innervates the skin of the palm and volar wrist. Hobbs et al.15 detailed the origin, course, and distribution of the palmar cutaneous nerve and related the nerve anatomy to the planning of incisions around the palmar aspect of the wrist and palm. The carpal tunnel is a gauntlet of inelastic connective tissue through which pass the median nerve and nine tendons, including the FPL, FDS, and FDP on their way to the distal palm and digits.16 The median nerve travels with the flexor tendons underneath the transverse carpal ligament. At the distal edge of the flexor retinaculum, the median nerve normally divides into six branches: the recurrent motor branch, three proper digital nerves, and two common digital nerves. Steinberg and 4 Szabo16 reported that the motor branch passed through a separate fascial tunnel immediately before entering the thenar muscles in 56% of the specimens studied. The other branches of the radial trunk and the ulnar trunk send common digital nerves to the radial side of the thumb and the first, second, and third web spaces. Sunderland17 reported that in 50% of cases, the median nerve innervated the FDP of the index and middle fingers whereas the ulnar nerve innervated those of the ring and little fingers. In the other 50%, nerve fibers overlapped so much that the median nerve often encroached upon the territory of the ulnar nerve. In the hand, the reverse is true, and the ulnar nerve tends to extend radially into the median nerve territory. The most variable muscles regarding innervation are the thenar muscles, first dorsal interosseous, and third and fourth lumbricals. Martin-Gruber anastomoses are motor connections between the median nerve and the ulnar nerve in the proximal forearm or, more distally, between the anterior interosseous nerve and the ulnar nerve (Fig. 7).14 The connections can be of four types.14,18 Based on a review of the literature, Leibovic and Hastings19 determined the SRPS • Volume 11 • Issue R7 • 2015 frequency of each type of connection and cited a 17% overall incidence of Martin-Gruber anastomoses. Riche-Cannieu anastomoses are motor connections between the median and ulnar nerves in the palm that are present in as many as 70% of people (Fig. 8).14 Three types of connections have been reported. Motor anastomoses are noteworthy in that they can mask the site of injury.20 For example, if both MartinGruber and Riche-Cannieu connections are present, a complete low ulnar nerve lesion might be accompanied by a normal appearing hand because the intrinsic muscles of the hand are innervated by the anterior interosseous branch of the median nerve (Fig. 9).20,21 Likewise, injury to the median nerve at the elbow might result in the loss of all intrinsic muscle functions of the hand, even if the ulnar nerve remains intact. Budak and Gönenç22 used electrophysiological studies to determine the frequency of innervation anomalies in the hand. Martin-Gruber anastomoses were found in 18% of cases, and the finding was bilateral in 73%. Martin-Gruber anastomoses most frequently innervated both the first dorsal interossei muscle and adductor digiti minimi muscle, with the next most frequent being innervation of only the first dorsal interossei muscle, and the third most frequent being innervation of only the adductor digiti minimi muscle. Dorsal scapular n. Lateral pectoral n. Lateral cord Subclavian n. Superscapular n. Posterior cord C4 C5 Medial cord C6 Axillary n. C7 T1 Radial n. Long thoracic n. Median n. Upper Trunks Middle Lower Ulnar n. Lower subscapular n. Thoracodorsal n. Medial brachial cutaneous n. Upper subscapular n. Medial antebrachial cutaneous n. Figure 6. Brachial plexus. n., nerve. (Modified from McCarthy et al.13) 5 SRPS • Volume 11 • Issue R7 • 2015 Riche-Cannieu anastomoses were found in 73% of cases, suggesting that this communication should be accepted as a normal anatomic variation rather than an abnormality. Incidences of neural communication between the median and ulnar nerves of 21% and 14% were observed for the first dorsal interosseous and adductor digiti minimi, respectively. In addition to aberrant motor innervation patterns, Stančić et al.23 described the Berrettini branch, or superficial palmar communication between the median and ulnar nerves. The Berrettini branch frequently is found at the level of the transverse carpal ligament or distally. Injury to this branch, particularly during endoscopic carpal tunnel release (ECTR), can result in altered sensation of the middle and ring fingers. 1 Median nerve 2 3 4 5 6 Ulnar nerve Flexor digitorum profundus 7 Figure 7. Median to ulnar (Martin-Gruber) and ulnar to median nerve anastomoses in the forearm. (Modified from Matloub and Yousif.13) 6 Figure 8. Ulnar to median (Riche-Cannieu) nerve anastomoses in the hand. 1, median nerve; 2, ulnar nerve; 3, recurrent motor branch; 4, opponens pollicis; 5, superficial head; 6, deep head. (Modified from Matloub and Yousif.13) Figure 9. Complete low ulnar nerve lesion can present with normal appearing hand when both ulnar to median nerve and Riche-Cannieu connections are present. (Modified from Mackinnon and Dellon.20) SRPS • Volume 11 • Issue R7 • 2015 Ulnar Nerve Classification In the forearm, the ulnar nerve gives off branches to the muscle bellies of the flexor carpi ulnaris (FCU) and the FDP of the ring and little fingers. It also innervates the ulnar artery by way of the nerve of Henle, which provides sensory branches to the distal forearm and proximal hypothenar eminence. The nerve of Henle was absent in 43% of dissections performed by McCabe and Kleinert.24 Sunderland17 and Seddon26 grouped nerve injuries into five grades according to the disrupted internal structures. Mackinnon and Dellon21 added a sixth degree of injury, which is a mixed injury and likely more applicable in most clinical settings (Table 2). The severity of the injury progresses from minimal derangement of the axon in neurapraxia (first degree) to complete disruption of all neural structures in neurotmesis (fifth degree). In the hand, the ulnar nerve divides at the level of Guyon canal. The deep (motor) branch supplies the hypothenar eminence and midpalm—all the interossei, the two ulnar lumbricals, adductor pollicis, and deep head of the flexor pollicis brevis (FPB). The superficial (sensory) branch of the ulnar nerve supplies the radiocarpal joint and ulnar aspect of the hand, the mid-longitudinal axis of the ring finger, and the entire little finger. The dorsal sensory branch of the ulnar nerve innervates the dorsum of the hand in approximately the same distribution. The palmar cutaneous branch of the ulnar nerve is absent when the nerve of Henle is present.24 Radial Nerve In the arm, the radial nerve gives off the posterior antebrachial cutaneous nerve, which descends along the lateral arm to supply the skin of the dorsal and lateral forearm to the wrist. The radial nerve then divides at the level of the elbow into a superficial (sensory) branch and a deep (motor) branch. The deep branch is known as the posterior interosseous nerve. This nerve gives off branches to the supinator muscle in the forearm and innervates the dorsal wrist capsule. Distal branches innervate all the muscles of finger and thumb extension and provide sensation to the radiocarpal and carpometacarpal joints. The superficial branch of the radial nerve divides into a lateral branch to the radial dorsal aspect of the thumb and a medial branch, which in turn divides into four dorsal digital nerves that supply the thumb, index finger, middle finger, and radial side of the ring finger. Nerve Injuries Peripheral nerve injuries result in loss of sensory, motor, and autonomic functions supplied by the involved nerve.25 McCabe and Kleinert24 indicated that neural injuries can be classified according to their chronicity. Acute injuries are caused by direct trauma, and the onset of symptoms is immediate. Chronic lesions (also known as entrapment neuropathies) are caused by repetitive microtrauma or longstanding compression. With chronic lesions, the onset of symptoms is gradual. The majority of substantial peripheral nerve injuries are chronic. Degree 1 and 2 injuries recover spontaneously with function that is far better than that achieved with surgical reconstruction. Degree 3 injuries also recover spontaneously to a more variable degree, although generally without the need for surgery. Degree 4 and 5 injuries will not recover without repair. Degree 6 injuries recover to variable degrees, depending on the relative degrees of injury involved.27 Ultrastructural Changes In 1850, Waller28 presented his classic experiments on the degeneration of peripheral nerves after transection. Ever since, posttraumatic distal changes in a nerve have been known as wallerian degeneration whereas changes in the proximal portion of a nerve are called axonal degeneration. Conventional wisdom holds that physical disintegration of the axon and myelin sheath extends only a few millimeters proximal to the point of injury. Distally, the axon of the transected nerve undergoes wallerian degeneration, while the sheath of Schwann, endoneurium, and blood vessels remain intact. Schwann cells in the distal stump proliferate into bands of Bungner and form a conduit that helps guide regenerating axons.29 Simultaneously, the proximal nerve cell body enlarges and its metabolism speeds up for 10 to 20 days. Axonal budding begins during that time.17 Peripheral nerve injury also results in substantial neuronal cell death proximally, 7 SRPS • Volume 11 • Issue R7 • 2015 Table 2 Classification of Nerve Injuries21 Histopathological Changes Degree of Injury Myelin Axon Endoneurium Perineurium I Neurapraxia ± II Axonotmesis + + III + + + IV + + + + V Neurotmesis + + + + VI Epineurium + Various fibers and fascicles show various pathological changes in the sensory neurons at the dorsal root ganglion and in the motor neurons in the spinal cord.29 Sensory neuronal death is more marked in distal injuries, and motor neuronal death occurs in association with proximal nerve injuries.30 Diagnosis A complete history of the injury and a precise record of all sensory and motor deficits must be obtained. Terzis31 offered a comprehensive approach to the preoperative evaluation of peripheral nerve lesions. The requirement for careful repeated clinical examination cannot be understated in diagnosing a peripheral nerve lesion and monitoring functional recovery. Investigations should be considered an adjunct. Muscle testing32 and electrophysiological studies33,34 help define the level of injury. Omer32 reviewed testing and grading techniques for patients with peripheral nerve injuries. He reported that the primary indication for electrodiagnostic studies is inability to arrive at a diagnosis. For unexplored closed injuries with suspected nerve involvement, a waiting period of 6 weeks or longer is appropriate before ordering electromyography (EMG) and nerve conduction velocity (NCV) studies. If clinical 8 Tinel Sign Present Progresses Distally − − + + + + + − + − + ± evidence of muscle return is detected in the interim, electrical studies are unnecessary. The EMG and NCV findings in various types of nerve lesions are listed in Table 3.35 Progress is monitored by repeat testing, presupposing a nerve regeneration rate of 1 mm per day (1 in/mo). Sable36 described the use of electrodiagnostic studies in evaluating the median and ulnar nerves at the level of the hand and wrist. Compartment Syndrome The differential diagnosis of peripheral nerve lesions in open wounds of the hand must include compartment syndrome. Clinical distinction between pain and paresthesia from direct nerve trauma or secondary to increased compartment pressure can be difficult. Compartment syndrome develops when tissue pressure progressively builds within a limiting fascial envelope, squeezing the contained neuromuscular structures. As the edema increases, the circulation is compromised37 and the conscious patient experiences agonizing pain. If the pressure is not released surgically, first muscle and then nerve necrosis occurs as tissue pressures reach 30 mmHg. The ischemic changes become irreversible after 6 hours in muscle and after 8 to 10 hours in nerves.21,38 Treatment SRPS • Volume 11 • Issue R7 • 2015 Table 3 Results of Electrical Studies According to Nerve Status35 Electromyogram Fibrillations and Positive Sharp Waves Nerve Conduction Study Voluntary Motor Unit Potentials Sensory and Motor Latency Amplitude Normal Absent Present Normal Normal Complete lesion Present Absent Absent Absent Incomplete lesion Present Decrease in numbers Normal or slightly prolonged Reduced Absent Absent across block, normal above and below block Neurapraxia Absent consists of urgent fasciotomies to relieve pressure in the tissues (Fig. 10).39 Untreated compartment syndrome can result in severe morbidity. Although the diagnosis is essentially clinical, continuous compartment pressure monitoring has been used as an adjunct by some authors.40 Continuous compartment pressure monitoring in the lower extremity has a sensitivity of 94% and specificity of 98% for diagnosis of acute compartment syndrome.41 Factors Affecting Nerve Surgery Both mechanical and biological factors affect nerve regeneration. Neurotropic guidance of axonal regeneration influences the extrinsic environment of the axon. In contrast, neurotrophic enhancement implies an intrinsic change in neuronal metabolism.42 At the nerve trunk level, axonal regeneration is specific and presumably guided by tropic factors.43 Neuronal survival and regeneration might require a continuous supply of neurotropic factors from their target cells.44,45 Nevertheless, as Lundborg et al.46 discovered, a well-organized nerve trunk can regenerate through a preformed mesothelial chamber without preexisting structural guidance as long as the gap is <15 mm. The authors noted strong neurotrophic activity in fluid collected from silicone chambers encasing the stumps of Normal above severed nerves.47,48 In cases in which axonal regeneration of the severed trunk is unlikely,42 ganglioside preparations have been used to stimulate collateral sprout formation from intact nerve terminals. Nerve growth factor (NGF) is a protein that supports cell survival and elicits neuritic outgrowth.44-51 NGF has been isolated from some sympathetic or adrenergic nerves and axons originating from sensory ganglion cells but not from somatic efferent or parasympathetic fibers. Neuronal changes occur after axonal injury, and they switch the neurons from a state of transmission to a growth state, resulting in upregulation of genes involved in cell survival and neurite outgrowth.25 Retrograde transport is also disrupted and provides negative signals to inform the cell body of the disconnection. NGF transport therefore decreases and provides an important negative feedback to the cell body. Artificial application of NGF to axotomized sensory neurons can delay axonal regrowth. Interested readers are directed to an in-depth publication illustrating current concepts in neurobiology of peripheral nerve regeneration.25 Other neurotrophic factors involved in promotion of cell survival include brain-derived neurotrophic factor, neurotrophin 4/5, glial derived neurotrophic factor, ciliary neurotrophic factor, leukemia inhibitor factor, and glial growth factor.29 9 SRPS • Volume 11 • Issue R7 • 2015 Figure 10. Preferred fasciotomy incisions for compartment decompression in upper extremity. (Modified from Rowland.36) Time of Repair The functional results of primary and early secondary nerve repair are equal when conditions are optimal and similar skills apply.52 Primary repair is probably best under the following circumstances: •• For proximal injuries •• In which the distal nerve ends can be pinpointed •• That are minimally contaminated •• Without associated injuries precluding skeletal stabilization or compromising tissue viability or skin coverage •• In an otherwise healthy patient •• When performed by a well-trained, well-rested operative team working with appropriate facilities53 If these conditions are met within 7 days, delayed primary repair seems reasonable; if not, secondary repair is indicated. Nerve stumps are approximated during the initial débridement procedure, and a specific date is set for reexploration and definitive repair. This approach ensures patient compliance and avoids further delay in case minimal return should tempt one to prolong the observation period. Delayed repairs within 6 months of injury yield better functional return than those performed after 6 months.54 10 The zone of injury for a nerve depends on the mechanism of injury: sharp, crush, saw, or avulsion. Avulsion is by far the worst mechanism, as discussed by Zachary et al.55 In their study, the histological injury in nerve avulsions continues to progress along the nerve up to 21 days after injury and requires aggressive resection if immediate reconstruction is performed. The zone of injury can extend up to 2 cm proximally and distally beyond the transection site. Therefore, primary repair is not recommended for injuries with any notable blunt or avulsive component. Waiting for 3 weeks allows the surgeon to define the damaged area of nerve and to resect judiciously based on gross appearance at the time of reconstruction. Resection of injured nerve ends at the time of definitive reconstruction must remove all damaged tissue to ensure regeneration can occur without injured or scarred tissue impeding growth. A conduit or nerve graft might be required to avoid inappropriate tension across the repair, as repair site tension induces additional scarring and decreased nerve regeneration. In other words, use of nerve conduit or graft for a tensionless repair is superior to primary nerve suture under excessive tension.56 The experimental data regarding the best time for suture of sharply lacerated peripheral nerves are contradictory. Cabaud et al.57 recommended repair as early as possible to take advantage of early axonal sprouting and to minimize scarring in the distal endoneurial space. Similarly, Ducker et al.58 and Sunderland17 supported primary or delayed primary repair within the first 10 days. Grabb59 and Kline and Hackett60 reported improved motor reinnervation after primary nerve repair in primates, and Van Beek et al.61 and Hatano62 reported greater end-organ recovery (muscle strength and reinnervated muscle weight) with primary sciatic nerve repair in the rat. Seddon,26 on the other hand, noted that fibrosis at 3 weeks offered mechanical advantages for optimal nerve suturing. Kleinert and Griffin63 recommended nerve repair at 7 to 18 days to coincide with maximal axoplasmic synthesis and to facilitate axonal sprouting across the gap. Citing the same reasons, Holmes and Young64 recommended nerve repair at 2 to 3 weeks after injury. As indicated by Zachary et al.,55 it is preferable to wait 3 weeks if the presence of an avulsive (versus sharp) mechanism of nerve injury is suspected. SRPS • Volume 11 • Issue R7 • 2015 Patient Age Tension of Repair When other variables are the same, functional results of peripheral nerve repair are better in direct correlation with younger patient age. Onne52 noted that for median and ulnar nerve repairs, regained 2-point discrimination (TPD) value in millimeters was approximately the same as the age of the patient, up to 20 years. Between ages 20 and 31 years, recovery was variable but generally poor, and after age 31 years, all sutured nerves showed poor functional recovery, with TPD >30 mm. In contrast, digital nerve repairs in that series showed good results up to age 50 years. Elasticity of neural tissues can be used to advantage to bridge the space between the stumps when a nerve is sutured primarily. Millesi54 stated that human nerve tissue tolerates elongation of 20%, at which point it reaches its elastic limit, beyond which nerve conductivity diminishes. Young et al.65 found useful TPD in 80% of patients younger than 20 years who underwent digital nerve repairs; protective sensation, but no TPD, was achieved by patients older than 40 years. Sunderland,17 on the other hand, noted good functional results in older patients and denied any correlation between patient age and outcome. The excellent function observed in children after nerve repair is ascribed to easier reeducation of central, cortical, sensory, and motor pathways and to a stronger tropic influence of one cell on another. Condition of Wound The degree of intraneural damage (contusion, hematoma, devitalized tissue, infection, scarring) is a factor of the traumatic agent. Severe intraneural damage is reflected at the neuron cell body level, where increased proximal degeneration is present and the regeneration potential is consequently reduced. Traction injuries, high-velocity missile wounds, shotgun wounds, and other types of severe trauma adversely affect the outcome of nerve repair. Level of Injury The more proximal the injury, the worse the prognosis for sensory and motor functional return.17,66 In proximally innervated muscles, recovery is faster and more complete when the nerve fibers are large and localized to the nerve trunk. Crossover during nerve regeneration might not adversely affect function if it occurs between synergistic muscle groups but can play havoc with muscles responsible for precise independent movements, such as the intrinsics. The experimental data are contradictory. Highet and Sanders67 studied the effects of stretching on canine popliteal nerves and noted no permanent elongation but rather long-term damage from continual stresses on the nerves. Likewise, Miyamoto et al.68 showed that tension of 50 g (or stretch >2 cm) impaired transverse anastomoses and later intraneural vascularization of fibular nerves in dogs. In contrast, Rydevik et al.69 working with rabbit tibial nerves, noted that endoneurial capillaries remained well perfused some days after neurolysis despite laceration of regional vessels and mobilization of fascicles for at least 2 cm. Those findings supported the contention held by Smith70 that nerves can probably be mobilized up to 6 to 8 cm and stand as empirical evidence for the existence of a duplicate longitudinal vascular supply at the fascicular level. Vasconez et al.71 noted functional recovery in rhesus monkeys that had 5 to 15 mm of median or ulnar nerve removed. Working with rats, Terzis et al.72 concluded that whereas severe tension predictably diminished functional recovery, mild stretching produced results similar to properly fitted nerve grafts. Gap Size Several authors59,70 have remarked on the worsening results of neurorrhaphy when the gap between the proximal and distal ends of the nerve exceeds 2.5 cm. The major reason for a nerve gap is elastic retraction on both sides of the injury. In the absence of primary repair,73 a fibrotic reaction takes place around the interfascicular epineurium that effectively anchors the nerve ends in a retracted position. The maximum gap that can be tolerated depends on the normal excursion for that nerve. For example, McLellan and Swash74 established that the greatest excursion for the median nerve is 3.5 cm, produced by extension of the wrist and fingers and flexion of the elbow. If the gap after resection of the injured portion is 2 cm, the nerve lacks 1.5 cm to accommodate full excursion and the gap should be bridged. 11 SRPS • Volume 11 • Issue R7 • 2015 Techniques for bridging a nerve gap include direct repair, grafting, nerve transfer(transfer of a redundant nerve or fascicle to reinnervate a critical sensory or motor territory), various tubes and conduits,75 nerve allografts, and tissue expansion.75 The ideal nerve conduit should be readily available, biodegradable, nonantigenic, easily vascularized, and porous enough to facilitate oxygen transport.75 Mackinnon75 reviewed experimental and clinical experience with polyglycolic acid (PGA) (Dexon; Covidien, Mansfield, MA) and Vicryl (Ethicon, Inc., West Somerville, NJ) tubes, autogenous veins, and degenerated muscle grafts in bridging nerve gaps. The authors reported that the best sensory results are obtained with muscle grafts in an acute setting and with PGA tubes if the nerve reconstruction is delayed. The first randomized, prospective, multicenter evaluation of a bioabsorbable conduit for nerve repair was presented by Weber et al.77 In that study, 136 nerve transections were randomized into either a standard repair (end-to-end or nerve graft) or repair using a PGA conduit. At 12 months of follow-up, no significant differences were observed between the two groups. Excellent or good results were obtained in 86% and 74% of the control and PGA conduit groups, respectively. Results were stratified retrospectively to groups with nerve gaps of <4 mm, 5 to 7 mm, and >8 mm, but differences among group sizes made it difficult to draw definitive conclusions. Conduits were superior to “standard” repair in the <4 mm and >8 mm groups but equivalent in the 5 to 7 mm group. The RANGER multicenter study evaluated digital nerve gaps of 5 to 15 mm that were reconstructed with nerve allograft. Ninety-two percent were found to achieve meaningful sensory recovery (grades S3-S4) and were reported to be as good as historical autograft controls. However, statistical significance was not reported.78 Mechanism of Injury Taha and Taha79 evaluated functional outcome and return to work after suture of radial, median, and ulnar nerves injured by low-velocity missiles. The authors found that nerve suture repair of injuries between the wrist and axilla often required supplemental techniques, such as permanent splints, tendon or muscle transfers, or fusions, to achieve good results and expectations for return to 12 work, with the exception of combined median and ulnar nerve injuries. Specifically, supplemental techniques were required after suture repair in 11% of radial, 45% of median, 72% of ulnar, and 100% of combined median and ulnar nerve injuries. After suture repair and supplemental techniques, if needed, patients returned to work in 100% of radial, 55% of median, 57% of ulnar, and 0% of combined median and ulnar nerve injury cases. Surgical Management Frykman et al.35 offered an algorithm for managing peripheral nerve injuries that is predicated on the results of clinical examination and electrodiagnostic studies (Fig. 11). Nerve Repair The goals of nerve repair are to align the severed nerve ends as accurately as possible with the fewest possible number of sutures and to dissect nerve ends only to the extent necessary to achieve proper alignment with minimal tension. Optimal function is possible only if the motor and sensory fascicles of a nerve are precisely and correctly matched. Anatomic, electrophysiological, histochemical, and immunohistochemical methods are now available for fascicular motor sensory differentiation intraoperatively and contribute to good surgical results of primary repair after nerve injuries.80 Nerve repairs are of three general types. To date, comparative analyses have not proved any one type to be superior to the others. The following descriptions are largely excerpted from the review presented by Daniel and Terzis.81 Epineurial Repair—Epineurial repair is the conventional technique for suturing divided peripheral nerves. It is used primarily for small digital nerves that can be aligned with two or three 9-0 or 10-0 nylon sutures. The advantages and disadvantages of epineurial repair are presented in Table 4.81 Perineurial (Fascicular or Funicular) Repair— Fascicular repair was described in 1917 by Langley and SRPS • Volume 11 • Issue R7 • 2015 Nerve is injured Nature of nerve lesion is known Yes Nerve is in continuity No No Repair as soon as appropriate Yes Yes Wait to allow 2.5 cm/month for re-innervation Wait 3 weeks or more Examine for return of muscle activity Yes Re-examine every 1–3 months Progressive return of muscle activity Yes Congratulations No No Electrical Studies Results: EMG [ NCV [ FIBS, PSW NO VMP NO MOTOR NO SENSORY Explore surgically and repair EMG [ FIBS, PSW ↓NO VMP NCV [ AMPLITUDES ↓ SENSORY AND MOTOR LATENCIES NORMAL OR SLIGHTLY ↑ Expect further return EMG [ NCV [ NO FIBS OR PSW VMP ABSENT MOTOR AND SENSORY NORMAL ABOVE AND BELOW BLOCK Expect full return EMG [ NO FIBS OR PSW NO VMP UNLESS PATIENT IS TRICKED NCV [ NORMAL Normal nerve No injury Figure 11. Algorithm for management of peripheral nerve injuries. EMG, electromyogram; FIBS, fibrillation potentials; PSW, positive sharp waves; VMP, voluntary motor potentials; NCV, nerve conduction velocity. (Modified from Frykman et al.32) 13 SRPS • Volume 11 • Issue R7 • 2015 Table 4 Advantages and Disadvantages of Epineurial Repair74 Advantages Disadvantages Short execution time Compromise of precise fascicular alignment Technical ease Sutured epineurium lying in same vertical plane as cut axonal interphases Minimal magnification Tension from normal retraction of cut nerve ends even if no nerve tissue is lost Intraneural contents not invaded Many sutures required to achieve structural integrity of repair Sutures placed only in outer investing sheath Controversial outcome Applicable to both primary and secondary repairs Likelihood of performance by improperly trained personnel Hashimoto.82 The superiority of fascicular over epineurial repair is still in question. Some investigators find no difference between the techniques,65,83-85 whereas others tend to favor perineurial repair over the funicular suture.86-91 Orgel85 stated that suture of the outer epineurium is the technique of choice for most acute nerve lacerations. The author found it to be easier, to be faster, and to require less manipulation of the delicate internal neural structure. Fascicular repair, on the other hand, is more likely to restore usable pathways when branched fiber systems are well localized at the nerve ends. It yields superior results, as judged by morphological and neurophysiological parameters.92 Jabaley93 defined the respective indications for epineurial and perineurial repair. Funicular repair is the technique of choice in nerve grafting and is applicable to nerves with fewer than five fascicles but is contraindicated for multi-fascicled nerves. Theoretical advantages are considerably more myelination of the stumps, better fascicular alignment by coaptation of perineurial tubes, more regenerating axons entering endoneurial tubes of the distal nerve, and greater functional recovery of motor and sensory end organs.82 Disadvantages include longer operative times, increased fibrosis at the suture site, possible vascular compromise of isolated fasciculi, inability to restore funicular continuity on a one-to-one 14 basis, crowding of the suture line with suture material, greater trauma to nutrient vessels at the nerve ends, and inability to approximate very small funiculi.83 In addition, identifying motor and sensory fascicles in the proximal and distal stumps to accurately match the transected ends might not be possible.53 Group Fascicular Repair—Group fascicular repair is possible when a nerve is transected at a level where the branches responsible for different functions are well formed and readily identified within the main trunk. Considering these conditions, motor and sensory components can be accurately coapted to avoid motor sensory cross-innervation. Although histochemical staining can distinguish between motor and sensory axons94-96 to aid in matching the appropriate fascicles, the lengthy process is not clinically practical at this time. Jabaley93 presented a list of specific indications for group fascicular suture at various levels in the upper extremity. Regarding the median nerve, the suitable length of nerve trunk for group fascicular repair extends from approximately 5 cm above the wrist crease to its termination in the palm and for several centimeters below the elbow. In the ulnar nerve, a likely area extends 7 to 8 cm proximal to the wrist crease. SRPS • Volume 11 • Issue R7 • 2015 Repair of Upper Extremity Peripheral Nerve Defects Trumble and McCallister97 described four techniques for achieving direct coaptation of nerve ends when repairing defects in peripheral nerves: nerve stump mobilization, nerve rerouting and transposition, joint positioning, and bone shortening. The authors provided a decision pathway for using these methods of reconstructing peripheral nerve injuries (Fig. 12). In addition to the four methods, the authors described neurotization, which involves embedding the distal end of a nerve directly into the recipient muscle and the use of autologous nerve grafts from adjacent nerves that have been irreparably damaged. Nerve mobilization can reduce tension at the repair site and requires release of constricting fascia, division of mesoneurial attachment, division or dissection of tethering nerve branches, and sometimes epineurolysis. Trumble and McCallister97 reviewed the literature regarding decrease in nerve blood flow as a result of stretching the nerve itself. The authors found a range of 4% to 25% nerve elongation through direct stretching to be safe, based on clinical and experimental studies. Mobilization as described above must be performed judiciously because nerve mobilization, even without stretching the nerve, can decrease nerve vascularity. Maki et al.98 found, in a rabbit sciatic nerve model, that a nerve can be maintained on its proximal intrinsic blood supply after transection and circumferential dissection along a length equal to 63 times its diameter. The nerve could also be supported on a single extrinsic blood vessel with nerve transection both proximally and distally (eliminating the intrinsic blood supply) for a length equal to 45 times its diameter. The results support the clinical use of extensive nerve mobilization to gain additional length for nerve repair in a tensionless fashion. Peripheral nerves can also be rerouted and transposed to decrease tension at the repair site. Anatomic locations in the upper extremity best suited to nerve transposition include the following:97 1.Ulnar nerve at the elbow 2.Recurrent motor branch of the ulnar nerve at the wrist as it enters the carpal tunnel 3.Median nerve at the elbow with release of the lacertus fibrosus, PT muscle, and FDS muscle Positioning of the joints nearest the site of injury results in the greatest reduction of tension across the repair site. As described, correcting joint position postoperatively starts no earlier than 2 to 3 weeks after repair and increases joint motion by 10% each week.97 However, concern exists regarding scarring as a result of tension at the nerve repair site. Nerve grafts or conduits can be used in an acute or chronic setting to avoid tension of nerve repairs in all extremes of joint position at the time of repair.56,99 However the number of axons within the regenerating front that succeed in crossing a neurorrhaphy site reduces with each additional suture line. Therefore, a degree of joint positioning might result in a higher number of axons reaching their target end organ if it allows a direct end-toend nerve repair. Bone shortening in the upper extremity is another technique of bridging nerve gaps.97 Humeral shortening, commonly indicated when multiple nerves have been injured in the setting of a comminuted fracture, facilitates end-to-end repair of both the median and ulnar nerves. Additional indications for humeral shortening include segmental defects of the ulnar nerve in children and adolescents and cases in which concomitant repair of the median and radial nerves is possible. By using these four techniques (nerve mobilization, rerouting and transposition, joint positioning, and bone shortening), Trumble and McCallister97 provided maximum lengths attainable for median, ulnar, and radial nerve repairs. The authors provided a detailed description of the anatomic specifics of the maneuvers. Functional Recovery Nerve Regeneration and the Reinnervation Process When the ends of a severed nerve are approximated, Schwann cell processes projecting from the distal nerve segment accept the progressing axonal buds and serve as conduits to bridge the gap. The regenerating axons are said to grow at a rate of approximately 1 to 3 mm per day through the old endoneurial conduits in the distal segment.17,53,100 According to Huber,101 regeneration of the peripheral end of a severed nerve depends on the outcome of the struggle between the down-growing axis cylinders 15 SRPS • Volume 11 • Issue R7 • 2015 Can the injury be repaired primarily without having to overcome a segmental defect in the nerve? Yes No Proceed with primary repair. Are the end organs (skin, muscle) supplied by the nerve irreparably damaged? Yes No Is the proximal portion of the nerve available for repair? Proceed with neurovascular island flaps or tendon transfers. No Yes Consider nerve transfer (e.g., intercostal nerve transfer to the musculocutaneous nerve). No Is the distal end of the nerve available for repair? Yes Consider neurotization. Can the nerve ends be approximated with mobilization, transposition, bone shortening, or joint positioning? No Bridge nerve defect with intercalary graft. Yes Repair end to end. Figure 12. Algorithm for management of peripheral nerve injuries. (Modified from Trumble and McCallister.93) 16 SRPS • Volume 11 • Issue R7 • 2015 and the developing connective tissue between the severed ends. Experimental studies revealed accelerated axonal regeneration of 4 to 5 mm per day in animal models.57,102-104 Cabaud et al.,57 working with rhesus monkeys, documented remyelination beginning at 1 to 2 weeks; by 3 weeks, it was far distal to the site of injury. Moreover, most new fibers passed down new endoneurial tubules, rarely using old conduits. Wallerian degeneration continued for 6 weeks. The rate and quality of the recovery depend on appropriate reinnervation of the muscles. Progress of the regenerating nerves is measured by Tinel sign, repeated sensory and motor testing, and electrodiagnostic studies. Clinically successful reinnervation depends to a large extent on the proximity of the lesion from its end organ and the denervation interval.31,105,106 Gutmann and Young106 studied the process of muscular reinnervation in the rabbit after variable periods of denervation. The proportion of end plates successfully reinnervated was inversely related to the length of denervation; even so, the authors found intact motor end plates 1 year and more after denervation. The number of end plates returning to function was thought to correspond with the proximity of the nerve injury to the muscle. Brushart and colleagues,91,107 working with rat sciatic nerves, documented reinnervation of peroneal muscles by appropriate motor neurons and by motor neurons that previously served their antagonists. Disappointing functional results, therefore, are not only caused by sensory-motor crossover but also by inappropriate motor reinnervation. Sunderland,17,105 citing cases in which excellent recovery was achieved 12 months or longer after injury, concluded the following: •• Restoration of muscle function requires more than reestablishment of axonal continuity with the terminal endings in the muscle. •• Reinnervated human muscles are capable of regaining almost complete function even after a year of denervation, provided a sufficient number of axons can be directed to their original or functionally similar end organs and the muscle has been maintained in good condition with therapy. •• Any functional deficit remaining after muscle reinnervation is often the result of an insufficient number of reinnervated muscle fibers, fiber mixing at the site of nerve repair, or persisting retrograde and transsynaptic central neuronal changes impairing activity patterns. Lundborg and Rosén108 assessed 54 patients after repair of transected median or ulnar nerves at the wrist level. The best results for recovery of hand sensibility were achieved in patients younger than 10 years. A rapid decline then occurred until age 18 years, at which time a plateau was reached. The authors concluded that the critical age period for regaining functional sensibility after nerve repair ends in the late teenage years. A growing body of evidence supports the role of an intact distal target in promoting nerve regeneration. This is relevant in nerve graft reconstruction, with which a proximal nerve end or donor nerve can be coapted to a nerve graft without a distal recipient neuromuscular unit being present. Such a scenario commonly occurs in cases of cross-facial nerve grafting, nerve pedicle transfer, and contralateral C7 transfer using pedicled vascularized ulnar nerve grafts. Goheen-Robillard et al.109 reported a significant increase in regenerating axons when a nerve graft is coapted to its distal nerve and/or muscle target at the time of proximal graft coaptation. The number of fibers crossing the neurorrhaphy site was double with coaptation to a distal target. The finding was also suggested by Hadlock et al.110 in a facial nerve injury model and by Rab et al.111 in an extremity model. Postoperative Rehabilitation Most authors agree that sensory and motor recovery after peripheral nerve injury, especially recovery to useful function, continues to improve for more than 2 years postoperatively. Immediately after surgery, immobilization for 7 to 10 days is sufficient in most cases.31 Wilgis112 suggested a slightly longer period of immobilization and charted his protocol for rehabilitation of nerve injuries (Fig. 13). 17 SRPS • Volume 11 • Issue R7 • 2015 Rapidly accumulating evidence indicates that a prolonged period of immobilization is not mandatory after digital nerve repair in the setting of an associated flexor tendon repair.113,114 In a cadaver model, the Duran protocol was performed without disruption of nerve coaptations when the nerve gap was 5 mm or less (Figs. 14 and 15).115,116 This is in direct contradiction to the conventional wisdom that dictates 3 to 4 weeks of casting after digital nerve repair.117 Once passive motion of the extremity begins, physiotherapy, direct muscle stimulation, and sensory reeducation are essential for optimal return of function. s ye a r on th 1 m 6 on th m 3 ks w ee 6 3 0 w ee ks s The use of early tactile stimulation after digital nerve repair has been reported by Cheng and colleagues118,119 to provide better sensibility compared with no early tactile stimulation. Tactile stimulation was started 3 weeks after nerve repair using a rotating tactile stimulator disc in addition to standard rehabilitation techniques. Additional innovations for rehabilitation after sensory nerve repair in the hand come from the research conducted by Rosén and colleagues120,121 in Sweden. During the early phase of sensory recovery, as sensation returns to the digits, repeated sessions of forearm anesthesia by topical application of EMLA cream (AstraZeneca, London, United Kingdom) might augment sensation for up to 4 to 6 weeks through rapid cortical reorganization. The durability of this early difference is not certain based on current evidence, considering that four treatments over a 2-week period did not have lasting effects after 8 to 11 months.122 It was recently reported that extension of the cutaneous anesthesia protocol along with sensory rehabilitation over the course of a year allowed reversal of “alexia” for Braille in a blind patient.123 Temporary cutaneous anesthesia of neighboring sensory territories remains a promising modality to augment sensory recovery after nerve repair, although a predictable and tested protocol has yet to be determined. Protection of repair Prevention of secondary deformity Splintage Increased range of motion Increased strength Sensory reeducation: Start when 256 cycles per second recovery and Tinel advancement to end plate Figure 13. Course of rehabilitation after peripheral nerve repair. (Modified from Wilgis.108) 18 SRPS • Volume 11 • Issue R7 • 2015 Figure 14. Mean gap at which disruption occurred for each digital nerve. No statistically significant difference was shown between mean disruption gaps for each of 10 digital nerves. For each nerve, n = 10. R, radial digital nerve; U, ulnar digital nerve within each digit; crosshatched bars, splinted; white bars, unsplinted. (Reprinted with permission from Chao et al.112) Figure 15. Effect of mobilization on nerve repairs and grafts. Percentage of nerves that remained intact for certain gaps after modified Duran and unsplinted protocols decreased with increasing gap. Increased rupture rates for unsplinted nerves at 5- and 7.5-mm gaps were shown to be statistically significant. n = 100 for each bar except as indicated for three bars. Crosshatched bars, splinted; white bars, unsplinted. (Reprinted with permission from Chao et al.112) 19 SRPS • Volume 11 • Issue R7 • 2015 Evaluating the Results by touch. Grading Systems Jerosch-Herold133 argued that to accurately assess the functional outcome of peripheral nerve suture in the hand, the evaluation must “include an additional measure of performance on daily living tasks.” The author’s conclusion was based on a study of the relevance of sensory tests to everyday functional activities. Analysis of the data revealed that the tests of sensibility do not predict patients’ ability to use their hands in everyday activities because patients are able to compensate for sensory deficit through the use of vision and bilateral use of the hands. To compare the published clinical results of peripheral nerve repair, one must rely on standardized, universally recognized, objective criteria. In 1946, Zachary and Holmes124 proposed a system that was based on a scheme presented by Highet and Sanders.67 The scheme was later modified by the Nerve Committee of the British Medical Research Council (Tables 5 and 6).21 The grading scale, although useful, does not fully reflect hand function. For example, an M3 level might be sufficient for effective motor function in some muscle groups but not in others, and the sensory criteria do not include meaningful sensation such as object recognition. Furthermore, MacAvoy and Green125 reported marked limitations in the ability of the Medical Research Council system to discern motor activity in the functional range (Grades 3−5), considering Grade 4 encompasses 96% of potential elbow flexion strength. The other 4% of detectable elbow strength must therefore be distributed among Grades 2, 3, and 5. Although Grade 2 (contraction with gravity removed) generally is not functional in daily use, the level of movement is clinically detectable. Non-obstetric Brachial Plexus Injury Brachial plexus injuries are most common in young men. The majority of injuries are sustained in motor vehicle accidents (traction or crush); less frequently, they occur as sequelae of penetrating wounds, tumors, obstetric trauma, or radiation.134,135 Brachial plexus injuries can occur at several levels134-136 and usually involve multiple structures of the thorax and upper extremity.137 Static TPD tests, such as described by Onne52 in 1962, are best at predicting hand activities that depend on the sensation of something being held between the fingers and correctly assessing the pinch strength needed to maintain grasp. This is a test of innervation density in slowly adapting fibers. In 1978, Dellon and colleague126,127 suggested a moving TPD test and correlated test results with object recognition or tactile gnosis. This is a test of innervation density in rapidly adapting fibers. Evaluation of a patient suspected of having brachial plexopathy must include a detailed medical history, clinical assessment of motor and sensory function in the upper extremity, and determination of the status of the diaphragm, pectoralis, serratus, latissimus, trapezius, and paraspinous muscles.138 To determine suitability of the intercostal nerves for transfer, any history of rib fractures is also elicited. Concomitant injuries to the spine, clavicle, ribs, shoulder girdle, and upper limb are important clues to the energy transfer involved and the likely grade and level. In routine use, the “ten test” described by Strauch and colleagues128,129 is a simple, practical, trackable, reliable, and repeatable examination with which the patient develops a ratio between light moving touch and diminished moving touch on a scale of 1 to 10. It is most likely a test of pressure threshold in rapidly adapting fibers. A more common assessment of pressure threshold in slowly adapting fibers is the use of Semmes-Weinstein monofilaments, which come as a calibrated set of at least five and as many as 20 filaments.130 Moberg131,132 proposed a “pick-up” test that has patients picking up objects from a table and placing them in a small box. The patient is subsequently blindfolded and asked to identify the objects The literature regarding brachial plexus injury is extensive, and a substantial proportion is historical. Contemporary management is determined by several factors, including the level of injury, grade of injury, timing of injury, evidence of neuronal regeneration, and intra- and extraplexal donor nerves available. Regarding level of injury, it is critical to determine whether the injury is pre- or post-ganglionic.139 An avulsion indicates that the spinal nerve has been avulsed from the spinal cord, with no connection between the central and peripheral nervous system, and is termed preganglionic. A rupture describes division of the peripheral nerve, usually caused by tractional force, which can occur at any level of the 20 SRPS • Volume 11 • Issue R7 • 2015 Table 5 Classification of Sensory Recovery21 Grade Recovery of Sensibility S0 No recovery of sensibility in the autonomous zone of the nerve S1 Recovery of deep cutaneous pain sensibility with the autonomous zone of the nerve S1+ Recovery of superficial pain sensibility S2 Recovery of superficial pain and some touch sensibility S2+ As in S2 but with overresponse S3 Recovery of pain and touch sensibility with disappearance of overresponse* S3+ As in S3 but localization of stimulus is good and recovery of 2-point discrimination is imperfect† S4 Complete recovery‡ *Classification modified to include classic 2-point discrimination range: S3 has 2-point discrimination >15 mm. †Classification modified to include classic 2-point discrimination range: S3+ includes 7- to 15-mm range. ‡Classification modified to include classic 2-point discrimination range: S4 includes 2- to 6-mm range. Table 6 Classification of Motor Recovery21 Grade Motor Recovery M0 No contraction M1 Return of perceptible contraction in proximal muscles M2 Return of perceptible contraction in both proximal and distal muscles M3 Return of function in both proximal and distal muscles to such a degree that all important muscles are sufficiently powerful to act against gravity M4 All muscles act against strong resistance, some independent movements are possible M5 Full recovery in all muscles 21 SRPS • Volume 11 • Issue R7 • 2015 brachial plexus and indicates disruption distal to the dorsal root ganglion (DRC) with an intact connection between the central and peripheral nervous system and is therefore postganglionic (Fig. 16).139 Preganglionic lesions have no proximal source of axons for nerve regeneration and therefore preclude repair or interpositional nerve grafting. Postganglionic lesions have a proximal source of axons for nerve regeneration and therefore interpositional nerve grafting can be considered. Level of injury is also anatomically classified as supraclavicular (75%) or infraclavicular (25%), with supraclavicular injuries involving the roots and trunks and infraclavicular injuries involving the divisions, cords, and terminal branches.140 Several other classifications have been published, including that presented by Terzis et al.141 who classified injury levels into supraclavicular preganglionic, supraclavicular postganglionic, and infraclavicular. In 2010, Chuang142 published a further classification of level of injury based on a review of 819 adult brachial plexus injuries operated on by a single surgeon. It should be noted that injury might involve more than one level. Clinical examination can indicate the level of injury based on dermatomal and myotomal neurological findings. Signs of Horner syndrome indicate sympathetic chain disruption and likely avulsion of the lower roots (C8−T1). Computed tomographic (CT) myelography can define pseudomeningocele from a dural tear; however, it is not pathognomonic for root avulsion and not used universally.143 Magnetic resonance imaging (MRI) can be useful when considering root avulsion. It offers the advantages of visualization of the whole brachial plexus and no radiation exposure. However, CT myelography remains the gold standard imaging for assessing root avulsion and is most accurate 3 to 4 weeks after injury.140 EMG and nerve conduction studies are routinely performed.144 They aid in diagnosing, localizing, and defining the extent of axonal loss, but they should be delayed for 3 to 4 weeks because Wallerian degeneration is responsible for the emergence of spontaneous electrical discharge.139,143 Sensory nerve action potentials require continuity between the axon and cell body, which is located in the dorsal root ganglion. Therefore, action Avulsion B Dorsal root ganglion C Stretch A D Rupture Figure 16. A, Transverse section through the cervical spine shows the dorsal root ganglion, dorsal and ventral rootlets, spinal cord, and osseous anatomy. B, With avulsion injuries, the rootlets are torn from the spinal cord. They are preganglionic injuries, as the injury occurs proximal to the dorsal root ganglion. C, If the injury occurs distal to the ganglion, it is postganglionic. Such injuries include stretch injuries (shown in panel C) and ruptures of the nerve (shown in panel D). (Modified from Giuffre et al.135) 22 SRPS • Volume 11 • Issue R7 • 2015 potentials are preserved in preganglionic lesions but not postganglionic lesions or combined pre- and postganglionic lesions. Somatosensory evoked potentials can also help to distinguish between preganglionic and postganglionic lesions.145 Grade of nerve injury has been classified by Sunderland146 and described earlier. From a clinical perspective, it is important to determine whether the injury is a conduction block (neurapraxia) or axonal injury. A conduction block requires no surgical intervention, and full recovery can be expected. Axonal injury results in Wallerian degeneration and prolonged recovery, the extent of which is determined by the distance from injury to end organ and the extent of connective tissue disruption. Three clinical findings can help to determine whether the patient has conduction block or axonal injury: Tinel sign, sweating, and neuropathic pain. Conduction block typically presents with absent Tinel sign, intact sweating (sympathetic supply), and no neuropathic pain. These injuries are common in cases of low-energy trauma. Axonal injury typically presents with Tinel sign, intact sweating, and neuropathic pain. Complete nerve division (rupture) presents with Tinel sign, absent sweating, and neuropathic pain (Table 7). Of note, severe neuropathic shooting pain into an anaesthetic territory is suggestive of a preganglionic injury. Timing of operative intervention is controversial, with a shift to a more proactive approach with earlier intervention.147,148 Timing of nerve injury has substantial impact on management and outcome. Patients often are referred to the Brachial Plexus Unit several weeks after initial injury, at which point suboptimal conditions exist for exploration, resulting in delayed surgical intervention. A recent publication reported that 25% of delayed explorations occurred in patients with injuries that would have been suitable to explore within the first 2 weeks.147 Earlier intervention is currently advocated on the basis of biological, technical, and pain-related factors.148 Birch148 argued that because neurons rely on neurotrophic factors to survive after axonotomy and proximal nerve injuries result in cell death in both the ventral horn and the dorsal root ganglion, reestablishing the nerve cells with neurotrophic factors in the peripheral nerve by early intervention should improve outcome. Some units have designed a protocol system for timing of exploration for brachial plexus injuries (Fig. 17). It is generally accepted that open injuries, vascular injuries, and iatropathic injuries dictate immediate exploration. Patients with adverse features, such as Tinel sign, autonomic dysfunction, and neuropathic pain, might benefit from early surgical exploration (<12 weeks). This strategy allows diagnosis, decompression, and neurolysis and, where indicated, nerve grafting or nerve transfer. Even where an incontinuity lesion exists, decompression optimizes the conditions for axonal regeneration and can greatly improve neuropathic pain. Patients without indication for immediate exploration should be regularly assessed by a peripheral nerve injury service to establish the pattern, grade, and extent of injury, which nerves have potential for spontaneous recovery, and what potential donor nerves are available for transfer. Once a diagnosis of rupture or avulsion is established, early exploration can be performed to confirm the diagnosis and reconstruction can then be accomplished with nerve grafting or transfer. Late intervention (>3 months) is indicated for those cases in which the expected progress has not been achieved, in cases that are not fit for earlier intervention, and in cases that were referred late. Conservative treatment for 3 months is recommended for patients with upper type injuries who have no clinical evidence of preganglionic lesions. Reinnervation of muscle is generally considered necessary by 12 months before irreversible motor end plate degeneration takes place. The timing of intervention depends on the type of procedure and the distance from the injury to end organ. Nerve transfer can convert a high-level injury to a low-level injury such that it can be considered at 10 months.149 However, proximal injuries treated with nerve grafting require intervention at an earlier time point to allow for the longer distance for axonal regeneration to the end organ. In a large series of patients with complete traumatic brachial plexus injuries, Bentolila et al.150 found that operative delay of less than 6 months from time of injury was a significant factor regarding recovery of the function of the biceps (P = 0.003). Ferenz151,152 reviewed the literature of brachial plexus injuries and discussed the current management trends for both acute and chronic injuries of the brachial plexus. Because up to 60% of brachial plexus injuries below the clavicle resolve gradually without medical intervention,153 surgery is postponed 2 to 5 months to 23 SRPS • Volume 11 • Issue R7 • 2015 Table 7 Clinical Signs of Injury Type Injury Type Tinel Sign Sweating Neuropathic Pain Conduction block − + − Axonal injury + + + Rupture + − + −, absent; +, present. await the spontaneous return of nerve function.136,144 The later the operative repair is performed, however, the more difficult the procedure is and the worse the results are.135 Patients should be examined for the availability of both intraplexal and extraplexal donors for reconstruction via nerve transfer and for donor nerve site for nerve grafting. Direct coaptation for the brachial plexus is limited to open sharp injuries. High energies involved in closed traction-type stretch or rupture injuries produces a zone of injury that precludes direct repair. This results in a requirement for functional reconstruction with a focus on reconstruction of key components of upper extremity function by nerve grafting, nerve transfer, tendon transfer, and free functioning muscle transfer. These techniques are discussed in detail later in the monograph. Terzis and Papakonstantinou154 drew on many years of experience with brachial plexus injuries in adults when they suggested the following aims of reconstructive procedures: 1.Stability of the shoulder with focus on return of function in the supraspinatus and deltoid muscles: Shoulder fusion is not advocated. 2.Restoration of biceps function: A hand that has been spared from injury will be useless unless elbow flexion is restored. 3.Reinnervation of the triceps to provide elbow joint stability: Not all authors attempt to regain triceps function, and, 24 in some cases, the triceps muscle can be transferred to provide elbow flexion. 4.Restoration of median nerve function to allow both sensory protection and finger flexion: Sensory protection can be achieved by neurotization of the median nerve from sensory intercostal nerves or from supraclavicular sensory nerves. 5.Lower root involvement: In cases of lower root involvement, it might be prudent to leave banked nerves at the elbow level for future free muscle transplantation for hand reanimation. Terzis et al.141 reported outcomes of brachial plexus reconstruction in 204 patients with adequate follow-up during an 18-year period. Most patients were young (mean patient age, 26 years), and 55% of injuries were caused by high-velocity motor vehicle accidents involving nerve avulsions. Nerve reconstruction included 577 nerve repairs (140 direct neurotizations and 437 nerve grafts). Microneurolysis was performed in 89 cases, and vascularized free nerve grafts were used in 120 repairs. Muscle transfers (29 pedicled and 78 free) were performed to enhance hand function. The results were good or excellent in 75% of suprascapular nerve reconstruction, 40% of deltoid reconstruction, 48% of biceps reconstruction, 30% of triceps reconstructions, 35% of finger flexion reconstructions, and 15% of finger extension reconstructions. The majority of patients experienced protective sensation and pain relief postoperatively. SRPS • Volume 11 • Issue R7 • 2015 Immediate • Open injuries • Vascular injuries • Latropathic injuries Early (<12 weeks) • Closed injuries with adverse features • Associated injury needing intervention • Evidence of avulsion and/or rupture clinically Late (>12 weeks) • Not fit initially • Failure to make expected progress • Late referral Figure 17. Birmingham peripheral nerve injury service protocol. Early is defined as within 12 weeks of injury. Late is defined as more than 12 weeks after injury. Complications of Nerve Injury Neuromas occurs at the site of the nerve injury or if partial injury exists, painful sequelae can develop because a portion of the nerve is transmitting relatively normal impulses and the remainder of the nerve is transmitting abnormal impulses. A pragmatic approach to neuroma prevention in patients with irreparable nerve division is simple resection to allow the cut ends to retract proximally into healthy soft tissue.155 If the soft-tissue bed is not adequate, the nerve is transected proximally where local trauma can be minimized. Neuromas can occur after injury or surgical repair. They can be classified according to their site: lateral, incontinuity, end, or amputation stump neuromas. Conventionally, neuromas are divided into those that occur after complete transection and those that occur after partial transection. Complete Transection The management of painful neuromas is controversial. In the event of an established, painful neuroma that cannot be repaired primarily, en bloc relocation to an unscarred, protected site is preferred.156,157 Alternatives are crushing,158 epineurial closure and ligation,159 transposition (either in-continuity or after resection),112 relocation of the intact neuroma,157 and resection and proximal nerve end implantation into muscle or bone.160 Neuromas behave unpredictably, particularly those forming in proximal peripheral nerves. Patients’ activities of daily living and work can be restricted because of the severity of the symptoms. Neuromas are also difficult to diagnose because of the fascicular arrangement of proximal nerves. Mackinnon and Dellon21 stated that when a peripheral nerve is divided, nerve fibers in the proximal nerve stump form regenerating units that contain numerous axon sprouts. If the sprouting fibers make appropriate sensory or motor distal connections, functional recovery occurs. If they do not make appropriate distal connections, loss of function occurs. Pain can develop at the level of the thwarted regeneration. Physical and chemical measures, such as freezing, cauterization, electrocauterization of the nerve stump, silicone-rubber capping, and corticosteroid injection, have been tried with variable success.161-163 Martini and Fromm164 described managing 68 painful neuromas by shortening the fascicles and sealing the epineurial cuff with Histoacryl topical skin adhesive (B. Braun Medical, Inc., Bethlehem, PA) to plug the nerve ends and prevent escape of regenerating axons. All except three of 36 patients were cured or improved. Per Mackinnon and Dellon,21 the inevitable consequence of complete division of a peripheral nerve is formation of a neuroma at the proximal site of the nerve division if the neuron’s attempt to regenerate distally is thwarted. Not all of these neuromas are painful. No consensus exists regarding why one neuroma is clinically symptomatic and another is not. If partial regeneration Novak et al.165 surveyed 70 patients to evaluate the long-term subjective outcome of surgical treatment of upper extremity neuroma. In all cases, the neuromas (n = 112) were resected and the involved nerve was transposed into proximal muscle away from overlying scar and denervated skin. Forty-five patients (64%) reported good pain relief and were able to return to work after surgery. 25 SRPS • Volume 11 • Issue R7 • 2015 No significant difference was observed in functional outcome that could be related to patient sex, postinjury time, follow-up time, number of previous operations, or site of nerve injury. Mackinnon and Dellon166 offered an algorithm for the surgical management of painful neuromas (Fig. 18). Partial Transection When no definite evidence of nerve severance is present, a 1-month waiting period for evidence of spontaneous recovery is reasonable. Serial examinations of the injured extremity to include Tinel test and electrodiagnostic studies (nerve conduction velocity and evoked response) should be correlated with the clinical picture to determine whether and at what point exploration is necessary. The findings at exploration will determine the course of action, as follows:81 •• In the event of residual sensory and motor function and a lateral neuroma, care should be taken not to convert a partial lesion into a complete one. Management is by appropriate resection and repair of the disrupted perineurium of the involved fascicles only. The resulting gap determines whether nerve graft is indicated. •• Where exploration reveals a spindleshaped neuroma and fascicular continuity, intraneural neurolysis (not resection and repair) should be performed regardless of the functional loss. •• Spindle-shaped neuromas associated with complete transection and no sensory or motor return should undergo subsequent resection and coaptation at the site of injury. Woodhall et al.167 noted significant functional recovery in 75% of patients with nerve injuries who had initial paralysis but no overt nerve severance. Evidence of regeneration was delayed beyond the expected time in half the patients, and those cases were subsequently explored. In that group, nerve regeneration and eventual recovery was better when the neuroma was not excised and the nerve not sutured. Because motor end plates tend to disintegrate after denervation and to lose most of their 26 reinnervation potential with time, 12 months should be considered the outer time limit for surgical intervention in suspected neuromas of the motor system. Certain anatomic sites are common for neuroma formation, including superficial radial nerve or branches, Palmar cutaneous branch of the median nerve, and digital nerves after amputation. Nonsurgical management should be considered in the initial treatment of neuromas and includes physical therapy, desensitization, and pharmacological treatment (gabapentin or pregabalin).168 A recent review advocated dividing surgical management into three categories:168 1.If appropriate distal nerve and sensory receptors are available, nerve grafting can direct regenerating nerve fibers from proximal to distal segment. 2.If a distal nerve is not available but critical function is required distally, free functioning neurocutaneous flap can be used to accept regenerating sensory nerve fibers in the proximal segment. 3.If the local environment is not suitable for nerve grafting or if multiple previous operations have been performed, resection of the neuroma and transposition of the proximal stump into muscle, vein, or bone is advised. Centro-centralization technique involves suturing two nerve fascicles in the proximal segment to each other and has shown clinical success. Autologous tissue wrapped around the nerve has also been used to help provide a well-vascularized tissue with the aim of producing a gliding surface and preventing fibrosis, adherence, and irritation to the peripheral nerve. Elliot169 reported complete resolution of all modalities of pain in eight of 14 patients undergoing teno-neurolysis and fascial nerve wrap. The author recommended the use of local flaps, including the Becker fasciocutaneous flap and the anterior forearm fascial flap. This usually is reserved for large forearm nerves (median or ulnar) in continuity to treat pain rather than distal sensation.169 SRPS • Volume 11 • Issue R7 • 2015 Arm Wrist Forearm Neuroma resection Bone Critical sensation Nerve graft Noncritical Brachioradialis muscle Distal nerve stump available Nerve graft No distal nerve Provide innervated free tissue Distal nerve available Nerve graft No distal nerve Bone Critical sensation Hand Noncritical Lower extremity Critical sensation Nerve graft Bone Noncritical Soleus muscle Figure 18. Surgical algorithm for management of painful neuroma. (Modified from Mackinnon and Dellon.161) Complex Regional Pain Syndrome Complex regional pain syndrome (CRPS) is a contemporary term to replace numerous historical terms including algodystrophy, algoneurodystrophy, Sudeck atrophy, reflex neurovascular dystrophy, causalgia, reflex sympathetic dystrophy, shoulder-hand syndrome, and fracture disease. For the sake of clarity, the term CRPS is used to replace previous terminology in much of the following section. Here is a history of CRPS based on reviews presented by Schutzer and Gossling,170 Wilder et al.,171 and Inhofe and Garcia-Moral.172 The history of CRPS began in 1864, when Mitchell and colleagues reported the sequelae of nerve injuries in victims of the Civil War and called the disorder causalgia. Sudeck, in 1900, described refractory pain, swelling, stiffness, vasomotor instability, trophic skin changes, and osteoporosis of the affected extremity. Hence Sudeck atrophy is the term used to describe demineralization of the bone. Lankford and Thompson categorized CRPS as either minor causalgia, which involves sensory nerve injury alone, and major causalgia, which involves mixed nerve injury. This somewhat confusing classification is not currently advocated.170-172 In 1991, the American Association for Hand Surgery appointed an ad hoc committee to define the disorder known as reflex sympathetic dystrophy (RSD) and to make recommendations regarding its diagnosis and management. Much of this summary of RSD is taken largely from that committee’s report173 and from reviews conducted by Schutzer and Gossling,170 Wilder et al.,171 and Inhofe and Garcia-Moral.172 According to the ad hoc committee of the American Association for Hand Surgery,173 reflex sympathetic dystrophy was the term given to a complex syndrome of posttraumatic pain. The pain typically is accompanied by loss of function, and evidence exists of “autonomic dysfunction” in the affected limbs. Based on the evidence, committee members thought that a more appropriate name for RSD would be sympathetic maintained pain syndrome. Traditionally, three diagnostic criteria were required for the diagnosis of RSD of the hand: 1) diffuse, intense, or unduly prolonged pain; 2) delayed functional recovery with some degree of loss of hand mobility; and 3) sympathetic dysfunction manifested by vasomotor 27 SRPS • Volume 11 • Issue R7 • 2015 disturbances and various associated trophic changes.170,173 Both over-diagnosis and under-diagnosis commonly occurred. The cause of CRPS is unknown. Most researchers think that chronic irritation of the peripheral sensory nerve(s) causes afferent input, resulting in continuous stimulation of sympathetic and motor efferent fibers. The altered sympathetic activity produces subsequent motor tone changes that culminate in CRPS. Some authors think that patients who are emotionally labile and have a low pain threshold are predisposed to CRPS, which, in those persons, might have a psychogenic origin. Other investigators think an excessive inflammatory response is behind the development of CRPS.174 To test that hypothesis, Oyen et al.174 obtained scintigrams by using indium-111-labeled immunoglobulin G in 23 patients with a diagnosis of CRPS of the hand. The results suggested that a flow-independent inflammatory component characterized by increased vascular permeability for macromolecules plays a role in the development of CRPS. Veldman et al.175 surveyed the signs and symptoms of CRPS in 829 patients. In its clinical presentation, CRPS exhibits three phases: 1) an initial warm phase for the first 2 to 3 months, 2) a phase of vasomotor instability that can last for several months, and 3) a terminal cold end phase.176 During its early phase, CRPS is characterized by pain and regional inflammation that is exacerbated by exercise. Both hypo- and hyperesthesia are present in up to 75% of patients. During the second and third phases, tissue atrophy, involuntary movements, muscle spasms, and pseudoparalysis can occur. Tremors and muscular incoordination are noted in approximately half the patient population. Sympathetic signs are infrequent and have no diagnostic value. The authors noted that the early symptoms of CRPS are those of an exaggerated regional inflammatory reaction and not those of a disturbance of the autonomic nervous system. A three-phase radionuclide bone scan is highly sensitive and specific in the diagnosis of CRPS.21 Heerschap et al.176 conducted a nuclear MRI study of the skeletal muscles of the leg in 11 patients with CRPS. The authors reported that the average tissue pH of the muscles of affected legs was significantly increased over that of control legs, as was the average inorganic 28 phosphate:phosphocreatine ratio of the muscles. The findings were in agreement with those of previous studies documenting reduced oxygen extraction and impairment of high-energy phosphate metabolism in cases of CRPS. Because the pathogenesis of CRPS is unclear, treatment is perforce empiric. Nonoperative measures include elevation of the extremity, physical therapy, transcutaneous electrical nerve stimulation, steroids, local and regional anesthetic blocks, and administration of a variety of drugs such as antidepressants, anticonvulsants, nonsteroidal antiinflammatory agents, vasodilators, calcitonin, and beta-adrenergic antagonists. Surgical management consists primarily of cervical sympathectomy.170 Watson and Carlson177 developed a program of progressive “stress loading” for treatment of CRPS in the hand, consisting of two simple activities called scrub and carry. The authors avoided the use of all other modalities, including immobilization and aggressive joint mobilization, until the pain began to subside. The patients exhibited improvement in pain, vasomotor activity, edema, motion, and strength, with 95% returning to normal activities and 84% returning to their pre-CRPS occupations. Although conservative treatment is said to be successful in approximately half of all cases of CRPS, a survey by Inhofe and Garcia-Moral172 showed that the long-term results are not so encouraging. Five years or more after diagnosis, a significant number of patients reported worsening of symptoms (56%), a negative impact on their daily lives (78%), and either a downgrading of their jobs or unemployment as a direct result of their condition. Wilder et al.171 presented an excellent review of CRPS in children, which has become increasingly common and is thought to be precipitated by the stress of sports activities. Appropriate treatment requires a multidisciplinary approach and consists primarily of noninvasive regimens, such as active physical therapy, transcutaneous electrical nerve stimulation, and biobehavioral intervention. Adjunctive sympathetic blocks are highly successful in relieving symptoms and accelerating the rate of mobilization. CRPS has become the preferred descriptor for SRPS • Volume 11 • Issue R7 • 2015 pain syndromes.178 The name was developed at the 1993 consensus workshop of the International Association for the Study of Pain. The new terminology emphasizes the current understanding that sympathetic dysfunction might not be present in every case of chronic pain. Two subgroups are defined, with Type I developing after minor or orthopaedic trauma without any detectable nerve injury and Type II occurring after a recognizable nerve injury. Type I corresponds to RSD, whereas Type II was traditionally called causalgia. In 2011, The Royal College of Physicians published guidelines for the diagnosis, referral and management of CRPS.179,180 CRPS is defined as “a debilitating condition, characterised by pain in a limb, in association with sensory, vasomotor, sudomotor, motor and dystrophic changes.” Diagnosis is undertaken using the Budapest criteria, and readers are directed to this guideline for a comprehensive contemporary approach to treatment, which is based on “four pillars” using an interdisciplinary approach: 1.Patient information and education 2.Pain relief (medication and procedures) 3.Physical and vocational rehabilitation 4.Psychological intervention Functional Reconstruction of the Upper Extremity Reconstruction of function in the upper limb after peripheral nerve injury should be undertaken using a systematic approach in a manner similar to the reconstructive ladder for soft-tissue reconstruction. A number of procedures are available depending on the timing of the injury, nerve recovery, and expendable donor muscle-tendon units, nerve fascicles, or muscles available. This armamentarium can be considered a functional reconstructive ladder (Table 8, Fig. 19). Ascension of the ladder involves interventions of greater complexity. Nerve Grafting Mackinnon and Dellon reviewed the history of nerve grafting, which they traced to Philipeaux and Vulpian in 1817. The modern era of nerve grafting dates to experiments conducted by Millesi and colleagues181-185 during the early 1970s. The authors recommended nerve 21 autografts for any gap >2 cm and, despite adverse surgical conditions, obtained excellent functional results in lesions of the radial, median, and ulnar nerves. Since those reports were published, nerve grafting has become accepted as a clinically feasible alternative to primary repair. Nerve grafting can be considered only when a proximal source of axons is available and a distal organ with the potential for full functional recovery is present. The following are highlights of the technique presented by Millesi185 for interfascicular nerve grafting: •• The fascicles are dissected both proximal and distal to the lesion in normal unscarred tissue. •• Not every single fascicle is dissected; rather, the dissection involves the connective tissue between groups of fascicles. •• When in doubt, resect; only healthy looking tissue should be preserved. •• Nerve junctures should be placed at different levels along the fascicular groups. •• A drawing of the cut surfaces helps in matching the stumps. •• Autografts are harvested as single strands and joined to fascicular groups of corresponding diameter. •• The grafts are cut slightly longer than the defect to be bridged. •• Only one or two 10-0 nylon stitches are needed to secure each fascicle or fascicular group. •• Each suture incorporates the perineurium of the fascicle of the proximal and distal stump and the epineurium of a single nerve graft. •• Four or five autografts are necessary for the ulnar and radial nerves and five or six for the median nerve. Recovery was more complete when the nerve grafts were performed between 6 and 12 months after injury, but the relationship between graft length and function was less clear. The sural nerve was the most common source of autograft, and the medial antebrachial cutaneous nerve was the next most common. 29 SRPS • Volume 11 • Issue R7 • 2015 Table 8 Upper Extremity Functional Reconstructive Ladder Complexity Procedure 4 Free functioning muscle transfer 3 Tendon transfer 2 Nerve transfer 1 Nerve grafting Figure 19. Upper extremity functional reconstructive ladder. FFMT, free function muscle transfer. Nunley et al.186 reported grafting the anterior branch of the medial antebrachial cutaneous nerve for the repair of defects of the digital nerve in 21 cases, including all fingers and thumb. Indications for nerve grafting were a gap in the digital nerve of >1 cm or excessive tension needed to oppose the severed nerve ends. At an average follow-up of 57 months, all except one finger had recovered the ability to distinguish between sharp and dull stimuli and all except three had TPD values between 5 and 15 mm. No painful neuromas were evident at the donor site. 30 Although grafts from sensory nerve donors have been the norm, a growing body of experimental evidence supports the use of nerve grafts from motor nerve donors for reconstruction of motor and mixed motor and sensory nerves.187 This concept has been termed preferential motor reinnervation.188 The difference seems to be a combination of neurotrophic,188 cellular,189 and physical190 factors that facilitate axonal regeneration across motor nerve grafts when compared with sensory nerve grafts. Although motor nerve grafts have traditionally been considered nonexpendable, the common use of muscle flaps in microsurgery indicates that some muscles, and thus SRPS • Volume 11 • Issue R7 • 2015 some motor nerves, are expendable when viewed from a different paradigm. Examples include the motor branch of the obturator nerve to the gracilis and the descending or transverse branch of the thoracodorsal nerve. In 1976, Taylor and Ham191 transferred a free vascularized nerve graft of the radial nerve to bridge a defect of the median nerve. Eight years later, Bonney et al.192 presented a report of 30 patients with brachial plexus injuries treated by vascularized ulnar nerve grafts. Koshima and Harii193,194 experimentally showed a significantly greater number of large-caliber, myelinated axons and earlier regeneration (4 versus 8 weeks) of vascularized nerve grafts compared with standard nerve grafts (P < 0.05). Breidenbach and Terzis195 noted that vascularized nerve grafts might be of benefit in scarred recipient beds, in proximal nerve lesions, in large nerve gaps, or as carriers for nonvascularized nerve grafts. Rose and Kowalski100 described features of the deep peroneal nerve-dorsalis pedis artery complex that make it ideal as a donor of segmental vascularized nerve grafts for digital sensory nerve reconstruction. Townsend and Taylor196 presented a discussion of the use of composite arterialized neurovenous systems in selected clinical cases. In 1982, Chiu et al.197 presented experimental evidence of nerve regeneration through a segment of autogenous interpositional vein graft. Eight years later, Chiu and Strauch198 evaluated their clinical results achieved with the technique in the repair of 15 palmar digital nerve gaps. Although all patients reported symptomatic relief and satisfactory sensory return, TPD values indicated superiority of direct repair and conventional nerve grafts over the vein graft technique. Nevertheless, the authors reported that autogenous vein grafts as nerve conduits are effective when selectively applied to bridge small (<3 cm) gaps in nonessential peripheral sensory nerves. Bain199 reported the use of peripheral nerve allotransplantation in seven patients. Six achieved good sensory recovery, three experienced motor recovery, and one had no recovery because of rejection. All patients required immunosuppression with combinations of cyclosporin A, azathioprine, prednisone, and FK506. Immunosuppression was withdrawn after Tinel sign progressed into the distal segment of the reconstructed nerve. No clinically significant complications of systemic immunosuppression were reported; however, the allograft was rejected in one patient. No patient experienced deterioration of nerve function after withdrawal of immunosuppression, and no evidence of chronic rejection was observed. Wyrick and Stern200 reviewed secondary nerve reconstruction and discussed alternatives to nerve grafting, such as tubulization (insertion of conduits for the passage of axons), elongation with tissue expanders, and direct muscular neurotization. Most brachial plexus injuries were previously managed primarily by nerve grafting.134-136 Millesi136 reported 64.5% “useful recovery” in complete lesions and 82.4% in incomplete lesions. Narakas134,135 noted 61% fair results achieved in cases of supraclavicular lesions. Chuang et al.201 presented a report of 15 patients with total root avulsion of the brachial plexus who were treated with cross-chest C7 nerve graft and then free muscle transplantation at a later stage in eight patients. After a long period of rehabilitation, useful function of the reconstructed limb was achieved, although no independent movement of the transferred muscles could be documented. Nagano202 recommended that nerve grafting should be performed if a ruptured root shows positive evoked spinal cord potentials or somatosensory evoked potentials in the trunk or in the cord. Exploration of the brachial plexus should be extended distally as far as possible to achieve good results after nerve grafting. When the exploration is thus extended, more than M3 power of the infraspinatus, deltoid, and biceps can be achieved in more than 70% of patients. Nerve Transfers Mackinnon and Colbert27 provided a contemporary update of indications and techniques in the rapidly expanding application of nerve transfers. The theoretical basis for nerve transfers is similar to that for tendon transfers: trivial or redundant motor units are surgically redistributed to reconstruct a critical missing function. Nerve transfers, however, have the capacity for reconstruction of sensation. Common clinical situations include high injuries to the radial, ulnar, and median nerves and to the brachial plexus. The technique of 31 SRPS • Volume 11 • Issue R7 • 2015 reconstruction using a distal motor nerve transfer is now considered superior to the use of long nerve grafts to the brachial plexus for brachial plexus and high-level nerve injuries.203 The management philosophy regarding nerve transfers is that of converting high-nerve injury to low-nerve injury. Nerve transfer also requires a distal organ with potential for functional recovery. Choosing nerve transfer for motor reconstruction is therefore time sensitive. Principles include the following: 1.Maintain anatomic and physiological integrity of musculotendinous units. 2.Minimize regeneration distance and time. 3.Target function of the transferred nerves. 4.Dissect in an unscarred field.203,204 Although nerve transfers have been described for proximal injuries, their successful use distal to the elbow opens a new spectrum of options for reconstruction. Table 9 provides a comprehensive list of commonly used nerve transfers.27 Proximal nerve transfers have involved donor nerves such as the thoracodorsal,205 medial pectoral,206 spinal accessory,207,208 phrenic,209,210 and intercostal nerves.211 The donor nerves can be divided into intraplexal (based on brachial plexus branches) or extraplexal (based on nerves outside the brachial plexus), with only the latter being available after total brachial plexus avulsion injuries. All these donor nerves must be noted as terminal nerve trunks, which are essentially sacrificed at the time of transfer with total loss of muscle function in their native territory. A paradigm shift occurred when Oberlin et al.212 began to use a single proximal fascicle of the intact ulnar nerve for reinnervation of the biceps. Doing so allowed the sacrifice of a highly selected fascicle of the ulnar nerve as a donor for transfer, with minimal to undetectable loss of function in the remaining ulnar nerve distribution because of highly redundant function among proximal fascicles. Similar strategies have since been used, with good success for distal transfers using an expendable fascicle from the median nerve as a donor for elbow flexion213 or radial nerve palsy (Fig. 20).214,215 The double fascicular transfer was described in 2005 as means of innervating both biceps and brachialis muscles to improve outcome in elbow flexion reconstruction.216 The contralateral C7 nerve root has been used 32 as a donor for multiple-root avulsion injuries of the brachial plexus. First described by Gu et al.217 in 1992, the contralateral C7 nerve root has been identified as expendable in the setting of a normal uninjured contralateral brachial plexus because of redundancy in function among the adjacent C6 and C8 nerve roots.218 Reports have indicated that the predominant morbidity after transfer is a transient sensory disturbance in the thumb, index, or long fingers, generally lasting 3 to 6 months.219 Either the entire C7 nerve root or a hemi-C7 donor can be selected, based on the needs at the recipient nerve. When selecting which portion of the C7 root to use in a hemi-C7 transfer, the anterior division generally is used for flexor functions and the posterior division for extensor functions. The posterior division of C7 contains a greater number of motor axons compared with the anterior division.201,220,221 A nerve graft usually is required because of the long distance between the donor and recipient nerves; pedicled or free vascularized ulnar nerve grafts are preferred, but sural or saphenous nerve grafts can also be used (Figs. 21-23).222 The contralateral C7 transfer has also been applied in children.223 Concerns remain regarding synkinesis of the contralateral donor arm to activate motion of the reconstructed recipient arm in a majority of patients, and attempts at elucidating the relevant neurophysiological adaptations are ongoing.224 Nagano202 recommended intercostal nerve transfers to restore elbow flexion in root avulsion type injury, and M3 or greater strength can be achieved in 70% of patients. Best results were obtained in patients younger than 30 years and in those who have been treated surgically within 6 months of injury. However, using the intercostal nerve as a donor dictates a longer regeneration distance to biceps in comparison with intraplexal distal donor nerves, which might explain poorer results after 6 months. Tendon Transfers The surgical practice of tendon transposition is based on the concept that the power of a functioning muscle can be used to activate a nonfunctioning nerve-muscletendon unit. Tendon transfers are justified for restoration of functional motion of the hand, not just motion. The surgical alternatives to tendon transfer include neurorrhaphy, tenorrhaphy, tenodesis, tendon graft, arthrodesis, and amputation. SRPS • Volume 11 • Issue R7 • 2015 Table 9 Common Nerve Transfers of the Upper Extremity27 Injured Nerve Missing Function Donor Nerve Recipient Nerve Motor Suprascapular Shoulder abduction, stabilization, external rotation Distal spinal accessory Suprascapular Axillary Shoulder abduction Triceps branch of radial nerve Axillary Musculocutaneous Elbow flexion Ulnar nerve fascicle to FCU, median nerve fascicle to FCR Brachialis branch, biceps branch Spinal accessory Shoulder elevation, abduction Medial pectoral Spinal accessory Spinal accessory Shoulder elevation, abduction C7 redundant fascicle Spinal accessory Ulnar Intrinsic hand Terminal AIN (branch to pronator quadratus) Ulnar nerve fascicles to deep motor branch Median and ulnar Intrinsic hand Radial nerve branches to ECU, EDQ Ulnar nerve deep motor branch Median Thumb opposition Terminal AIN (branch to pronator quadratus) Median (recurrent) motor Median Finger flexion Ulnar nerve fascicle to FCU AIN Median Pronation Ulnar nerve fascicle to FCU, intact branches to PL or FCR, radial nerve branch to ECRB Median nerve branch to pronator teres Radial Wrist extension, finger extension Median nerve branches to FCR, FDS, PL Radial nerve branch to ECRB, PIN Median sensory Thumb-index finger key pinch area sensation Median common sensory branch to third web space in upper trunk (C5, C6) injury Median common sensory branch to first web space Median sensory Thumb-index finger key pinch area sensation Ulnar common sensory branch to fourth web space Median common sensory branch to first web space Median sensory Thumb-index finger key pinch area sensation Dorsal sensory branch of ulnar nerve Median common sensory branch to first web space Median sensory Thumb-index finger key pinch area sensation Sensory branch of radial nerve Median common sensory branch to first web space Median sensory Noncritical median distribution sensation Ulnar common sensory branch to fourth web space (side to end) Median common sensory branch to second and/or third web space Ulnar sensory Ring and small finger sensation Median common sensory branch to third web space Ulnar common sensory branch to fourth web space, ulnar digital nerve to small finger Ulnar sensory Ulnar border of hand sensation Median sensory at distal forearm (side to end) Ulnar sensory nerve at distal forearm Ulnar sensory Ulnar border of hand sensation Lateral antebrachial cutaneous Dorsal sensory branch of ulnar nerve Median sensory at distal forearm (side to end) Dorsal sensory branch of ulnar nerve Sensory Ulnar sensory FCU, flexor carpi ulnaris; FCR, flexor carpi radialis; AIN, anterior interosseous nerve; ECU, extensor carpi ulnaris; EDQ, extensor digitorum quinti; PL, palmaris longus; ECRB, extensor carpi radialis brevis; FDS, flexor digitorum superficialis; PIN, posterior interosseous nerve. 33 SRPS • Volume 11 • Issue R7 • 2015 Injury Open Surgical Reconstruction Explore No Improvement/Nerve Conduction Studies No Injury Observe (3 months) Improvement Done No Improvement/Nerve Conduction Studies Closed Surgical Reconstruction Observe (3 months) Improvement Done Figure 20. Radial nerve palsy algorithm. (Reprinted with permission from Lowe et al.209) When injury to a nerve renders a muscle-tendon unit useless, nerve repair often is indicated. The prognosis for function depends on the type of injury, time since injury, distance from lesion to muscle, patient age, type of nerve, etc. (see above). de Medinaceli et al.225 reviewed the results of primary nerve repair in 2181 fresh nerve injuries of the upper extremity at a reconstructive surgery center in France. Digital injuries accounted for 68% of all cases. Forty-one percent of patients had injury of only one nerve, whereas the other 59% had injury of two or more nerves. In 18% of cases, the injured limb was lost or beyond repair. Of the remaining 1800 or so injuries, end-to-end suture was successful 87% of the time and not possible in 11%, even with nerve graft. The authors concluded that primary neurorrhaphy was possible in a very large majority of peripheral nerve injuries of the upper limb and consequently that true loss of substance was rare in civilian practice. Direct suture of an injured tendon is not applicable in every case but should not be ignored in the decision. When the prognosis for function after tendon transfer is poor, tenodesis is a definite consideration. The mere provision of a fixed point of origin for a tendon will 34 sometimes position a joint so that it moves acceptably. The best approach to an irreparable tendon injury often lies between a tendon graft and a tendon transfer. Tendon grafts use muscles that already have the correct power, amplitude, and direction, require no relearning of task, and involve no loss of other function. On the minus side, tendon grafts require two junctures rather than one, have no blood supply, and are more prone to adhesions. Fusing a joint occasionally provides sufficient stabilization to simulate the desired muscle action (e.g., the interphalangeal [IP] joint of the thumb after loss of the FPL). Amputation of a ray or digit might be indicated in extreme cases to shorten the rehabilitation time and restore useful function. Tendon transfer might be indicated for functional reconstruction: •• After nerve injury •• After loss of muscle-tendon unit (acute or chronic) •• As a rebalancing procedure SRPS • Volume 11 • Issue R7 • 2015 Procedure Selection—Several factors need to be assessed when considering undertaking tendon transfers: 1.General factors 2.Patient factors 3.Recipient factors 4.Donor factors Figure 21. Custom-made metal passers for cross-chest tunneling of nerve grafts. Upper, passer for ulnar nerve; lower, passer for saphenous nerve. (Reprinted with permission from Terzis and Kokkalis.217) 5.Surgical factors and technique General Factors Task Analysis The patient’s need or desire to perform a certain task is of paramount importance in planning a transfer procedure. To accomplish that task (e.g., open a door), the fingers must be positioned in the proper way to engage the doorknob and must have the power to grasp and twist it. A transfer designed to restore intrinsic function to the fingers for grasp is of little use unless it also carries enough power for closing around the knob and turning it. Transfers, therefore, should be designed to accomplish tasks rather than motions. Figure 22. Selected bundles of anterior and posterior divisions of C7 have been neurectomized and coapted in end-to-end manner to two cross-chest saphenous nerve grafts. (Reprinted with permission from Terzis and Kokkalis.217) Nature of the Disease If the functional loss is caused by systemic or local disease, it should be controlled before restoration is attempted. If the disability is secondary to trauma, tendon transfer after neurorrhaphy can maintain muscle function until nerve recovery is complete. Patient Factors Motivation Figure 23. “Banked” cross-chest saphenous nerve grafts are depicted. Orange, upper graft connected to selected bundles of posterior division will be used for neurotization of free-muscle graft for finger extension restoration; green, lower graft is connected to selected bundles of anterior division and is destined for neurotization of free muscle for finger flexion. (Reprinted with permission from Terzis and Kokkalis.217) For any method of functional restoration to succeed, the patient must first be aware of the loss and be willing to do something about it. Patients who are unconcerned about the disability are unlikely to retrain the muscle that is newly empowered by a transferred tendon. Age Tendon transfers are relatively contraindicated in the elderly because joint stiffness during splinting increases with age. The process of relearning or teaching a muscle 35 SRPS • Volume 11 • Issue R7 • 2015 to perform a new task is not only lengthier but also more difficult at an advanced age, and the corresponding need for power movements also decreases. Cognition The patient must be capable of understanding both the nature of the surgery and its limitations. Additionally he or she must understand the motor reeducation program necessary for movement of the recipient tendon by activation of the transferred tendon. by approximately one motor grade when the muscle is detached from its normal insertion and is placed opposite a new antagonist. Reinnervated muscles are not ideal and should have a Medical Research Council grade of 4 or greater and be used only in exceptional circumstances. In general, muscle power corresponds to muscle cross-section, but by placing the tendon insertion farther from the joint axis and as close to 90 degrees as possible, the effective power of the muscle can be increased during transfer. Excursion Compliance The patient’s adherence to the required immobilization and activity limitation during the period of healing and to the therapy regimen necessary for retraining of the transferred tendon and mobilization of stiffened joints must be reasonably certain. Assessing patient compliance to preoperative physiotherapy can provide a useful predictor of compliance postoperatively. Recipient Site Factors •• Soft tissue: The soft tissue should be pliable and free from scar tissue and overlying skin graft. •• Skeletal Stability: Joints should be stabile to maximize the efficiency of the transfer. •• Sensation: Normal neurovascular status is required to provide useful function. •• Supple joints: Joints should be supple with a full range of motion. A tendon transfer will not rehabilitate a stiff joint. Ideally, the donor muscle should have the same amplitude or excursion as the muscle it replaces. Stretching the donor tendon over a number of joints improves total excursion. Wrist flexors and extensors have excursions of approximately 3 cm, finger extensors 5 cm, and finger flexors 7 cm. Effective amplitude can be increased by the effect of tenodesis or by freeing of fascial attachments to the muscle-tendon unit. A muscle of too much power and too little excursion can be inserted closer to the joint axis to minimize the distance it must contract to produce the desired motion. One Tendon, One Function Ideally, each tendon transfer should be designed to perform a single action. Effectiveness of a tendon transfer is reduced when designed to produce two different functions. Additionally, the donor muscle should be independently innervated (e.g., the sublimis) and not act in concert with other motors (e.g., the lumbrical). Performance Donor Muscle Factors A useful mnemonic with which to remember the donor muscle factors is PEOPLES: Power and control; Excursion; One tendon, one function; Performance; Line of pull; Expendability; Synergism. Power and Control The donor muscle power should be similar to that which it replaces and must be adequate for the function it will assume after transfer, remembering that it will diminish 36 A donor muscle must be able to perform under voluntary control. Muscles prone to spasticity or involuntary contraction will function poorly. Line of Pull Tendon transfers work more efficiently when traveling in a direct line of pull from its origin to new insertion. Where this is not possible, the tendon should pass through no more than one Pulley and the design should avoid acute angulation. SRPS • Volume 11 • Issue R7 • 2015 Expendability The replacement tendon must be carefully selected to avoid substantial functional deficit as a consequence of transfer.226-239 One must be confident that transfer will not result in loss of a critical hand movement from lack of the donor function. For example, in cases of radial nerve palsy, transfers for wrist and finger flexion should ensure that at least one functioning wrist flexor remains intact. Smith and Hastings239 offered an algorithm for selecting appropriate donor muscles to restore function in the hand. Synergism Muscles transferred within synergistic groups that produce composite movement are generally easier to retrain. Synergistic muscles include the fist group (wrist extensors, finger flexors, digital adductors, thumb flexors, and forearm pronators) and the open hand group (wrist flexors, finger extensors, digital abductors, and forearm supinators). The operative plan must also be designed to avoid hand imbalance at any of the affected joints. A tendon transfer can benefit a patient who has lost a certain movement. The substitute tendon is chosen, aligned, and inserted according to the patient’s need for a specific motion rather than based on a standard prescribed formula. For complex functional problems or multistage reconstructions, the surgeon should list the potential donor muscles for each desired function to maximize the benefit of the available donors and prevent using a muscle to power one action only to find out later that it could have been better used in another capacity. Consideration must be given to the balance of the transfer. An overly powerful muscle will overcompensate and, in time, deform the joint. Timing of tendon transfers depends on the cause and prognosis of the motor imbalance, the neurophysiological problems for the patient, and the condition of the involved extremity.240 In cases in which stiffness and deformity can be expected to increase with time, however, the transfer should be performed early, such as after median nerve injury with a poor chance of recovery, in which case a tendon transfer to secure thumb opposition will prevent the supination and/or adduction deformity that is likely to develop as the patient substitutes new motions in an attempt to preserve function. Surgical Factors and Techniques •• Incisions: When creating incisions, avoid crossing the path of the transferred tendon. The tendon should course through subcutaneous tissue free of scar to avoid adhesions that might tether it and limit its function. •• Operate in reverse order: Have the recipient site and tunnel ready before raising the muscle. •• Avoid interference with critical structures: Ensure that the transferred tendon does not compress nerves. •• Apply correct tension: The tension at the insertion should be set to produce the necessary movement of the joint with maximal muscle contraction. Because it tends to “stretch” slightly, some initial overcorrection is indicated by many surgeons for certain transfers (extensor indicus-extensor pollicis longus [EPL]). However, it has been shown that overstretching the muscle into the passive tension portion of the Blix curve decreases the potential contractile force to 28% of its maximum force.241 Therefore, setting the donor muscle near its normal length might be optimal for force generation.241 •• Tendon insertion: Tendon insertion should be at an angle of 90 degrees to the joint of motion to maximize power and excursion. The insertion can be moved away from the axis of the joint to improve power but at the cost of diminished excursion. A single insertion is best; when dual insertions are contemplated, motion will be effected at the tighter one (can occasionally be used to advantage in complex movements, with which one insertion is tighter during one phase of motion and the other takes over during another phase). •• Tendon coaptation: Methods include Pulvertaft weave, side-to-side, and bone fixation. Whichever method is used, it 37 SRPS • Volume 11 • Issue R7 • 2015 should be robust enough to allow early active mobilization and to minimize adhesions. A recent in vitro study using human cadaver tendons comparing the Pulvertaft weave and side-to-side methods showed average detected failure loads of 182 N and 96 N, respectively.242 •• Joint immobilization: The affected joint should be immobilized for an appropriate length of time in a position that relieves tension at the insertion. Specific Deficits Low Median Nerve Palsy Palsy of the median nerve of the hand often occurs after a forearm injury that typically includes disruption of the tendons, blood vessels, and ulnar nerve. When isolated, the median nerve injury produces sensory loss in the palm, sensory loss in the radial side of the hand and fingers, and loss of motor innervation to much of the thenar musculature. Loss of sensation to the pulp of the thumb, index finger, or long finger severely inhibits pulp-to-pulp pinch, a delicate motion which, unlike gross pinch, requires constant receptor input. Under optimal conditions, only 25% to 30% of patients achieve satisfactory thumb position (M3) and TPD of <15 mm (S3) with direct nerve repair,243 dictating the need for tendon transfer to improve hand function. Sensory restoration to the contact surfaces of the digits using neurovascular island flaps244 or digital nerve transfers245 might be necessary to complete the feedback loop for successful control of pinch. The principal motor deficit associated with a low median nerve injury is loss of thumb opposition. Thumb opposition is a composite of two movements: one rotates the pulp of the thumb to face the pulp of the index ray, and the other abducts the thumb metacarpal ray from the palmar plane.246 The combined action normally is brought about by the opponens pollicis (rotation and/or pronation of the thumb ray) and the abductor pollicis brevis, assisted by the superficial head of the FPB (some pronation, palmar abduction, and metacarpophalangeal [MP] flexion).247 Although placing the thumb in opposition 38 prepares the thumb for pinch, pinch is not a power move that requires a large and powerful muscle. Synergistic muscles include the finger flexors, wrist extensors, PL, and hypothenar muscles. The standard transfer places the FDS of the ring finger248 through a pulley249 in the region of the pisiform246 to its insertion. Other possible transfers for thumb opposition in cases of low median nerve palsy were summarized by Smith and Hastings (Table 10).239 Bunnell246 suggested pulling the thumb metacarpal toward the carpal pisiform by a tendon coming from that direction to create the proper vector of motion. Littler250 reported preference for a pull in the direction of the adductor pollicis brevis (APB), and Thompson249 chose a more transverse route across the palm. The transferred tendon must insert on the radial side, either at the APB250 or halfway into the APB and halfway into the EPL, in a dual insertion,251 to increase the extension of the IP joint and help balance the flexor force of an intact FPL at that joint. The dual insertion transfers some of the force of the FPL to the more proximal joints and helps stabilize the MP joint. A modification splits the insertion between the EPL and the adductor pollicis longus (APL) to try to neutralize the adductor and/or supinator forces.252 Tension at the insertion is adjusted to move the thumb into full opposition with the wrist in neutral. With the wrist in neutral position, a minimum of 25 to 30 mm of tendon excursion is needed for adequate thumb ray function, even more if the transfer is to function in all wrist positions. In 1929, Camitz253 described the PL abductorplasty for severe thenar atrophy secondary to carpal tunnel syndrome (CTS). The concept was later revisited by Littler and Li254 and by Braun.255 More recently, Terrono et al.256 reviewed the outcomes of the PL transfer described by Camitz in 33 hands (29 patients). Surgery was secondary in three patients and primary (at the time of initial release of the carpal tunnel) in 26. At a mean follow-up duration of 17 months, 94% of the patients were satisfied with the results of surgery because their thumb function had improved. SRPS • Volume 11 • Issue R7 • 2015 Table 10 Low Median Nerve Palsy: Transfers for Opposition239 Donor Pulley Graft Insertion on Thumb FDS (ring) Distal FCU None Dorsal-ulnar base PP FDS (ring) FCU None APB FDS (ring) FCU None APB and EPL at PP FDS (ring) Guyon canal None APB and EPL EIP Ulnar side of wrist None PP ADQ None None APB PL Carpal tunnel Rerouted EPB EPB PL None Palmar fascia Radial side MP joint Half FPL Around radial side of thumb None Dorsal PP FPL Translocated dorsally None PP FPL Transverse carpal ligament None PP and FDP APL PL None Base metacarpal ECU Ulnar side of wrist Rerouted EPB EPB ECRL or ECRB Ulnar side of wrist Free graft EPL FDS, flexor digitorum superficialis; FCU, flexor carpi ulnaris; PP, pronator profundus; APB, adductor pollicis brevis; EPL, extensor pollicis longus; EIP, extensor indicis proprius; ADQ, abductor digiti quinti; PL, palmaris longus; EPB, extensor pollicis brevis; MP, metacarpophalangeal; FPL, flexor pollicis longus; FDP, flexor digitorum profundus; APL, adductor pollicis longus; ECU, extensor carpi ulnaris; ECRL, extensor carpi radialis longus; ECRB, extensor carpi radialis brevis. A tendon transfer designed to restore thumb ray opposition must include release of any long-standing adduction contracture and lengthening of the web fascia. The EPL and APL act to supinate the thumb in an adducted position, so much so that a substitute for pinch in the absence of median nerve abduction involves rotating of the thumb into this position, which is relatively stable, and bringing the index pulp to meet it. Effective powerful pinch requires a stable MP joint to avoid collapse, which in turn demands careful balance of extensor and flexor forces. The FPB prevents extension of the joint as the thumb pulp contacts the index pulp. In isolated low median nerve injuries, the tendon retains marked ulnar innervation in approximately half the cases257 so that MP joint stabilization is not needed; when complicated by low ulnar palsy, however, any effective transfer for power pinch must include MP flexion and/or stabilization.258,259 An alternative method for restoring opposition after median nerve injury was suggested by Ustün et al.260 Based on a cadaver study, the authors recommended transfer of the motor branch to the pronator quadratus muscle from the anterior interosseous nerve to the thenar motor branch. They noted that this would allow for reestablishment of some motor function with a minor loss of function after denervation of the pronator quadratus muscle. This technique has been brought to clinical fruition by Vernadakis et al.261 who used a nerve graft from the distal anterior interosseous nerve (pronator quadratus branches) to the thenar motor branch. 39 SRPS • Volume 11 • Issue R7 • 2015 High Median Nerve Palsy Injuries to the median nerve above the level of the forearm increase the likelihood of ulnar nerve damage, limiting the transfer alternatives to radially innervated muscles. In addition to severe motor and sensory deficit of the index finger and thumb, loss of forearm pronation and loss of flexion of the index and long fingers and the FPL of the thumb occur. The goal for the reconstruction is gross key pinch rather than fine discriminatory tasks. Loss of forearm pronation creates a marked disability. A hand that is allowed to supinate for a long time after nerve injury will develop a contracture that is difficult to reverse. It is therefore important to maintain range of motion of the forearm. If reinnervation of the pronators is unlikely or delayed, one should consider rerouting the biceps. Smith and Hastings239 listed various options for tendon transfer in high median nerve palsy.255,262-268 Because the powerful finger flexors that contribute to wrist flexion have been lost, two wrist flexors can be transferred to the digits without compromising wrist balance. When the two deep flexors on the ulnar side of the hand still have power, all the profundus tendons can be sutured together to achieve mass finger flexion. If strong radial hand motion is required, the extensor carpi radialis longus (ECRL) can be transferred to the profundi of the index and long fingers. Disparate excursions of the ECRL and profundus tendons demand fine adjustment of tension at the insertion to enable finger flexion without contracture from this dynamic tenodesis. The brachioradialis can be used to duplicate the action of the FPL. Once again, the tension of the transferred tendon can be set loose enough to prevent flexion contracture while the wrist tenodesis serves to flex the thumb. Low Ulnar Nerve Palsy In the absence of normal ulnar innervation, hand function is severely limited. Most fine hand movements depend on muscles innervated by the ulnar nerve. Sensory loss along the ulnar border of the hand constitutes a substantial disability, even though it is less of a problem than the 40 motor deficit. The affected muscles are the four dorsal and three volar interossei, the ring and small finger lumbricales, the APL and deep head of the FPB, and the hypothenar mass. In cases of low ulnar nerve palsy, the deficit in the moment potential of MP joint flexion decreases by varying amounts, depending on the digit.269 The middle, ring, and small fingers lose moment potential by 7%, 13%, and 28%, respectively. Each muscle should be specifically tested before embarking on surgical reconstruction, however, because of frequent anomalous communications between the median and ulnar nerves. These crossconnections might alter the severity of the deficit and negate the standard patterns of ulnar = ring and small fingers and median = index and long fingers. Functionally, the deficit manifests as follows: •• Coordinated finger flexion and IP joint extension are lacking because of loss of the lumbricals and interossei, causing the fingers to roll into the palm in an ineffective grasp rather than encircle an object. The loss of active digital abduction and/or adduction produces the typical “claw” deformity as the extrinsics hyperextend the MP joints while trying to extend the IP joints. This is sometimes accompanied by an abducted little finger (Wartenberg sign) resulting from unopposed abduction by the extensor digiti minimi.270 •• Thumb-index finger pinch of all kinds are diminished from the combined loss of effective support to the thumb MP joint and adductor pollicis and loss of the first dorsal interosseous, which severely limits abduction strength in the index finger. Sideto-side movement of the fingers, necessary for fine manipulation of objects within the hand, is prevented by loss of the interossei. •• Power grip is absent because of loss of the intrinsics for grasping and the hypothenar mass for cupping an object. •• Sensation in the palm along the ulnar border is lost, and the loss might extend to the dorsal surface in cases of lesions proximal to the origin of the dorsal sensory SRPS • Volume 11 • Issue R7 • 2015 branch of the nerve (approximately 6–8 cm above the wrist). Clawhand is an indication for intrinsic muscle transfer in all four fingers. Various tendon transfers, with and without tendon grafts, have been suggested for the correction of clawhand.239 Common sites of insertion are the lateral bands, proximal phalanx, and A2 pulley. Hastings and Davidson270 analyzed the results achieved in 34 patients with ulnar nerve palsy and clawing who underwent reconstruction by various techniques of tendon transfer. Of the 14 digits treated for low ulnar deficit, correction of clawing was achieved in all instances with Brand and Riordan transfers and in two-thirds to three-fourths of those treated by Stiles-Bunnell transfer and Zancolli lasso transfer. Grasp strength was not substantially improved by any of the procedures, however, leading the authors to conclude that only the addition of a tendon motor from the wrist will add power. Limiting full finger extension at the MP joint theoretically allows the extrinsic extensors to exert force for active IP extension and prevents recurrence of clawing. In a strong hand, the extensor muscles tend to stretch with time and eventually become ineffective. Mikhail271 suggested placing a bone slab on the dorsal side of the MP joint to limit extension. Bunnell272 reported that MP arthrodesis achieves a similar effect, with obvious restriction of full finger flexion into the palm. These techniques achieve neither coordinated finger flexion to grasp a large object nor improved grip strength and should be strictly reserved for patients with limited motor resources from multiple paralyses. Zancolli273 originally advanced the volar plate of the MP joint proximally while excising volar palmar skin to passively restrain the MP joint. Modifications of the Zancolli procedure have since been described.274,275 Bunnell276 incised the A1 and part of the A2 pulleys on their volar aspect to bowstring the flexor tendons and increase their power at the MP joint. The basic technique of tenodesis routes a tendon (ECRL or extensor carpi ulnaris) attached to the proximal phalanx or dorsal hood through the intermetacarpal spaces to end beneath the deep transverse metacarpal ligament.251,277-280 Wrist flexion is made possible by attaching the proximal juncture to the wrist dorsum.280 An unstable thumb MP joint, loss of thumb adduction, and insufficient index finger abduction all contribute to inadequate pinch in ulnar palsy. Tendon transfers succeed in increasing available power to only 30% to 50% of normal strength.281 Some of the more common transfers designed to improve pinch include the tendon loop presented by Bunnell276 (extensor digitorum communis [EDC] to index finger with a tendon graft around the ulnar border of the hand to the adductor tubercle of the thumb), the extensor indicis proprius (EIP) transfer presented by Brown281 (passed between the metacarpals to insert on the adductor tubercle), and the brachioradialis or ECRL presented by Boyes282 (to insert on the adductor pollicis). Another pinch-improving strategy aims at increasing the abduction power of the index for use against the thumb. Brand283 reminded us that most people can support a key-pinch simply by flexing all the adducted fingers together, so that the thumb presses against a fistful of fingers. High Ulnar Nerve Palsy Injuries to the ulnar nerve above the elbow affect the ring and small finger FDP and the FCU in addition to producing the disabilities described under low ulnar nerve palsies. The major functional problem resulting from the injuries is further weakening of power grip. Despite high ulnar injury, finger flexion on the ulnar side might still be possible because of variable innervation of the FDP and intramuscular fascial connections. Patients with high ulnar injury can be treated with suturing of the FDP of all fingers in the forearm to distribute the available power more evenly across the full width of the hand. FCU loss can be similarly ignored in most cases, although the tendon plays an important role in certain motions involving wrist flexion and ulnar deviation (e.g., swinging a hammer).283 A forearm muscle being used to motor the proximal phalanx must cross the wrist, affecting it in some way. In cases of high ulnar palsy, the wrist is already overpowered on the radius/extensor side; it therefore makes sense to use a radial wrist extensor as the donor tendon to counteract that force. When the transfer approaches the insertion from the volar wrist, the moment arm is increased by 41 SRPS • Volume 11 • Issue R7 • 2015 flexion, raising the power of the motion correspondingly. Various authors250,274-285 have suggested transferring the ECRL through the interosseous membrane across the volar aspect of the wrist to insert proximally. Alternatively, the extensor carpi radialis brevis (ECRB) elongated by a graft can be passed across the dorsal wrist and through the intermetacarpal spaces.286 Fowler280 and Riordan278 reported a preference for the EIP, and Fritschi287 recommended the PL. Brand283 advocated transferring the brachioradialis or FCR tendon to supplement the function of the FCU in selected cases. Combined Median and Ulnar Nerve Palsy Tendon transfers in cases of combined nerve palsies are more complicated than those in cases of isolated nerve injuries because of the usually complex upper extremity trauma, poor proprioception, weakness of muscles available for transfer, and need for multistage procedures. finger extension is best motored by the synergistic FCR; the EPL remains motored by the PL. Green294 presented details and illustrations of the various transfer techniques that can be used in radial nerve palsy, and Smith and Hastings239 listed the options. Beasley295 reported a preference to adjust the tension in the fingers and thumb first. With the wrist in full extension, the fingertips should reach the palm on passive flexion; with the wrist in full flexion, complete MP extension should be possible. The tension on the wrist extension transfer is set last and should be as tight as possible. Lowe et al.296 described the use of nerve transfers to the radial nerve as an alternative to tendon transfers in radial nerve injuries. The authors presented the cases of two patients who underwent transfers of motor branches to the PL and FDS off the median nerve to branches of the posterior interosseous nerve and motor branch to the ECRB muscle. At 13 to 14 months postoperatively, both patients had a motor strength score of 4 of 5 at the wrist and fingers. Radial Nerve Palsy Injuries to the radial nerve impair the hand tremendously by voiding extension power to the wrist, fingers, and thumb and preventing radial abduction of the thumb. Wrist extension is critical for stability, which in turn is essential for grip and for function of the many tendons passing over it to mobilize distant joints. Tendon transfers for radial nerve palsy are well defined and highly effective. The “standard” transfers include PT to ECRB for wrist dorsiflexion;288 FCU to EDC to fingers II through V; and PL to EPL for thumb extension.289,290 Boyes and colleagues291,292 suggested an alternative superficialis transfer (PT to ECRB and ECRL for wrist extension, long finger FDS to EDC for finger extension, ring finger FDS to EIP and EPL for thumb and index extension, and FCR to APL and extensor pollicis brevis for thumb radial abduction). The author reasoned that the FCU should be preserved because the normal direction of wrist flexion is from dorsoradial to volar ulnar, a motion to which the FCU substantially contributes. The transfer preferred by Brand293 for cases of radial nerve palsy consists of PT to ECRB for wrist extension, FCR to EDC for finger extension, and PL to EPL for thumb extension. The logic behind this protocol is that 42 Other Transfers Peripheral nerve injuries in the upper extremity are the primary reason why tendon transfers are undertaken. In addition, loss of muscle power in the hand occurs from many other causes, and restoration of some degree of function can be attempted by performing transfers. Omer240 noted that in poliomyelitis, 50% of the residual strength of a muscle is recovered within 3 months of onset and 75% within 6 months. If voluntary muscle test at 3 months shows only a trace of activity in a pertinent muscle-tendon unit, tendon transfer is indicated. High Cervical Quadriplegia and/or Tetraplegia Freehafer297,298 described his experience with tendon transfers in tetraplegic patients. During a 30-year period, 272 of 285 patients experienced improvement in upper extremity function after tendon transfers after high-level spinal cord injuries. Recommended transfers to achieve independence in those patients included wrist extension procedures, posterior deltoid transfers, opponens transfers, and transfers for finger flexion. The author reported that patients with C6, C7, and C8 complete neurological SRPS • Volume 11 • Issue R7 • 2015 injuries were almost as independent after the tendon transfers as paraplegics. They could transfer from a chair, insert a catheter, write, type, hold a book, take care of their toilet needs, bathe themselves, eat, drink, dress themselves, and perform other activities of daily living. A few patients with C5 neurological level required automatic grasp or used a wrist-driven splint. After the procedures, the patients achieved active wrist extension, elbow extension, thumb pinch, and finger grasp. A key concept in hand reconstruction for tetraplegia is the use and surgical augmentation of tenodesis motion, whereby active extension and passive flexion with gravity at the wrist are parlayed into digital motion by rebalancing the digital flexor and extensor tendons through shortening and/or bone attachment. This is the principle behind the Moberg key pinch and House series grasp reconstructions.299 Other authors have reported their experiences and made recommendations for patients with spinal cord injury.300-312 Rehabilitation Physical therapy is essential to regain joint mobility lost during splinting and to train the tendon to glide in its new course. Even more important is teaching patients to activate the replacement muscle when they want a certain response, a process that must be repeated many times to establish the new neural pathways before it becomes automatic. The more the patient feels the disability, the greater the motivation and the easier the retraining. Despite a long history of tendon transfer surgery and the multiple surgical procedures described, there is a paucity of literature regarding management of the hand after tendon transfer. Toth313 outlined the basic principles of rehabilitation as follows: Preoperatively, it is imperative to obtain maximum passive range of motion, quality of soft tissues, and strength of donor muscles. Postoperative care is administered in four stages: 1.The protective phase begins at surgery and lasts 3 to 5 weeks. During that time, therapeutic objectives are to protect the transfer by immobilization (protective splinting), control edema, and mobilize the uninvolved joints. 2.During the mobilization phase, which begins when tendon healing is adequate to begin activation of the transfer (usually 3 to 5 weeks postoperatively), one should strive to mobilize the tendon transfer, immobilize the soft tissue, continue mobilization of uninvolved joints to prevent joint stiffness from disuse, reinforce preoperative teaching and patient education, continue edema control, continue protective splinting, and begin a home hand rehabilitation program. 3.The intermediate phase begins 5 to 8 weeks after transfer. At that time, the patient gradually increases hand activity (active and passive range-of-motion exercises), and limited functional movements are permitted. 4.During the final resistive phase, beginning at 8 to 12 weeks when tendon junctions are strong enough to withstand increasing resistance, the therapeutic objective is to increase endurance and strength of transferred muscles. Work-related simulated tasks are begun to patient tolerance. Toth313 further delineated the management of specific tendon transfers in his excellent article, which is highly recommended to all hand surgeons. Surgical execution is only part of the key to the success of a tendon transfer; postoperative rehabilitation is in large measure responsible for the eventual functional outcome. The volume of evidence for rehabilitation after tendon transfer compared with tendon repairs is relatively small. A recent systematic review of rehabilitation regimens administered after tendon transfers found that although early mobilization offers some short-term benefits (superior range of motion, grip strength, pinch strength, and tendon transfer integration), the benefit for long-term outcome is not conclusive.314 Free Functioning Muscle Transfer Local muscle transfers used for functional reconstruction in the upper extremity include a Steindler flexorplasty, a pectoralis major transfer, a latissimus dorsi transfer, and a triceps transfer. However, a muscle loses a grade of 43 SRPS • Volume 11 • Issue R7 • 2015 power on transfer and a lack of choice of donor muscle often occurs because of neurological injury concomitant to the brachial plexus injury. Free functioning muscle transfer (FFMT) is becoming increasingly popular in upper extremity reconstruction, and results from these procedures are superior to those achieved with local muscle transfers. FFMT are particularly useful in cases of global plexopathy.315,316 Indications for FFMT include acute or chronic root avulsion, rupture with failed nerve transfer, muscle loss caused by trauma, electrical burns, and tumor resection. If time from nerve injury to intervention is approaching a year, FFMT should be considered.139 Elbow flexion is a critical function in the upper limb and the most common indication for FFMT. Other key functions that should be considered include shoulder abduction, elbow extension, and finger flexion and extension. Donor muscles include gracilis, latissimus dorsi, and rectus abdominis muscles. Chuang316 undertook 647 FFMT between 1987 and 2003 and provided insight into the use of FFTM in brachial plexus avulsion injuries. Doi et al.258 from Japan presented the results of reinnervated free muscle transplantation in 24 patients with brachial plexus injuries. Time from injury was longer than 1 year in 21 patients. After conventional diagnostic maneuvers, including physical examination, myelography, and electromyography, all patients underwent surgical exploration of the brachial plexus and intraoperative electrodiagnosis. Twenty-one of 23 traumatic injuries showed complete avulsion of the C5 to T1 nerve roots, two had avulsion of the C5 to C7 nerve roots, and one had avulsion of the C5 and C6 nerve roots. The donor muscles (gracilis, latissimus dorsi, and rectus femoris muscles) were selected according to their length (i.e., the transplanted muscle had to reach from clavicle to forearm). Both the accessory nerve and the intercostal nerve were used to motor the transplanted muscles. Restoration of functions, such as elbow and finger flexion, required up to 20 muscle transplants in combination with multiple nerve transfers. Venous thrombosis with subsequent partial or total muscle loss developed in four cases and delayed reinnervation considerably. Analysis of results showed no significant difference in time to reinnervation among the three donor muscles (P > 0.002), but muscles reinnervated by the intercostal nerves took approximately 2 months longer to show EMG signs of reinnervation (average, 5.25 months) than those reinnervated by the spinal accessory 44 nerve (average, 3.3 months). The authors reported that the combination of the latissimus dorsi muscle reinnervated by the spinal accessory nerve led to the most powerful contraction and greatest range of joint motion. Another report by Doi et al.317 described the use of double free muscle transfer in a large series of patients with complete avulsion of the brachial plexus. Twenty-six of 32 patients were followed for at least 24 months after the second free muscle transfer. In most cases, the gracilis muscles were used for both the first and second muscle transfer; however, in some cases, latissimus dorsi or rectus femoris muscle was used. After initial exploration of the brachial plexus, the first free muscle transfer was used to restore elbow flexion and finger extension and was reinnervated by the spinal accessory nerve. The second muscle transfer, to restore finger flexion, was reinnervated by the 5th and 6th intercostal nerves. The motor branch to the triceps brachii was reinnervated by the 3rd and 4th intercostal nerves to restore elbow flexion. Hand sensibility was restored by suturing the sensory rami of the intercostal nerves to the median nerve or the ulnar nerve component of the medial cord. Secondary reconstructive procedures, such as arthrodesis, were also used.317 In the series presented by Doi et al.,317 96% of patients had satisfactory elbow flexion and 65% had satisfactory prehension (more than 30 degrees of total active motion of the digit). In addition, 54% of patients could position the hand in space, negating simultaneous flexion of the elbow, while moving fingers at least 30 degrees. They could also use the reconstructed hand for activities requiring the use of two hands, such as holding a bottle while opening the cap and lifting a heavy object. The authors concluded that the double free muscle procedure can provide reliable and useful prehensile function for patients with complete avulsion of the brachial plexus. In an earlier study, Doi et al.318 used a free gracilis muscle transfer to restore hand function after injuries to the lower brachial plexus. The authors presented the results achieved in three patients who had good outcomes after intercostal nerve transfers to the transferred gracilis muscle at long-term follow-up. SRPS • Volume 11 • Issue R7 • 2015 •• When the history and physical examination suggest that more than one nerve might be compressed in a single extremity and no history of trauma is reported, a more proximal source of compression should be considered. Compression Neuropathies Diagnosis Nerve compression is a common cause of peripheral neuropathy in the upper extremity. In a comprehensive, thoughtful treatise, Dellon319 discussed patient evaluation and treatment considerations for patients with compression neuropathies. Among the highlights of his review are the following points: •• When obtaining the history of the problem, begin with questions related to which areas are numb, tingling, or a source of pain, because they might relate to the innervation pattern of a specific peripheral nerve. In particular, ask questions regarding the small finger, because it is the only digit without any median nerve innervation. •• The most common misconception is to assume that “weakness” or “dropping things” is causally related to CTS. Symptoms related to weakness or loss of coordination are causally related to ulnar nerve dysfunction, most often to entrapment of the ulnar nerve at the elbow. •• Nocturnal disturbance is a hallmark of CTS. Neck pain should not be present. •• Neck pain deserves extensive evaluation, especially when concomitant with bilateral upper extremity symptoms. •• Chronic compression of a peripheral nerve results in a predictable sequence of events. The key to understanding the physical examination and to successfully diagnosing and managing the problem is to translate the physiological events into clinical manifestations. •• Testing with the tuning fork is the simplest and most efficient screening test for the hand surgeon to use in the office. •• Physical examination of a patient who has isolated peripheral nerve compression should reveal localized signs of entrapment of that nerve at the known area of anatomic narrowing (e.g., Phalen sign, Tinel sign). •• The diagnosis of chronic peripheral nerve compression in a single extremity should not represent a challenge in most cases. Bianchi et al.320 described the use of a highfrequency ultrasonographic examination of the hand and wrist and suggested that the modality could be used to depict the median nerve at the wrist level. Anatomic variants such as accessory branches, proximal bifurcations, and the presence of a persistent median artery can be identified, which is important before endoscopic release to avoid injury to the aberrant structures. Flattening or deformity of the nerve within the carpal tunnel, bulbous swelling of the nerve proximal to the tunnel, and palmar bowing of the flexor retinaculum might also suggest median nerve compression. Ultrasound can also be used to identify space-occupying lesions, which could be contributing to nerve compressions at the wrist. Color Doppler sonography can identify reactive hyperemia resulting from severe compression.321 The cross-sectional area of the median nerve shown by ultrasonographic examination has been positively correlated with electrophysiological derangements routinely associated with CTS.322,323 Although ultrasonography is still considered to be far from a standard diagnostic modality, a growing body of evidence suggests ultrasonography as an adjunct for diagnosis of CTS.324-326 Yassi327 reviewed the field of repetitive strain injuries and categorized the disorders according to affected part (tendon, muscle, joint, peripheral nerve, or blood vessel): Tendon-related disorders Tendonitis Tenosynovitis Stenosing tenosynovitis Periodontitis Ganglion cyst Epicondylitis (lateral or medial) 45 SRPS • Volume 11 • Issue R7 • 2015 Muscular disorders Focal dystonia Fibromyositis Tension-neck syndrome Myositis Myalgia Peripheral nerve entrapment Carpal tunnel syndrome Guyon tunnel syndrome Radial tunnel syndrome Pronator teres syndrome Cubital tunnel syndrome Joint and joint capsule disorders Osteoarthritis Bursitis Synovitis Adhesive capsulitis Neurovascular and vascular disorders Hand-arm vibration syndrome (Raynaud syndrome) Ulnar artery thrombosis The review article provides a comprehensive overview of the spectrum of repetitive strain injuries, their costs to society, and the role of the physician in preventing, recognizing, and managing these problems. It is important to differentiate nerve compression syndromes from other disorders that present with similar sensory disturbances. Careful attention should be given to patients who have experienced hand-arm vibration exposure, usually in work-related settings. Such patients are likely to complain of cold intolerance and difficulty with handwriting, picking up small objects, opening 46 lids, lifting, and carrying. Hand-arm vibration syndrome is a chronic condition that might not reverse when vibration exposure stops. Because it cannot be treated pharmacologically or surgically, prevention is the best approach.328-330 Pathophysiology The sequence of events that follows chronic compression of a peripheral nerve is clinically observed in the sensory system first as numbness or tingling, which can be measured by noting alterations in the cutaneous pressure threshold or cutaneous vibratory threshold.331 In the motor system, the first change noted by the patient is weakness of pinch and grip.331 Muscle atrophy occurs with progression of the compression. Rydevik et al.69 studied the effects of gradual compression on intraneural blood flow. At pressures of 20 to 30 mmHg, interference with venular flow occurred; at 40 to 50 mmHg, impairment of arteriolar and interfascicular capillary flow occurred; and at 60 to 80 mmHg, complete blockage of nerve perfusion occurred. The authors surmised that acute compression of a nerve can cause mechanical injury to the intraneural vessels and result in long-term impairment of circulation to that nerve. Rempel et al.332 reviewed the pathophysiology of nerve compression syndromes and provided the following conclusions from the literature regarding the role of tissue loads in the pathogenesis of peripheral nerve entrapment: •• Elevated extraneural pressures can, within minutes or hours, inhibit intraneural microvascular blood flow, axonal transport, and nerve function. This results in endoneurial edema with increased intrafascicular pressure and displacement of myelin. •• On the basis of animal models, lowmagnitude, short-duration extraneural pressure can initiate a process of nerve injury and repair and can cause structural tissue changes that persist for at least 1 month. The cascade of biological response to compression includes endoneurial edema, demyelination, inflammation, distal axonal SRPS • Volume 11 • Issue R7 • 2015 degeneration, fibrosis, growth of new Median Nerve axons, remyelination, and thickening of the CTS perineurium and endothelium. •• In healthy people, non-neutral positions of the fingers, wrist, and forearm and loading of the fingertips can elevate extraneural pressure in the carpal tunnel in a doseresponse manner. •• In a rat model, exposure of the hind limb to vibration causes intraneural edema, structural changes in myelinated and unmyelinated fibers of the sciatic nerve, and functional changes both in nerve fibers and non-neuronal cells. •• Exposure of vibrating hand tools at work can lead to permanent nerve injury with structural neuronal changes in the finger nerves and in the nerve trunks just proximal to the wrist. Mackinnon and Dellon21 and other authors319,333-335 reviewed current concepts of the pathophysiology of nerve compression and discussed the presenting symptoms, appropriate diagnosis, and management of entrapment neuropathies (Table 11). Mackinnon and Novak336 commented on the pathogenesis and treatment of cumulative trauma disorders, which, in their view, are complex disorders with multiple components. The authors hypothesized a multifactorial origin for cumulative trauma disorders, including organic causes such as neuropathic compression, myofascial pain, and muscle imbalance. These are overlaid by psychological and sociopolitical considerations having to do with repetitive tasks, low-prestige occupations, and insurance reimbursement patterns. Mackinnon and Novak recommended a correspondingly broad treatment plan that takes into account all areas of possible dysfunction and deals with them accordingly. Much has been written about CTS, which is now the most common entrapment disorder affecting peripheral nerves. Between 1% and 10% of the United States population will exhibit symptoms of carpal tunnel compression at some time. CTS results from repetitive prolonged wrist extension causing mechanical irritation, synovitis, and eventually compressive neuropathy of the median nerve. Certain occupations such as cashier, checker, butcher, and baker are associated with increased frequency of CTS symptoms (23% of grocery store workers).337 The condition also frequently occurs in rowers and bicyclists.338 The normal anatomy of the nerves and tendons at the level of the carpal tunnel was illustrated by Eversmann.339 As described, the carpal tunnel is bordered by the transverse carpal ligament volarly and by the bones of the wrist dorsally. The median nerve shares this space with nine flexor tendons. At the level of the carpal tunnel, the median nerve gives off a motor branch to the thenar eminence muscles, a branch to the first and second lumbricals, and sensory branches to the radial three and a half digits.16,340 Olave et al.341 performed a biometric study of the superficial palmar arch and of the communicating branch between the ulnar and median nerves as related to the flexor retinaculum. The authors found that in men, the distance between the distal wrist crease and the site at which the communicating branch originates from the ulnar component had an average of 34 ± 6 mm on the right side and 30 ± 8 mm on the left. The difference between the distal wrist crease and the junction of the communicating branch with the common palmar digital nerve of the third interosseous space was 44 ± 7 mm on the right and 40 ± 6 mm on the left. Conversely, in 15% of cases, the communicating branch was observed to emerge from the common palmar digital nerve of the third interosseous space. The distance between the retinaculum and the superficial palmar arch in the axial line of the fourth metacarpal bone was on average 7 ± 4 mm on the right and 8 ± 4 mm on the left. At the same level, the distance between the retinaculum and the communicating branch was 6 ± 4 mm on the right and 5 ± 3 mm on the left. The measurements in women were consistently smaller throughout (Tables 12 and 13).341 These 47 SRPS • Volume 11 • Issue R7 • 2015 Syndrome Median nerve Carpal tunnel syndrome Pronator syndrome Anterior interosseous nerve syndrome Table 11 Nerve Compression Syndromes Site of Compression Carpal tunnel Numbness radial three and a half digits, thenar atrophy, positive Tinel sign at wrist, positive Phalen sign, increased symptoms with tourniquet, decreased NCV Where median nerve passes deep to both Reproduction of proximal forearm pain heads of pronator teres, at thickening of on resistance to forearm pronation and lacertus fibrosus or flexor superficialis arch wrist flexion, possible median numbness, Tinel sign, EMG changes Where nerve crosses tendinous origin of deep head of pronator teres Supracondyloid process syndrome Bony spur 3 to 5 cm above medial epicondyle, ligament of Struthers between spur and condyle (1%), fascial band from biceps to volar forearm fascia Radial nerve Radial tunnel syndrome Signs and Symptoms Proximal to radial tunnel by fibrous bands, at takeoff of radial recurrent artery (vascular leash of Henry), at tendinous origin of ECRB, or at arcade of Frohse “Pinch attitude” of hand caused by paralysis of FPL and FDP to index and long fingers, no sensory component, varies if Martin-Gruber anastomosis is present Possible median nerve symptoms after trauma EDC to finger(s) paralyzed Posterior interosseous nerve syndrome Localized to the arcade of Frohse were the All EDC and FCU paralyzed, loss of MP posterior interosseous nerve pierces the extension, wrist in dorsoradial extension, two heads of the supinator no sensory component Superficial radial nerve syndrome Variable Pain in proximal forearm, hypesthesia of dorsal thumb At first rib or costoclavicular area Pain with neck rotation, variable sensory and/or motor symptoms Arcade of Struthers syndrome Where the ulnar nerve passes into the posterior compartment of the distal arm Variable sensory and/or motor symptoms Cubital tunnel syndrome Distal to medial epicondyle where nerve passes through tow heads of flexor carpi ulnaris, at common origin of FCU, FDP, and FDS Pure motor or sensory loss of ulnar one and a half fingers or combination sensory and motor impairment, possible slowed NCV Guyon canal syndrome In Guyon canal at pisiform or hook of hamate Loss of intrinsic muscle function to small and ring fingers with or without sensory loss Ulnar nerve Cervical syndromes NCV, nerve conduction velocity; EMG, electromyogram; FPL, flexor pollicis longus; FDP, flexor digitorum profundus; ECRB, extensor carpi radialis brevis; EDC, extensor digitorum communis; FCU, flexor carpi ulnaris; MP, metacarpophalangeal; FDS, flexor digitorum superficialis. 48 SRPS • Volume 11 • Issue R7 • 2015 Table 12 Distance from Distal Wrist Crease to Exit Level and to Junction Level of Communicating Branch341 Sex Junction Level (mean ± SD in mm) Exit Level (mean ± SD in mm) Right Left Right Left Male 33.9 ± 5.5 30.2 ± 8.2 43.6 ± 6.9 40.2 ± 6.2 Female 28.5 ± 6.2 27.1 ± 3.3 40.7 ± 7.8 34.4 ± 1.6 Table 13 Distance from Distal Margin of Flexor Retinaculum to Communicating Branch341 Communicating Branch Level (mean ± SD in mm) Sex Right Left Male 6.2 ± 3.7 5.1 ± 2.8 Female 5.3 ± 3.7 4.0 ± 1.9 measurements can be used as a reference during surgical procedures in the palmar tunnel, particularly ECTR. Lanz342 studied the anatomy of the median nerve at the carpal tunnel and identified three major patterns of branching of the recurrent motor branch: extraligamentous (46%–90%), subligamentous (31%), and transligamentous (23%). Amadio343 confirmed the anatomic work presented by Lanz342 in his own series of 275 carpal tunnel releases and noted that anomalies within the carpal tunnel are common. More recently, Szabo and Pettey344 reported a bilateral group III variation in which the radial-most branch of each median nerve occupied an accessory ligamentous compartment beneath its transverse carpal ligament. Green and Morgan345 further emphasized the anatomic variability of and potential for iatrogenic injury to the thenar motor branch. In their series of 1400 consecutive patients who underwent open carpal tunnel release (OCTR) through a thenar crease incision, 25.5% of patients displayed an extraligamentous or transligamentous course, which rendered the thenar motor branch more vulnerable to injury. The authors recommended caution when thenar muscle fibers are located superficial to or within the transverse carpal ligament, considering the incidence of anomalous motor branch was 93% in those cases. They reported numerous representative cases in which motor branch nerve fibers were clearly located within the standard field for OCTR and were vulnerable to inadvertent injury. Based on the findings presented by Green and Morgan, the precise location of the incision is extremely important in avoiding the vulnerable thenar 49 SRPS • Volume 11 • Issue R7 • 2015 motor branch nerve fibers. Kaplan346 presented a classic description of the thenar motor branch as lying at the intersection of axes defined by the radial border of the long finger and the “cardinal line,” which extends from the apex of the first web space parallel to the middle palmar crease. The location was recently clarified by Eskandari et al.347 in patients undergoing OCTR. Based on the radiographic measurements presented by Eskandari et al., the thenar motor branch is 12.6 mm ulnar and 4.4 mm proximal to the location described by Kaplan. Therefore, the motor branch is more in line with the mid-axis of the long finger than with its radial border, which is a cause for caution when using the traditional landmarks for OCTR. Finally, the anatomic definition of the cardinal line presented by Kaplan was reinvestigated by Vella et al.,348 indicating that it is best drawn from the apex of the first web space to the hook of the hamate. The authors confirmed the finding reported by Eskandari et al. that the cardinal line described by Kaplan is not a reliable landmark for localizing the thenar motor branch, but they indicated that the cardinal line is consistently 18 mm proximal to the superficial palmar arch. Whereas this topography is important for the safety of open releases, knowledge of the limitation of these landmarks is crucial in limited-incision and endoscopic techniques. Sugimoto et al.349 evaluated the blood supply to the median nerve using dynamic, contrast-enhanced MRI. The authors found significant circulatory disturbances in the median nerve of patients with CTS, suggesting that chronic hypoxia in the nerve rather than nerve compression alone produces the symptoms of CTS. The role of the lumbrical muscles in the cause of carpal tunnel disorder has been sporadically investigated for many years. In the 1970s, Eriksen350 and Jabaley351 reported symptoms of CTS associated with hypertrophy of a lumbrical muscle the origin of which was high enough to crowd the carpal tunnel during active finger flexion. Desjacques et al.352 noted that in nearly all cases of CTS that showed complete denervation of the thenar muscles, the first and second lumbricals maintained part of their innervation. Cobb et al.353 from the Mayo Clinic evaluated the significance of lumbrical muscle incursion within the carpal tunnel. The authors documented a progressive increase in carpal tunnel pressure for each degree of finger 50 flexion in cadaveric hands with intact lumbricals. This stood “in sharp contrast to a relatively stable carpal tunnel pressure during finger flexion for the group without lumbrical muscles.” The authors concluded that lumbrical muscle incursion into the carpal tunnel can result in elevation of carpal tunnel pressure and could be a cause of work-related CTS. Braun et al.354 described provocative stress testing for patients suspected of having CTS. Of 40 patients who were evaluated, 34 had increased hand volume but only 17 had impaired sensibility associated with the swelling, and those 17 were considered to have dynamic CTS requiring surgery. Osamura et al.355 began to elucidate the role of alterations in material properties of the carpal tunnel subsynovial connective tissue (SSCT) in patients with idiopathic CTS. They reported a mean shear modulus of 22.8 kPa in patients with CTS versus 2.7 kPa in normal control patients and mean maximum shear strength of 54.6 kPa in the CTS group versus 23.3 kPa in the control group. A more recent study using human cadaveric wrist models investigated the effect of high- (60 mm/s) and low- (2 mm/s) velocity tendon excursion of the SSCT. The results showed that increasing velocity lowers the threshold for SSCT damage with resistance energy greater at all excursions (P < 0.031).356 The authors suggested that this finding might be important in understanding SSCT and occupational links with CTS. Associated evidence suggests that digital motion at extremes of wrist flexion and extension might lead to shear injury to the SSCT and volume changes within the carpal tunnel.357 Much controversy exists regarding the most appropriate surgical technique for decompression of the carpal tunnel. A number of operative procedures have been recommended, from simple operative release of the flexor retinaculum at the wrist, to release and exploration of either the motor branch to the thenar muscles or the palmar cutaneous branch of the median nerve, to release and neurolysis of the median nerve. The various approaches are passionately defended by their practitioners. Open Carpal Tunnel Release—Steinberg and Szabo16 SRPS • Volume 11 • Issue R7 • 2015 argued for the traditional approach of open surgical decompression of the median nerve with full visualization of the transverse carpal ligament because of the potential for anomalous branching patterns of the nerve. Details of the “classic” technique they prefer are presented in their article. Ariyan and Watson358 reviewed their extensive experience with CTS and described their operative technique. Heckler and Jabaley359 emphasized that the treatment of CTS is nerve dissection and decompression rather than transverse carpal ligament release. Jakab et al.360 described a technique of transverse carpal ligament reconstruction in surgery for CTS that resulted in complete resolution of symptoms in 93% of 104 hands. The authors described the operation as requiring substantially more dissection, with release of Guyon canal and mobilization of the ulnar nerve and artery. Serra et al.361 described the site of a short incision through which it is possible to completely section the carpal ligament without damaging the carpal contents. The technique involves a specially designed, blunt-tipped, rigid cannula and three Senn-Miller retractors to guide the scalpel and avoid injury to the median nerve. The resulting incision is 1.5 to 2 cm long. The authors note that their approach combines the advantages of the endoscopic technique—minimal scar, no tenderness, early recovery— with those of the classic open technique—exploration of the carpal contents. First reported by Lee and Strickland362 in 1998, the Indiana Tome (Biomet Orthopaedics, Inc., Warsaw, IN) is an instrument that allows sectioning of the transverse carpal ligament through a limited palmar incision. The approach used is extrabursal (outside the synovial confluence of flexor tendons and median nerve), proceeding from distal to proximal over a protective guide. The technique was designed as a hybrid technique that offers the advantages of both open and endoscopic techniques by allowing direct visualization of the carpal tunnel contents and a limited incision. The results achieved with this technique were comparable to those achieved with other techniques, with 72.6% of patients experiencing complete relief and 19.6% experiencing near-complete relief. In the initial report of 694 hands, one partial and one complete median nerve transection occurred (0.29%) because of a second pass of the tome after difficulty with the initial pass. After that move was eliminated, no other transections occurred. Lee et al.363 reported 13-year outcomes for 1332 patients who had undergone carpal tunnel release with the Indiana Tome. The complication rate was 0.83%. Third common digital neurapraxia occurred most commonly. One can assume higher grade injury to the third common digital nerve in four patients (0.30%). Two of those cases were confirmed by surgical exploration, and two were unexplored but displayed persistent increase in TPD. The authors unequivocally stated the following: “The key to reducing complication in any minimally invasive technique, including the Indiana Tome, is to have a low threshold of converting to a standard open approach whenever the anatomy appears unclear or the technique does not proceed easily. It is the responsibility of all surgeons, whatever their degree of experience, to recognize this potential for complications and to do what is necessary to ensure patient safety.” Lowry and Follender364 evaluated the benefits of adjunctive interfascicular neurolysis in the treatment of severe CTS and found no significant difference between the study group (n = 25) and the control group (n = 25) who underwent standard ligament release alone. The authors concluded that no benefit is derived from adjunctive interfascicular neurolysis in carpal tunnel surgery. In the absence of substantial adhesions to the nerve, Seiler et al.365 also noted no improvement in nerve blood flow after epineurotomy. Mackinnon et al.366 agreed with that opinion on the basis of a prospective, randomized study comparing carpal tunnel release with and without internal neurolysis. The authors stated that the addition of internal neurolysis to division of the transverse carpal ligament does not add notable improvement in the sensory or motor outcomes for patients with primary CTS. A similar prospective comparative study by Blair et al.367 found no difference between the epineurotomy and non-epineurotomy groups in terms of TPD, physical and neurophysiological findings, and patient perception of outcome. These studies do not support the use of routine epineurotomy as an adjunctive procedure during carpal tunnel decompression. Similarly, it has been reported that simultaneous release of Guyon canal is no longer recommended for patients with carpal tunnel and paresthesias in the little 51 SRPS • Volume 11 • Issue R7 • 2015 finger. MRI findings showed that the dimensions of Guyon canal enlarge with carpal tunnel release alone.368 This finding has been clinically substantiated.16,369 Singh et al.370 surveyed the results of surgical decompression of the carpal tunnel in 265 patients (303 hands) who were diagnosed as having CTS. The femaleto-male ratio was 6:1, and 40 patients had bilateral involvement. Only 39% of hands showed numbness and paresthesias along the distribution of the median nerve. Tinel sign was positive in 63% of hands, and Phalen maneuver was positive in 67%. EMG studies were universally positive for carpal tunnel compression but were not conducted in every case (i.e., some patients were operated on solely on the basis of the clinical findings). A variety of operative procedures were performed, and all patients reported improvement in symptoms regardless of the type of surgery. The most common complication was a sensitive palmar scar, which bothered 62% of patients for up to 5 months postoperatively. Follow-up duration was 4 to 32 months (mean, 22 months). Lindau and Karlsson371 presented a 6-year followup of 91 patients undergoing OCTR. The results are presented in Table 14. Other complications of carpal tunnel release include infection, transection of the motor branch of the median nerve, injury to the palmar cutaneous branch of the median nerve, partial severance of the carpal ligament, pisotriquetral pain syndrome, RSD, interpillar pain, and transection of digital sensory nerves. The diagnosis and management of these and other complications were reviewed by Kuschner et al.372 Shurr et al.373 reported improvement in all clinical and electrodiagnostic studies after carpal tunnel release: motor and conduction velocities and TPD studies were significantly improved as early as 2 weeks postoperatively; sensory and motor latencies improved by 3 and 6 months; and pinch and grip strengths improved by 6 and 9 months (P < 0.05).374,375 In addition, ulnar nerve symptoms, present in 33% of patients with CTS, improved with carpal tunnel release alone.369 Wintman et al.376 correlated the severity of preoperative symptoms of CTS with surgical outcomes in 54 hands. The authors measured hand, wrist, and forearm pain, night pain and paresthesias, intermittent 52 paresthesias, hand clumsiness, hand weakness, constant numbness, and difficulty with work-related tasks before and after carpal tunnel release. Although all symptoms showed considerable improvement 3 months postoperatively, patients whose hand weakness, pain, numbness, and clumsiness were rated more severe showed less improvement of function and less satisfaction with overall symptom relief. Patients with preoperative night symptoms and intermittent paresthesias tended to be more satisfied with the results of surgery. Cartotto et al.377 reported two unusual but extremely serious complications of carpal tunnel release. One patient had massive necrosis of the palm, which required free flap coverage, and another had complete severance of the median nerve for which direct repair failed, necessitating grafting. Endoscopic Carpal Tunnel Release—Okutsu et al.378 and Chow379 first introduced ECTR in 1987 and 1989. Initial reception of the endoscopic approach was colored by clinical series performed with the original Agee device and using the transbursal technique, which were associated with complication rates four times higher than those associated with the current extrabursal technique.380 Since that time, a single-portal operation with the retooled Agee device381 and a dual-portal technique presented by Chow379 have been used extensively in the United States for carpal tunnel decompression. Nagle382 reviewed the literature of ECTR and compared its surgical outcomes with those of OCTR. Overall, the author found no difference between the two approaches in terms of tendon, artery, or nerve lacerations, hematomas, incomplete releases, or time to resolution of paresthesias or pain. In general, ECTR is more commonly associated with neurapraxia and stitch infection than is OCTR, but ECTR also leads to more rapid recovery of pinch and grip strength and wrist range of motion and less midpalm tenderness than does OCTR. In a presentation at the annual meeting of the American Society for Surgery of the Hand in 1995, Palmer and Toivonen383 reported the results of a survey of the membership regarding complications of ECTR and OCTR (Table 15). Brown et al.384 analyzed the results of carpal tunnel release through the open and endoscopic methods in SRPS • Volume 11 • Issue R7 • 2015 Table 14 Open Carpal Tunnel Release: Preoperative and 6-Year Postoperative Findings371 Preoperative (n = 85) 6 Years Postoperative (n = 91) Relative Risk (95% confidence interval) Numbness at night 78 29 0.35 (0.14−0.85) Numbness during day 63 29 0.43 (0.22−0.83) Weakness of grip 17 39 2.14 (1.09−4.21) Occasional pain at rest 24 19 0.74 (0.37−1.48) Decreased sensibility 15 11 0.68 (0.30−1.59) Decreased wrist motion 0 28 Sensitivity to cold 0 33 Pain with loading 0 35 Positive Phalen sign 61 9 0.14 (0.06−0.32) Positive Tinel sign 55 7 0.12 (0.05−0.29) Thenar atrophy 5 10 1.87 (0.61−5.71) Dysesthesia in scar 0 8 Pain in scar 0 1 Subjective symptoms Objective findings Table 15 Nerve Laceration Complications Associated with Endoscopic Carpal Tunnel Release and Open Carpal Tunnel Release383 Endoscopic (708 surgeons) Open (616 surgeons) 100 147 Ulnar nerve 88 29 Digital nerve 77 54 Tendon 69 19 Superficial arch 86 21 Ulnar artery 34 11 Laceration Median nerve 53 SRPS • Volume 11 • Issue R7 • 2015 169 hands and found no significant differences between the two groups regarding sensation, motor strength, or subjective relief of symptoms. The open technique was associated with more tenderness of the scar than was the endoscopic method. The open approach also resulted in longer time to return to work (median, 28 days) than did the endoscopic technique (median, 14 days). On the other hand, endoscopic release was associated with more frequent complications, such as partial nerve transections, contusions, and wound hematomas. The authors concluded that although function is restored faster when the endoscopic method is used, the higher rate of complications is a sign that intraoperative safety needs to be improved before ECTR can be performed on a widespread basis. Murphy et al.385 also reported a case of transection of the median nerve and pseudoaneurysm of the superficial palmar arch after ECTR. A review of the literature disclosed marked differences in return-to-work times of patients after carpal tunnel release by either the endoscopic or the open approach.380,381,386-389 Jimenez et al.389 provided an extensive review of published articles on ECTR and OCTR. The authors concluded that the reported success, complication, and failure rates of ECTR are acceptable and comparable to those of OCTR in adequately selected patients. Furthermore, many patients who underwent ECTR returned to work and activities of daily living sooner than did the patients who underwent OCTR.389 In addition, the patients who underwent ECTR reported less pain and tenderness during the postoperative period. The authors were unable to recommend one procedure over the other but preferred subligamentous or extrabursal techniques to transverse methods. Ultimately, surgeons should perform the procedure with which they are most comfortable. Thoma et al. compared cost and effectiveness of ECTR and OCTR using a decision analysis economic model. Based on medical costs from a Canadian university hospital, the calculation strongly favors OCTR performed in a day surgery unit when compared with ECTR performed in the main operating room. When both OCTR and ECTR are performed in the day surgery unit, the findings are equivocal depending on the variable occurrence of increased scar sensitivity (OCTR) or increased RSD (ECTR). Although the results were based on a cost structure and operating room setup that were 390 54 unique to a specific institution, the study indicated the important factors of surgery resource utilization, operating room equipment availability, and logistics when weighing the two procedures in the setting of limited health resources. A meta-analysis of randomized controlled trials conducted by Thoma et al.391 compared ECTR and OCTR (Fig. 24). Based on the 13 available Level I trials in the world literature, including five non-English language publications, the authors found support to favor ECTR in reduction of scar tenderness and increase in grip and pinch strength at 12-week follow-up examinations. Data were inconclusive for symptom relief and return to work. Reversible nerve damage was three times more likely to occur in ECTR cases, but irreversible nerve damage was rare with either technique (Fig. 24).391 The authors called for a multicenter randomized controlled trial to obtain a definitive comparison between the techniques. More recently, Atroshi et al.392 conducted a randomized trial comparing outcomes of OCTR and ECTR performed in 63 patients each with 5-year followup. No difference in outcomes was shown by the validated Carpal Tunnel Questionnaire (CTQ). Of note, persistent palm and scar tenderness were equal in both groups. A number of validated outcome assessment questionnaires have been used to compare outcomes in cases of CTS. Hand-specific outcome instruments include the CTQ, Michigan Hand Outcomes Questionnaire, and Disability of Arm, Shoulder and Hand Questionnaire (DASH). These have all been compared and shown to be responsive to change after carpal tunnel release.393-395 CTS in Children—Lamberti and Light396 reviewed the literature and described 64 cases of pediatric CTS. The most frequent age of diagnosis and treatment was between 6 and 8 years; however, some patients were as young as 2 years. Fifty percent of the cases were secondary to lysosomal storage disease, whereas 25% were considered idiopathic. Most children presented with modest complaints and were often described as being clumsy with their hands. Other complaints included wrist and hand pain, and older children often complained of paresthesias and nocturnal symptoms similar to those of adults with CTS. More than half the patients had evidence of thenar SRPS • Volume 11 • Issue R7 • 2015 Figure 24. Graphs show effect size and odds ratio for outcomes. OCTR, open carpal tunnel release. ECTR, Endoscopic carpal tunnel release. (Reprinted with permission from Thoma et al.384) 55 SRPS • Volume 11 • Issue R7 • 2015 atrophy, and, in some cases, it was severe enough to suggest the possibility of congenital absence or hypoplasia of the thenar muscles. Findings of provocative tests, such as Tinel and Phelan tests, often are normal for children with longstanding nerve compression, and electrodiagnostic studies are essential to establish the diagnosis. Pediatric cases tend to include bilateral electrophysiological abnormalities even if only one hand is clinically involved. Therefore, it is recommended that electrodiagnostic studies be performed bilaterally in all suspected pediatric cases. Causes of CTS in the review presented by Lamberti and Light396 include genetic abnormalities such as lysosomal storage disease, mucopolysaccharidosis, mucolipidosis, primary familial CTS, Schwartz-Jampel disorder, and hemophilia. Nongenetic causes include massive hemangiomatosis, macrodactyly, melorheostosis, sports-related activities, and aberrant anatomy. Sixty of the 64 cases underwent carpal tunnel release, and the vast majority of patients experienced either partial or complete recovery of sensation and strength. The authors implied that operative release is the only treatment documented to be effective in cases of pediatric CTS. Choudry et al.397 summarized a series of 32 children (age 16 years or younger) with CTS at the Mayo Clinic between 1974 and 2005. Overuse syndrome was the most frequent (44%) cause of CTS in that series, with less frequent causes being genetic conditions (25%), familial CTS (16%), trauma (13%), and ganglion cyst (3%). The mean patient age was 14 years, with a range of 5 to 16 years. The experience mirrors the findings presented by Lamberti and Light396 regarding surgery as the preferred treatment for CTS secondary to genetic disorders and trauma. However, Choudry et al. found conservative therapy to be effective in 79% of cases related to overuse syndrome, with only 14% of those cases requiring surgery. Recurrent CTS—Although the vast majority of patients are relieved of their symptoms after carpal tunnel decompression, Hunter398 noted an increasing number of patients (approximately 20% of those undergoing carpal tunnel surgery) who failed to return to the work force postoperatively because of recurrent CTS. Approximately 50% of those patients had histories of previous injury to the wrist, and Hunter speculated that something other 56 than simple compression neuropathy of the median nerve was at work. The median nerve might have been fixed in adhesions, either from the trauma or the failed surgery, and rapid-pace recovery programs might have aggravated the problem by forcing the hand into pronation and causing radial neuropathy. Such a complex of events could even lead to brachial plexus traction disorders. Hunter stated that the best approach to recurrent CTS is complete mobilization of the median nerve through the hand and wrist. O’Malley et al.399 recommended reexploration for patients with unrelieved carpal tunnel symptoms after release if they have positive Phalen test findings, nocturnal symptoms, symptoms exacerbated by activities, or a short or transverse initial incision. In the absence of these criteria, reexploration will not result in a satisfactory outcome. Rose et al.400 added a palmaris brevis turnover flap to internal neurolysis of the chronically scarred median nerve in cases of recurrent CTS. With that protocol, they documented improvement in strength, bulk, and sensibility in all hands in their study. Botte et al.401 identified the common causes of recurrent CTS and prescribed treatment as follows: •• For incomplete release of a transverse carpal ligament, conduct reexploration and repeat release of the transverse carpal ligament or perform incision or excision of the reformed transverse carpal ligament. •• For fibrous proliferation or scarring within the carpal tunnel, perform epineurolysis or use local muscle flaps or local or remote free fat grafts with or without internal neurolysis, perform excision or Z-plasty of painful scar, or insert a nerve barrier (e.g., silicone sheet, vein wrapping). •• For recurrent or exuberant flexor tenosynovitis, repeat tenosynovectomy or appropriate chemotherapy. Anterior Interosseous Syndrome Eversmann333 and Howard402 reviewed the anatomy, diagnosis, and treatment of the anterior interosseous syndrome. SRPS • Volume 11 • Issue R7 • 2015 Pronator Syndrome Olehnik et al.403 presented their experience with median nerve compression in the proximal forearm. The nerve might be impinged upon by the lacertus fibrosus, by vascular leashes across the nerve canal, or by abnormalities of the PT muscle or the FDS muscle. Less commonly, the median nerve is pinched beneath the ligament of Struthers from a supracondylar process on the distal humerus. Nerve conduction testing is ineffective for confirming clinical impression of proximal median nerve entrapment. The treatment of choice for this disorder is surgical release of the median nerve in the proximal forearm. Radial Nerve At the level of the elbow, the radial nerve can be compromised at four different sites along its submuscular course by the following: 1) fibrous bands proximal to the radial tunnel; 2) the vascular leash of Henry (radial recurrent artery); 3) the tendinous margin of the ECRB; or 4) the arcade of Frohse (the proximal edge of the superficial supinator), where the posterior interosseous nerve travels between the superficial and deep heads of the supinator.333,404 Radial Tunnel Syndrome Compression of the radial nerve at the level of the elbow manifests clinically as radial tunnel syndrome.333,402 This condition is distinct from lateral epicondylitis, or “tennis elbow,” which also exhibits pain in the region of the mobile wad and common extensor origin.405 Anatomically, the radial tunnel contents include both the superficial radial nerve (the primary sensory branch) and the motor component of the radial nerve (nerves to brachioradialis, ECRL, ECRB, supinator, and posterior interosseous nerve). Symptoms are exertional pain in the extensorsupinator muscle mass in the proximal forearm that can radiate distally, with paresthesia in the dorsal radial aspect of the hand; patients also frequently complain of a weak grip.332,402 EMG studies are not diagnostic unless marked denervation is present,333 but radial nerve block is prognostically helpful: Ritts et al.404 noted excellent surgical results in patients who experienced relief of pain when their radial nerve was anesthetized. In a recent study of the long-term results of surgical decompression of the radial tunnel by a brachioradialis muscle-splitting approach, Jebson and Engber406 noted that complete pain relief and return to activities after radial tunnel surgery is not as predictable as previous studies indicated. The authors tracked the functional results of surgical release in 24 extremities for an average of 8 years after surgery. The criteria outlined by Roles and Maudsley407 and Ritts et al.404 were used to determine outcomes. Based on the method presented by Roles and Maudsley, eight extremities (33%) were judged to be fair or poor. Based on the method presented by Ritts et al., seven extremities (29%) were judged to be fair or poor. Five patients changed their occupations because of continued discomfort. These findings are in line with the 30% fair or poor results reported by Lawrence et al.408 in a retrospective review of 30 decompressions of the radial nerve. Posterior Interosseous Nerve Syndrome Compression of the posterior interosseous nerve at the fibrous arcade of Frohse gives rise to the posterior interosseous nerve syndrome. The nerve supplies branches to the extensor digitorum, extensor digiti minimi, extensor carpi ulnaris, abductor pollicis longus, EPL, extensor pollicis brevis, and EIP muscles.402 Symptoms are paresis of the hand with loss of finger extension, loss of thumb abduction, and radial deviation of the hand during wrist extension. The radial tunnel and arcade of Frohse can be approached anteriorly or posteriorly. When the compression neuropathy can be localized to the arcade of Frohse, Mackinnon and Dellon21 prefer a lazy-S musclesplitting incision extending posteriorly between the brachioradialis muscle and the ECRL (Fig. 25).404 Patients with radial nerve entrapment syndrome that cannot be localized to the arcade of Frohse are best served by a zigzag anterolateral incision to expose the radial tunnel for exploration.333 Kotani et al.409 reported four cases of posterior interosseous nerve paralysis with multiple constrictions. At the time of surgery, the constrictions were found between the arcade of Frohse and a point of bifurcation of the supinator motor branch. External neurolysis with epineurotomy under the microscope was performed in all cases, and full recovery was obtained. 57 SRPS • Volume 11 • Issue R7 • 2015 7 1 2 3 8 9 4 10 5 6 Figure 25. Sites of compression of the posterior interosseous nerve in association with radial tunnel syndrome. 1, radial nerve; 2, brachialis; 3, brachioradialis; 4, arcade of Frohse; 5, extensor carpi radialis brevis; 6, superficial radial nerve; 7, bicep; 8, median nerve; 9, radial recurrent artery; 10, fibrous bands. (Modified from Ritts et al.397) Ulnar Nerve Mackinnon and Dellon,21 Eversmann,333 and Leffert410 reviewed the anatomy, diagnosis, and treatment of ulnar neuropathies. Cubital Tunnel Syndrome The ulnar nerve lies on the medial head of the triceps muscle, enters the cubital tunnel behind the medial epicondyle, and continues distally beneath the arcade of fascia joining the heads of the FCU (Fig. 26).338,411 The nerve might be constricted in its course through the cubital tunnel at the level of the elbow. Ulnar nerve constriction manifests as pain on the medial side of the proximal forearm and paresthesias, dysesthesias, or anesthesia in the little and ring fingers.412 Clinical signs and a positive percussion test over the ulnar nerve are diagnostic of entrapment; nerve conduction velocity studies are inconclusive.333 When the ulnar nerve is squeezed by the aponeurosis of the FCU, surgical decompression 58 Figure 26. Zigzag incision for anterolateral approach to radial tunnel when compression cannot be localized to arcade of Frohse. (Modified from Eversmann.327) alone is sufficient for treatment.333-413 Patients for whom decompression has failed or whose ulnar neuropathy is the result of trauma, cyst, or congenital anomaly are candidates for anterior transposition of the ulnar nerve. Learmonth414 described the submuscular technique of anterior transposition, and Leffert410 recounted his experience with anterior submuscular transposition of the ulnar nerve in 38 patients. Ulnar nerve mobilization necessitates decompression of the cubital tunnel, fasciotomy of the FCU, and dissection along the ulnar nerve for at least 8 cm proximal to the medial epicondyle.413 Controversy surrounds the selection of an appropriate technique for ulnar nerve decompression in each individual patient.415 Numerous prospective randomized controlled trials have shown comparable results between simple decompression and either SRPS • Volume 11 • Issue R7 • 2015 submuscular or subcutaneous transposition.416-419 Bartels et al.416 found that simple decompression has a lower complication rate when compared with subcutaneous transposition (9.6% versus 31.1%), with sensibility loss around the scar and superficial infection occurring most commonly. In a companion study, the same group showed that total costs were substantially lower for simple decompression (1124 Euros versus 2730 Euros).420 Limitations of those studies were failure to include all treatment modalities and inadequate distribution of patients with mild, moderate, and severe symptom staging. A meta-analysis was conducted by Mowlavi et al.421 Based on an evaluation of 30 studies, the authors determined that patients with mild stage cubital tunnel syndrome benefit equally from nonoperative and all surgical treatments, those with moderate stage respond best to submuscular transposition, and those with severe stage show inconsistent efficacy for all available modalities, with medial epicondylectomy displaying the poorest result. Lowe and Mackinnon422 emphasized that all procedures have pitfalls that can lead to an overall recurrence or failure rate of 25% after surgery. The authors inferred the importance of expert technique and awareness of potential problems in performing the surgical modality of choice. Race and Saldana423 described the anatomic course of the medial cutaneous nerves of the arm, the fibers of which overlie the medial epicondyle and supply the skin over the olecranon. Because the standard incision used for ulnar nerve transposition severs the terminal branches of these cutaneous nerves, the authors recommended a posterior approach for transposition of the ulnar nerve. Lowe et al.424 further characterized these branches in clinical practice by using a standard approach posterior to the medial humeral epicondyle. In those cases, a branch of the medial antebrachial cutaneous nerve crossed the incision line 1.8 cm proximal to the epicondyle 61% of the time and 3.1 cm distal to the epicondyle 100% of the time (Fig. 27). Meticulous dissection is required at the time of surgery to preserve these branches despite use of a posterior approach. Ulnar Tunnel Syndrome In its passage from forearm to hand, the ulnar neurovascular bundle crosses the wrist by way of Guyon canal. The roof of the space is the volar carpal ligament, which begins at the pisiform and extends radially to the hook of the hamate and attaches to the transverse carpal ligament. The floor of Guyon canal consists of the muscles of the hypothenar eminence, their fibers of origin, and the flexor retinaculum. Fatty tissue separates the proximal and distal segments of the roof of the canal.1376,333,334 Compression of the ulnar nerve in Guyon canal at the wrist is known as ulnar tunnel syndrome. Clinical signs are varied, and the site of the lesion determines whether both motor and sensory abnormalities exist or whether only motor or sensory paralysis is present.425 The latter is rare.425 The most common cause of Guyon canal entrapment is a carpal ganglion, and the second most common cause is repeated trauma to the hypothenar area, usually related to occupation.425 Treatment is by surgical exploration of Guyon canal, decompression, and resection of any obstructing mass (e.g., a ganglion).425 König et al.426 studied the hypothenar region in 23 cadaveric hands and noted that the contents of Guyon canal exit through two distinct areas termed the deep distal hiatus and the superficial distal hiatus. The deep motor branch of the ulnar nerve, with its deep branch of the ulnar artery and venae comitantes, which anastomose with the deep palmar vascular arch, runs beneath the opponens digiti minimi and flexor digiti minimi through the deep distal hiatus. This deep ulnar neurovascular bundle exits deep toward the midpalmar space, where the finger flexors lie and, in 61% of specimens, is bound by a potentially compressive fibrous arcade. The clinical implication of this finding is that when a release of the ulnar nerve is indicated, surgery should not only open the roof of Guyon space to release the superficial sensory branch to the ring and small fingers but also the fibrous arcade of the deep distal hiatus to release the deep motor branch to the intrinsic muscles. Otherwise, intrinsic weakness is very likely to be incompletely treated. Traction Neuritis Occasionally, patients develop persistent pain after OCTR or transposition of the ulnar nerve at the elbow. The pain might be the result of a neuroma of one of the subcutaneous nerves in the area, incomplete release of the anatomic structure causing compression of the nerve, scarring of the nerve to adjacent structures, or 59 SRPS • Volume 11 • Issue R7 • 2015 Figure 27. Standard 8-cm surgical incision for primary cubital tunnel surgery. a, average distance of proximal crossing branch from medial epicondyle is 1.8 cm; b, average distance of distal branch from medial epicondyle. (Reprinted with permission from Lowe et al.417) devascularization of the nerve. Conventional treatment involves initial reexploration of the nerve and external neurolysis and then epineurotomy, epineurectomy, or internal neurolysis. Most patients improve after surgery, but a small group of patients are virtually crippled by debilitating pain. The pain usually is exacerbated by direct pressure over the incision or by flexion and extension of the adjacent joints. The patients who experience this pain have developed traction neuritis because of adhesive scarring of the nerve, either to the overlying skin or to the underlying tendons, which can progress to interfascicular scarring and segmental devascularization of the nerve. Typical examples include the following: 1) traction 60 neuritis of the median nerve after OCTR, sometimes referred to as adhesive median neuritis427 or epineural fibrous fixation,398 which is treated by secondary reexploration and neurolysis; and 2) traction neuritis of the ulnar nerve after unsuccessful cubital tunnel decompression, treated by either medial epicondylectomy, subcutaneous, or submuscular transposition.428 The concept of circumferential wrapping of a peripheral nerve involved in end stage traction neuritis with highly vascularized free flaps or pedicled flaps evolved out of frustration with the failure of conventional treatment and the successful precedent of relieving the SRPS • Volume 11 • Issue R7 • 2015 pain of brachial plexus neuritis by coverage with greater omentum. Circumferential wrapping might cushion the nerve from external pressure on the overlying skin, might isolate the nerve from the traction forces of adjacent moving tendons to allow improved gliding of the nerve, and might promote revascularization of a scarred nerve. In a report by Jones et al.428 nine patients with chronic severe pain caused by end stage traction neuritis of an intact peripheral nerve underwent external neurolysis and epineurectomy or epineurectomy and internal neurolysis and then circumferential wrapping of the devascularized segment of nerve. The flap was transferred on a pedicle or by microvascular anastomosis and consisted of subcutaneous adipose tissue, fascia, or muscle. During a follow-up period ranging from 1 to 5 years, seven patients experienced substantial relief of pain whereas two patients had no decrease in pain. The two patients without improvement had pure-fascia radial forearm fascial flaps without any overlying skin. Jones et al.428 expressed reservations regarding the effectiveness of such a flap compared with subcutaneous fatty tissue or muscle and discussed the pros and cons of various flap tissues and territories for circumferential nerve wrapping in the treatment of traction neuritis. Recent enthusiasm for using collagen-based nonvascularized materials for wrapping nerves affected by traction neuritis remains relatively unsubstantiated in clinical or scientific practice.429 Experimental studies conducted by Magill et al.430 using a rat model indicated that Seprafilm (Genzyme Corporation, Cambridge, MA), an antiadhesive hyaluronic acid and carboxymethylcellulose membrane sheet, is safe for use in nerve wrapping and might have benefits for decreasing perineurial scar bands. Multiple Nerves The term double crush syndrome is used to characterize the case of a single nerve compressed at two separate levels.431,432 According to a definition presented by Mackinnon,433 multiple crush syndrome implies compression of a single nerve at multiple sites or multiple structures compressing a nerve within a single anatomic area. The three major nerves of the upper extremity can be subject to multiple compressions as they pass through the structures of the forearm into the wrist and hand.431 Mackinnon433 reported that the histological changes in chronic nerve compression are slow but progressive. According to Mackinnon, once the changes are established, unless the cause is a systemic one that can be alleviated, the process will progress. Ideally, patients with multiple entrapment neuropathy or multiple crush syndrome are treated with job modification and education to avoid positions that exacerbate pressure on the entrapment sites. Should the modifications fail to eliminate the complaints, more distal surgical procedures are recommended and frequently obviate the more complex thoracic outlet decompression.433 Multiple compressions along a nerve have a cumulative effect on conduction, both antegrade and retrograde. The proximal source of compression can be subclinical yet partially responsible for the compression symptoms of more distal parts. Strict attention to the patient’s history and physical findings permits separation of the multiple points of compression and facilitates the choice of a site for surgical decompression.431 61 SRPS • Volume 11 • Issue R7 • 2015 REFERENCES 1.Gutowski KA. Hand II: Peripheral Nerves and Tendon Transfers. vol 9. Dallas: University of Texas; 2003. 2.Van Beek AL, Jacobs SC, Zook EG. Examination of peripheral nerves with the scanning electron microscope. Plast Reconstr Surg 1979;63:509–519. 3.Lundborg G. The intrinsic vascularization of human peripheral nerves: Structural and functional aspects. J Hand Surg Am 1979;4:34–41. 4.Sunderland S. The intraneural topography of the radial, median, and ulnar nerves. Brain 1945;68:243–299. 5.Jabaley ME, Wallace WH, Heckler FR. Internal topography of major nerves of the forearm and hand: A current view. J Hand Surg Am 1980;5:1–18. 6.Williams HB, Jabaley ME. The importance of internal anatomy of the peripheral nerves to nerve repair in the forearm and hand. Hand Clin 1986;2:689–707. 7.O’Daly JA, Imaeda T. Electron microscopic study of Wallerian degeneration in cutaneous nerves caused by mechanical injury. Lab Invest 1967;17:744–766. 8.Kline DG, Hudson AR. Selected recent advances in peripheral nerve injury research. Surg Neurol 1985;24: 371–376. 9.Ganong WF. Review of Medical Physiology. 7th ed. Los Altos: Lange Medical Publications; 1975. 10.Terzis JK, Publicover N. Clinical electrophysiologic peripheral nerve recordings. Clin Plast Surg 1984;11:47–52. 11.Van Beek A, Hubble B, Kinkead L, Torros S, Suchy H. Clinical use of nerve stimulation and recording techniques. Plast Reconstr Surg 1983;71:225–240. 12.Wilbourn AJ. The electrodiagnostic examination with peripheral nerve injuries. Clin Plast Surg 2003;30:139–154. 13.McCarthy JG, Galiano RD, Boutros SG. Brachial plexus. Current Therapy in Plastic Surgery. Philadelphia; Saunders: 2005. 14.Matloub HS, Yousif NJ. Peripheral nerve anatomy and innervation pattern. Hand Clin 1992;8:201–214. 15.Hobbs RA, Magnussen PA, Tonkin MA. Palmar cutaneous branch of the median nerve. J Hand Surg Am 1990;15:38–43. 16.Steinberg DR, Szabo RM. Anatomy of the median nerve at the wrist: Open carpal tunnel release: Classic. Hand Clin 1996;12:259–269. 17.Sunderland S. Degeneration, regeneration: A classification of nerve injury. In, Sunderland S (ed): Nerves and Nerve Injuries. 2nd ed. Edinburgh: Churchill Livingstone; 1978:69–141. 62 18.Thomson A. Third annual report of the Committee of Collective Investigation of the Anatomical Society of Great Britain and Ireland for the year 1891–1892. J Anat Physiol 1893;27:183–194. 19.Leibovic SJ, Hastings H II. Martin-Gruber revisited. J Hand Surg Am 1992;17:47–53. 20.Spinner M. Injuries to the Major Branches of the Peripheral Nerves of the Forearm. Philadelphia: WB Saunders; 1978: 245–252. 21.Mackinnon SE, Dellon AL. Surgery of the Peripheral Nerve. New York: Thieme Medical Publishers; 1988. 22.Budak F, Gönenç Z. Innervation anomalies in upper and lower extremities (an electrophysiological study). Electromyogr Clin Neurophysiol 1999;39:231–234. 23.Stančić MF, Mićović V, Potocnjak M. The anatomy of the Berrettini branch: Implications for carpal tunnel release. J Neurosurg 1999;91:1027–1030. 24.McCabe SJ, Kleinert JM. The nerve of Henlé. J Hand Surg Am 1990;15:784–788. 25.Allodi I, Udina E, Navarro X. Specificity of peripheral nerve regeneration: Interactions at the axon level. Prog Neurobiol 2012;98:16-37. 26.Seddon HJ. Three types of nerve injury. Brain 1943;66:237–288. 27.Mackinnon SE, Colbert SH. Nerve transfers in the hand and upper extremity surgery. Tech Hand Up Extrem Surg 2008;12:20–33. 28.Waller A. Experiments on the section of the glossopharyngeal and hypoglossal nerves of the frog, and observations of the alterations produced thereby in the structure of their primitive fibers. Philosoph Trans R Soc Lond (Biol) 1850;140:423–429. 29.Terenghi G. Peripheral nerve regeneration and neurotrophic factors. J Anat 1999;194:1-14. 30.Terenghi G, Hart A, Wiberg M. The nerve injury and the dying neurons: diagnosis and prevention. J Hand Surg Eur Vol 2011;36:730-734. 31.Terzis JK. Clinical microsurgery of the peripheral nerve: The state of the art. Clin Plast Surg 1979;6:247–267. 32.Omer GE Jr. Physical diagnosis of peripheral nerve injuries. Orthop Clin North Am 1981;12:207–228. 33.Bralliar F. Electromyography: Its use and misuse in peripheral nerve injuries. Orthop Clin North Am 1981;12: 229–238. 34.Stewart JD. Electrodiagnostic techniques in the evaluation of nerve compressions and injuries in the upper limb. Hand Clin 1986;2:677–687. SRPS • Volume 11 • Issue R7 • 2015 35.Frykman GK, Wolf A, Coyle T. An algorithm for management of peripheral nerve injuries. Orthop Clin North Am 1981;12:239–244. neurotrophic functions. In, Burnstock G, Vrbova G, and O’Brian R (ed): Somatic and Autonomic Nerve-Muscle Interactions. Amsterdam: Elsevier; 1982:213–252. 36.Sable AW. Median and ulnar nerves in the hand. Phys Med Rehabil Clin North Am 1998;9:737–753. 52.Onne L. Recovery of sensibility and sudomotor activity in the hand after nerve suture. Acta Chir Scand Suppl 1962;[suppl 300]:1–69. 37.Matsen FA III, Krugmire RB Jr. Compartmental syndromes. Surg Gynecol Obstet 1978;147:943–949. 38.Rorabeck CH, Castle GS, Hardie R, Logan J. Compartmental pressure measurements: An experimental investigation using the slit catheter. J Trauma 1981;21: 446–449. 39.Rowland SA. Fasciotomy: The treatment of compartment syndrome. In, Green DP (ed): Operative Hand Surgery. 2nd ed, vol 1. New York: Churchill Livingstone; 1988:665–707. 40.Duckworth AD, Mitchell SE, Molyneux SG, White TO, Court-Brown CM, McQueen MM. Acute compartment syndrome of the forearm. J Bone Joint Surg Am 2012;94:e63. 41.McQueen MM, Duckworth AD, Aitken SA, CourtBrown CM. The estimated sensitivity and specificity of compartment pressure monitoring for acute compartment syndrome. J Bone Joint Surg Am 2013;95:673-677. 42.McQuarrie IG. Peripheral nerve surgery: Today and looking ahead. Clin Plast Surg 1986;13:255–268. 43.Politis MJ. Specificity in mammalian peripheral nerve regeneration at the level of the nerve trunk. Brain Res 1985;328:271–276. 44.Varon S, Adler R. Nerve growth factors and control of nerve growth. Curr Top Dev Biol 1980;16:207–252. 45.Varon S, Adler R. Trophic and specifying factors directed to neuronal cells. Adv Cell Neurobiol 1981;2:115–163. 46.Lundborg G, Dahlin LB, Danielsen N, Hansson HA, Johannesson A, Longo FM, Varon S. Nerve regeneration across an extended gap: A neurobiological view of nerve repair and the possible involvement of neuronotrophic factors. J Hand Surg Am 1982;7:580–587. 47.Lundborg G, Longo FM, Varon S. Nerve regeneration model and trophic factors in vivo. Brain Res 1982;232: 157–161. 48.Varon S, Lundborg G. In, Haber B, Perez-Polo R, and Giuffrida AM (ed): Control of Nervous System Regeneration. New York: A Liss; 1982. 49.Levi-Montalcini R. The nerve growth factor: Its mode of action on sensory and sympathetic nerve cells. Harvey Lect 1966;60:217–259. 50.Varon S. Nerve growth factor and its mode of action. Exp Neurol 1975;48:75–92. 51.Varon S, Skaper SD. Nerve growth factor and 53.Madden JW, Peacock EE Jr. Some thoughts on repair of peripheral nerves. South Med J 1971;64:17–21. 54.Millesi H. Interfascicular nerve grafting. Orthop Clin North Am 1981;12:287–301. 55.Zachary LS, Dellon AL, Seiler WA IV. Relationship of intraneural damage in the rat sciatic nerve to the mechanism of injury. J Reconstr Microsurg 1989;5:137–140. 56.Dellon AL. Resection: Nerve repair’s most neglected technique. Plast Surg Tech 1995;1:191–199. 57.Cabaud HE, Rodkey WG, Nemeth TJ. Progressive ultrastructural changes after peripheral nerve transection and repair. J Hand Surg Am 1982;7:353–365. 58.Ducker TB, Kempe LG, Hayes GJ. The metabolic background for peripheral nerve surgery. J Neurosurg 1969;30:270–280. 59.Grabb WC. Median and ulnar nerve suture: An experimental study comparing primary and secondary repair in monkeys. J Bone Joint Surg Am 1968;50:964–972. 60.Kline DG, Hackett ER. Reappraisal of timing for exploration of civilian peripheral nerve injuries. Surgery 1975;78:54–65. 61.Van Beek A, Glover JL, Zook E. Primary versus delayedprimary neurorrhaphy in rat sciatic nerve. J Surg Res 1975;18:335–339. 62.Hatano E. A comparative study on primary and secondary nerve repair. Plast Reconstr Surg 1981;68:760–767. 63.Kleinert HE, Griffin JM. Techniques of nerve anastomosis. Orthop Clin North Am 1973;4:907–915. 64.Holmes W, Young JZ. Nerve regeneration after immediate and delayed suture. J Anat 1942;77:63–96. 65.Young L, Wray RC, Weeks PM. A randomized prospective comparison of fascicular and epineural digital nerve repairs. Plast Reconstr Surg 1981;68:89–93. 66.Gutmann E, Guttmann L. Factors affecting recovery of sensory function after nerve lesions. J Neurol Psychiatry 1942;5:117–129. 67.Highet WB, Sanders FK. The effects of stretching nerves after suture. Br J Surg 1943;30:355–371. 68.Miyamoto Y, Watari S, Tsuge K. Experimental studies on the effects of tension on intraneural microcirculation in sutured peripheral nerves. Plast Reconstr Surg 1979;63: 63 SRPS • Volume 11 • Issue R7 • 2015 398–403. the Hand. J Hand Surg Am 1979;4:201–211. 69.Rydevik B, Lundborg G, Nordborg C. Intraneural tissue reactions induced by internal neurolysis: An experimental study on the blood-nerve barrier, connective tissues and nerve fibres of rabbit tibial nerve. Scand J Plast Reconstr Surg 1976;10:3–8. 84.Cabaud HE, Rodkey WG, McCarroll HR Jr, Mutz SB, Niebauer JJ. Epineurial and perineurial fascicular nerve repairs: A critical comparison. J Hand Surg Am 1976;1: 131–137. 70.Smith JW. Factors influencing nerve repair: I. Blood supply of peripheral nerves. Arch Surg 1966;93:335–341. 71.Vasconez LO, Mathes SJ, Grau G. Direct fascicular repair and interfascicular nerve grafting of median and ulnar nerves in the rhesus monkey. Plast Reconstr Surg 1976;58:482–489. 72.Terzis J, Faibisoff B, Williams B. The nerve gap: Suture under tension vs. graft. Plast Reconstr Surg 1975;56:166–170. 73.Millesi H. The nerve gap: Theory and clinical practice. Hand Clin 1986;2:651–663. 74.McLellan DL, Swash M. Longitudinal sliding of the median nerve during movements of the upper limb. J Neurol Neurosurg Psychiatry 1976;39:566–570. 75.Mackinnon SE. Surgical management of the peripheral nerve gap. Clin Plast Surg 1989;16:587–603. 76.Manders EK, Saggers GC, Diaz-Alonso P, Finn L, Sipio JC, Glumac T, Au VK, Wong RK, Mottaleb M. Elongation of peripheral nerve and viscera containing smooth muscle. Clin Plast Surg 1987;14:551–562. 77.Weber RA, Breidenbach WC, Brown RE, Jabaley ME, Mass DP. A randomized prospective study of polyglycolic acid conduits for digital nerve reconstruction in humans. Plast Reconstr Surg 2000;106:1036–1045. 78.Rinker BD, Ingari JV, Greenberg JA, Thayer WP, Safa B, Buncke GM. Outcomes of short-gap sensory nerve injuries reconstructed with processed nerve allografts from a multicenter registry study. J Reconstr Microsurg 2015;Apr 20. [Epub ahead of print] 79.Taha A, Taha J. Results of suture of the radial, median, and ulnar nerves after missile injury below the axilla. J Trauma 1998;45:335–339. 80.Deutinger M, Girsch W, Burggasser G, Windisch A, Joshi D, Mayr N, Freilinger G. Peripheral nerve repair in the hand with and without motor sensory differentiation. J Hand Surg Am 1993;18:426–432. 85.Orgel MG. Epineurial versus perineurial repair of peripheral nerves. Clin Plast Surg 1984;11:101–104. 86.Bora FW Jr. Peripheral nerve repair in cats: The fascicular stitch. J Bone Joint Surg Am 1967;49:659–666. 87.Goto Y. Experimental study of nerve autografting by funicular suture [in Japanese]. Nippon Geka Hokan 1967;36:478–494. 88.Grabb WC, Bement SL, Koepke GH, Green RA. Comparison of methods of peripheral nerve suturing in monkeys. Plast Reconstr Surg 1970;46:31–38. 89.Wise AJ Jr, Topuzlu C, Davis P, Kaye IS. A comparative analysis of macro- and microsurgical neurorrhaphy techniques. Am J Surg 1969;117:566–572. 90.Yamamoto K. A comparative analysis of the process of nerve regeneration following funicular and epineurial suture for peripheral nerve repair [in Japanese]. Nippon Geka Hokan 1974;43:276–301. 91.Brushart TM, Tarlov EC, Mesulam MM. Specificity of muscle reinnervation after epineurial and individual fascicular suture of the rat sciatic nerve. J Hand Surg Am 1983;8:248–253. 92.Orgel MG, Terzis JK. Epineurial vs. perineurial repair: An ultrastructural and electrophysiological study of nerve regeneration. Plast Reconstr Surg 1977;60:80–91. 93.Jabaley ME. Current concepts of nerve repair. Clin Plast Surg 1981;8:33–44. 94.Gruber H. Identification of motor and sensory funiculi in cut nerves and their selective reunion. Br J Plast Surg 1976;29:70–73. 95.Engel J, Ganel A, Melamed R, Rimon S, Farine I. Choline acetyltransferase for differentiation between human motor and sensory nerve fibers. Ann Plast Surg 1980;4:376–380. 96.Riley DA, Lang DH. Carbonic anhydrase activity of human peripheral nerves: A possible histochemical aid to nerve repair. J Hand Surg Am 1984;9:112–120. 81.Daniel RK, Terzis JK. Reconstructive Microsurgery. Boston: Little, Brown; 1977. 97.Trumble TE, McCallister WV. Repair of peripheral nerve defects in the upper extremity. Hand Clin 2000;16:37–52. 82.Langley JN, Hashimoto M. On the suture of separate nerve bundles in a nerve trunk and on internal nerve plexuses. J Physiol 1917;51:318–346. 98.Maki Y, Firrell JC, Breidenbach WC. Blood flow in mobilized nerves: Results in a rabbit sciatic nerve model. Plast Reconstr Surg 1997;100:627–633. 83.Sunderland S. The pros and cons of funicular nerve repair: Founder’s lecture: American Society for Surgery of 99.Weber RV, Mackinnon SE. Bridging the neural gap. Clin Plast Surg 2005;32:605–616. 64 SRPS • Volume 11 • Issue R7 • 2015 100.Rose EH, Kowalski TA. Restoration of sensibility to anesthetic scarred digits with free vascularized nerve grafts from the dorsum of the foot. J Hand Surg Am 1985;10: 514–521. 101.Huber GC. A study of the operative treatment for loss of nerve substance in peripheral nerves. J Morphol 1895;11:629–740. 102.Forman DS, Berenberg RA. Regeneration of motor axons in the rat sciatic nerve studied by labeling with axonally transported radioactive proteins. Brain Res 1978;156:213–225. 103.Forman DS, Wood DK, DeSilva S. Rate of regeneration of sensory axons in transected rat sciatic nerve repaired with epineurial sutures. J Neurol Sci 1979;44:55–59. 104.Guth L, Jacobsen S. The rate of regeneration of the vagus nerve of the cat. Exp Neurol 1966;14:439–447. 105.Sunderland S. Factors influencing the course of regeneration and the quality of the recovery after nerve suture. Brain 1952;75:19–54. 106.Gutmann E, Young JZ. The re-innervation of muscle after various periods of atrophy. J Anat 1944;78:15–43. 107.Brushart TM, Mesulam MM. Alteration in connections between muscle and anterior horn motor neurons after peripheral nerve repair. Science 1980;208:603–605. 108.Lundborg G, Rosén B. Sensory relearning after nerve repair. Lancet 2001;358:809–810. 109.Goheen-Robillard B, Myckatyn TM, Mackinnon SE, Hunter DA. End-to-side neurorrhaphy and lateral axonal sprouting in a long graft rat model. Laryngoscope 2002;112:899–905. 110.Hadlock T, Sheahan T, Heaton J, Sundback C, Mackinnon S, Cheney M. Baiting the cross-face nerve graft with temporary hypoglossal hookup. Arch Facial Plast Surg 2004;6:228–233. 111.Rab M, Koller R, Haslik W, Kamolz LP, Beck H, Meggeneder J, Frey M. The influence of timing on the functional and morphological result after nerve grafting: An experimental study in rabbits. Br J Plast Surg 2002;55: 628–634. 112.Wilgis EF. Nerve repair and grafting. In, Green DP (ed): Operative Hand Surgery. 2nd ed, vol 2. New York: Churchill Livingstone; 1988:1373–1404. 113.Yu RS, Catalano LW III, Barron OA, Johnson C, Glickel SZ. Limited, protected postsurgical motion does not affect the results of digital nerve repair. J Hand Surg Am 2004;29: 302–306. 114.Vipond N, Taylor W, Rider M. Postoperative splinting for isolated digital nerve injuries in the hand. J Hand Ther 2007;20:222–230. 115.Malczewski MC, Zamboni WA, Haws MJ, Johnson RM, Smoot EC, Russell RC. Effect of motion on digital nerve repair in a fresh cadaver model. Plast Reconstr Surg 1995;96:1672–1675. 116.Chao RP, Braün SA, Ta KT, Palesty JA, Mine R, Syed SA, Chang DJ, Thomson JG. Early passive mobilization after digital nerve repair and grafting in a fresh cadaver. Plast Reconstr Surg 2001;108:386–391. 117.Miller EM. An experimental study to determine the strength of the suture line. Arch Surg 1921;2:167–175. 118.Cheng AS. Use of early tactile stimulation in rehabilitation of digital nerve injuries. Am J Occup Ther 2000;54:159–165. 119.Cheng AS, Hung L, Wong JM, Lau H, Chan J. A prospective study of early tactile stimulation after digital nerve repair. Clin Orthop Relat Res 2001;384:169–175. 120.Rosén B, Björkman A, Lundborg G. Improved sensory relearning after nerve repair induced by selective temporary anaesthesia: A new concept in hand rehabilitation. J Hand Surg Br 2006;31:126–132. 121.Björkman A, Weibull A, Rosén B, Svensson J, Lundborg G. Rapid cortical reorganisation and improved sensitivity of the hand following cutaneous anaesthesia of the forearm. Eur J Neurosci 2009;29:837–844. 122.Lundborg G, Björkman A, Rosén B. Enhanced sensory relearning after nerve repair by using repeated forearm anaesthesia: Aspects on time dynamics of treatment. Acta Neurochir Suppl 2007;100:121–126. 123.Björkman A, Rosén B, Lundborg G. Recovery of nerve injury-induced alexia for Braille using forearm anaesthesia. Neuroreport 2008;19:683–685. 124.Zachary RB, Holmes W. Primary suture of nerves. Surg Gynecol Obstet 1946;82:632-651. 125.MacAvoy MC, Green DP. Critical reappraisal of Medical Research Council muscle testing for elbow flexion. J Hand Surg Am 2007;32:149–153. 126.Dellon AL. The moving two-point discrimination test: Clinical evaluation of the quickly adapting fiber/receptor system. J Hand Surg Am 1978;3:474-481. 127.Dellon AL, Kallman CH. Evaluation of functional sensation in the hand. J Hand Surg Am 1983;8:865–870. 128.Strauch B, Lang A, Ferder M, Keyes-Ford M, Freeman K, Newstein D. The ten test. Plast Reconstr Surg 1997;99:1074– 1078. 129.Strauch B, Lang A. The ten test revisited. Plast Reconstr Surg 2003;112:593–594. 65 SRPS • Volume 11 • Issue R7 • 2015 130.Bell-Krotoski J, Tomancik E. The repeatability of testing with Semmes-Weinstein monofilaments. J Hand Surg Am 1987;12:155–161. 131.Moberg E. Objective methods of determining functional value of sensibility in the hand. J Bone Joint Surg Br 1958;40:454–476. 132.Moberg E. Criticism and study of methods for examining sensibility in the hand. Neurology 1962;12:8–19. 133.Jerosch-Herold C. Measuring outcome in median nerve injuries. J Hand Surg Br 1993;18:624–628. 134.Narakas AO. Brachial plexus surgery. Orthop Clin North Am 1981;12:303–323. Hand Surg Eur Vol [Epub ahead of print, July 8, 2014]. 148.Birch R. Timing of surgical reconstruction for closed traumatic injury to the supraclavicular brachial plexus. J Hand Surg Eur Vol [Epub ahead of print, July 8, 2014]. 149.Tung TH. Nerve transfers. Clin Plastic Surg 2014;41: 551–559. 150.Bentolila V, Nizard R, Bizot P, Sedel L. Complete traumatic brachial plexus palsy: Treatment and outcome after repair. J Bone Joint Surg Am 1999;81:20–28. 151.Ferenz CC. Review of the brachial plexus: Part I. Acute injuries. Orthopedics 1988;11:479–486. 135.Narakas AO. The treatment of brachial plexus injuries. Int Orthop 1985;9:29–36. 152.Ferenz CC. Review of the brachial plexus: Part II. Reconstruction after chronic upper plexus injuries. Orthopedics 1988;11:491–495. 136.Millesi H. Brachial plexus injuries: Management and results. Clin Plast Surg 1984;11:115–120. 153.Alnot JY. Infraclavicular lesions. Clin Plast Surg 1984;11:127–131. 137.Cobb TK, Carmichael SW, Cooney WP. Guyon’s canal revisited: An anatomic study of the carpal ulnar neurovascular space. J Hand Surg Am 1996;21:861–869. 154.Terzis JK, Papakonstantinou KC. The surgical treatment of brachial plexus injuries in adults. Plast Reconstr Surg 2000;106:1097–1122. 138.Burge P, Rushworth G, Watson N. Patterns of injury to the terminal branches of the brachial plexus: The place for early exploration. J Bone Joint Surg Br 1985;67:630–634. 155.Tupper JW. Discussion of “Treatment of the painful neuroma by neuroma resection and muscle implantation” by AL Dellon and SE Mackinnon. Plast Reconstr Surg 1986:77:437. 139.Giuffre JL, Kakar S, Bishop AT, Spinner RJ, Shin AY. Current concepts of the treatment of adult brachial plexus injuries. J Hand Surg Am 2010;35:678-688. Erratum in: J Hand Surg Am 2010;35:1226. 156.Tupper JW, Booth DM. Treatment of painful neuromas of sensory nerves in the hand: A comparison of traditional and newer methods. J Hand Surg Am 1976;1:144–151. 140.Moran SL, Steinmann SP, Shin AY. Adult brachial plexus injuries: Mechanism, patterns of injury, and physical diagnosis. Hand Clin 2005;21:13-24. 157.Herndon JH, Eaton RG, Littler JW. Management of painful neuromas in the hand. J Bone Joint Surg Am 1976;58:369–373. 141.Terzis JK, Vekris MD, Soucacos PN. Outcomes of brachial plexus reconstruction in 204 patients with devastating paralysis. Plast Reconstr Surg 1999;104:1221–1240. 158.Huber CG, Lewis D. Amputation neuromas. Arch Surg 1920;1:85–113. 142.Chuang DC. Adult brachial plexus reconstruction with the level of injury: Review and personal experience. Plast Reconstr Surg 2009;124[suppl 6]:e359-e369. 143.Terzis JK, Papakonstantinou KC. The surgical treatment of brachial plexus injuries in adults. Plast Reconstr Surg 2000;106:1097–1122. 144.Kline DG, Judice DJ. Operative management of selected brachial plexus lesions. J Neurosurg 1983;58:631–649. 145.Jones SJ, Parry CB, Landi A. Diagnosis of brachial plexus traction lesions by sensory nerve action potentials and somatosensory evoked potentials. Injury 1981;12:376–382. 146.Sunderland S. A classification of peripheral nerve injuries producing loss of function. Brain 1951;74:491-516. 147.Hems TE. Timing of surgical reconstruction for closed traumatic injury to the supraclavicular brachial plexus. J 66 159.Chapple WA. Prevention of nerve bulbs in stumps. Br Med J 1918;1:399. 160.Dellon AL, Mackinnon SE. Treatment of the painful neuroma by neuroma resection and muscle implantation. Plast Reconstr Surg 1986;77:427–438. 161.Swanson AB, Boeve NR, Lumsden RM. The prevention and treatment of amputation neuromata by silicone capping. J Hand Surg Am 1977;2:70–78. 162.Guttman L, Medawar PB. The chemical inhibition of fibre regeneration and neuroma formation in peripheral nerves. J Neurol Neurosurg Psychiatry 1942;5:130–141. 163.Smith RJ, Gomez NH. Local injection therapy of neuromata of the hand with triamcinolone acetonide: A preliminary study of twenty-two patients. J Bone Joint Surg Am 1970;52:71–83. 164.Martini A, Fromm B. A new operation for the prevention SRPS • Volume 11 • Issue R7 • 2015 and treatment of amputation neuromas. J Bone Joint Surg Br 1989;71:379–382. Group. Complex regional pain syndrome in adults: Concise guidance. Clin Med 2011;11:596–600. 165.Novak CB, van Vliet D, Mackinnon SE. Subjective outcome following surgical management of upper extremity neuromas. J Hand Surg Am 1995;20:221–226. 180.Goebel A, Barker CH, Turner-Stokes L. Complex Regional Pain Syndrome in Adults: UK Guidelines for Diagnosis, Referral and Management in Primary and Secondary Care. London: Royal College of Physicians; 2012. 166.Mackinnon SE, Dellon AL. Algorithm for neuroma management. Contemp Orthop 1986;13:15–27. 167.Woodhall B, Nulsen FE, White JC, Davis L. Neurosurgical implications. In, Woodhall B, Beebe GW (ed): Peripheral Nerve Regeneration. Washington DC: United States Government Print Office; 1956. 168.Watson J, Gonzalez M, Romero A, Kerna J. Neuromas of the hand and upper extremity. J Hand Surg Am 2010;35: 499–510. 169.Elliot D. Surgical management of painful peripheral nerves. Clin Plast Surg 2014;41:589–613. 170.Schutzer SF, Gossling HR. The treatment of reflex sympathetic dystrophy syndrome. J Bone Joint Surg Am 1984;66:625–629. 171.Wilder RT, Berde CB, Wolohan M, Vieyra MA, Masek BJ, Micheli LJ. Reflex sympathetic dystrophy in children: Clinical characteristics and follow-up of seventy patients. J Bone Joint Surg Am 1992;74:910–919. 172.Inhofe PD, Garcia-Moral CA. Reflex sympathetic dystrophy: A review of the literature and a long-term outcome study. Orthop Rev 1994;23:655–661. 173.Amadio PC, Mackinnon SE, Merritt WH, Brody GS, Terzis JK. Reflex sympathetic dystrophy syndrome: Consensus report of an ad hoc committee of the American Association for Hand Surgery on the definition of reflex sympathetic dystrophy syndrome. Plast Reconstr Surg 1991;87:371–375. 181.Millesi H, Meissl G, Berger A. The interfascicular nervegrafting of the median and ulnar nerves. J Bone Joint Surg Am 1972;54:727–750. 182.Millesi H. Treatment of nerve lesions by fascicular free nerve grafts. In, Michon J, Moberg E (ed): Traumatic Nerve Lesions. Edinburgh: Churchill Livingstone; 1975:91–100. 183.Millesi H, Meissl G, Berger A. Further experience with interfascicular grafting of the median, ulnar, and radial nerves. J Bone Joint Surg Am 1976;58:209–218. 184.Millesi H. Nerve grafting. Clin Plast Surg 1984;11: 105–113. 185.Millesi H. Fascicular nerve repair and interfascicular nerve grafting. In, Daniel RK, Terzis JK (ed): Reconstructive Microsurgery. Boston: Little, Brown; 1977:430–442. 186.Nunley JA, Ugino MR, Goldner RD, Regan N, Urbaniak JR. Use of the anterior branch of the medial antebrachial cutaneous nerve as a graft for the repair of defects of the digital nerve. J Bone Joint Surg Am 1989;71:563–567. 187.Moradzadeh A, Borschel GH, Luciano JP, Whitlock EL, Hayashi A, Hunter DA, Mackinnon SE. The impact of motor and sensory nerve architecture on nerve regeneration. Exp Neurol 2008:212;370–376. 188.Brushart TM, Gerber J, Kessens P, Chen YG, Royall RM. Contributions of pathway and neuron to preferential motor reinnervation. J Neurosci 1998;18:8674–8681. 174.Oyen WJ, Arntz IE, Claessens RM, Van der Meer JW, Corstens FH, Goris RJ. Reflex sympathetic dystrophy of the hand: An excessive inflammatory response? Pain 1993;55:151–157. 189.Höke A, Redett R, Hameed H, Jari R, Zhou C, Li ZB, Griffin JW, Brushart TM. Schwann cells express motor and sensory phenotypes that regulate axon regeneration. J Neurosci 2006;26:9646-9655. 175.Veldman PH, Reynen HM, Arntz IE, Goris RJ. Signs and symptoms of reflex sympathetic dystrophy: Prospective study of 829 patients. Lancet 1993;342:1012–1016. 190.Lloyd BM, Lugibuhl RD, Brenner MJ, Rocque BG, Tung TH, Myckatyn TM, Hunter DA, Mackinnon SE, Borschel GH. Use of motor nerve material in peripheral nerve repair with conduits. Microsurgery 2007;27:138-145. 176.Heerschap A, den Hollander JA, Reynen H, Goris RJ. Metabolic changes in reflex sympathetic dystrophy: A 31P NMR spectroscopy study. Muscle Nerve 1993;16:367–373. 177.Watson HK, Carlson L. Treatment of reflex sympathetic dystrophy of the hand with an active “stress loading” program. J Hand Surg Am 1987;12:779–785. 178.Albazaz R, Wong YT, Homer-Vanniasinkam S. Complex regional pain syndrome: A review. Ann Vasc Surg 2008;22:297–306. 179.Turner-Stokes L, Goebel A; Guideline Development 191.Taylor GI, Ham FJ. The free vascularized nerve graft: A further experimental and clinical application of microvascular techniques. Plast Reconstr Surg 1976;57: 413–426. 192.Bonney G, Birch R, Jamieson AM, Eames RA. Experience with vascularized nerve grafts. Clin Plast Surg 1984;11: 137–142. 193.Koshima I, Harii K. Experimental study of vascularized nerve grafts: Morphometric study of axonal regeneration 67 SRPS • Volume 11 • Issue R7 • 2015 of nerves transplanted into silicone tubes. Ann Plast Surg 1985;14:235–243. 194.Koshima I, Harii K. Experimental study of vascularized nerve grafts: Multifactorial analyses of axonal regeneration of nerves transplanted into an acute burn wound. J Hand Surg Am 1985;10:64–72. 195.Breidenbach W, Terzis JK. The anatomy of free vascularized nerve grafts. Clin Plast Surg 1984;11:65–71. 196.Townsend PL, Taylor GI. Vascularized nerve grafts using composite arterialised neuro-venous systems. Br J Plast Surg 1984;37:1–17. 197.Chiu DT, Janecka I, Krizek TJ, Wolff M, Lovelace RE. Autogenous vein graft as a conduit for nerve regeneration. Surgery 1982;91:226–233. 198.Chiu DT, Strauch B. A prospective clinical evaluation of autogenous vein grafts used as a nerve conduit for distal sensory nerve defects of 3 cm or less. Plast Reconstr Surg 1990;86:928–934. 199.Bain JR. Peripheral nerve and neuromuscular allotransplantation: Current status. Microsurgery 2000;20:384–388. 200.Wyrick JD, Stern PJ. Secondary nerve reconstruction. Hand Clin 1992;8:587–598. 201.Chuang DC, Wei FC, Noordhoff MS. Cross-chest C7 nerve grafting followed by free muscle transplantations for the treatment of total avulsed brachial plexus injuries: A preliminary report. Plast Reconstr Surg 1993;92:717–725. 202.Nagano A. Treatment of brachial plexus injury. J Orthop Sci 1998;3:71–80. 203.Tung Th, Mackinnon SE. Nerve transfers: Indications, techniques, and outcomes. J Hand Surg Am 2010;35: 332–341. 204.Nath RK, Mackinnon SE. Nerve transfers in the upper extremity. Hand Clin 2000;16:131–139. 205.Novak CB, Mackinnon SE, Tung TH. Patient outcome following a thoracodorsal to musculocutaneous nerve transfer for reconstruction of elbow flexion. Br J Plast Surg 2002;55:416–419. 206.Brandt KE, Mackinnon SE. A technique for maximizing biceps recovery in brachial plexus reconstruction. J Hand Surg Am 1993;18:726–733. 207.Suzuki K, Doi K, Hattori Y, Pagsaligan JM. Longterm results of spinal accessory nerve transfer to the suprascapular nerve in upper-type paralysis of brachial plexus injury. J Reconstr Microsurg 2007;23:295–299. 208.Colbert SH, Mackinnon S. Posterior approach for double nerve transfer for restoration of shoulder function in upper brachial plexus palsy. Hand (N Y) 2006;1:71–77. 68 209.Gu YD, Ma MK. Use of the phrenic nerve for brachial plexus reconstruction. Clin Orthop Relat Res 1996;323: 119–121. 210.Xu WD, Gu YD, Lu JB, Yu C, Zhang CG, Xu JG. Pulmonary function after complete unilateral phrenic nerve transection. J Neurosurg 2005;103:464–467. 211.Kawabata H, Shibata T, Matsui Y, Yasui N. Use of intercostal nerves for neurotization of the musculocutaneous nerve in infants with birth-related brachial plexus palsy. J Neurosurg 2001;94:386–391. 212.Oberlin C, Béal D, Leechavengvongs S, Salon A, Dauge MC, Sarcy JJ. Nerve transfer to biceps muscle using a part of ulnar nerve for C5-C6 avulsion of the brachial plexus: Anatomical study and report of four cases. J Hand Surg Am 1994;19:232–237. 213.Tung TH, Novak CB, Mackinnon SE. Nerve transfers to the biceps and brachialis branches to improve elbow flexion strength after brachial plexus injuries. J Neurosurg 2003;98:313–318. 214.Lowe JB III, Sen SK, Mackinnon SE. Current approach to radial nerve paralysis. Plast Reconstr Surg 2002;110: 1099–1113. 215.Mackinnon SE, Roque B, Tung TH. Median to radial nerve transfer for treatment of radial nerve palsy: Case report. J Neurosurg 2007;107:666–671. 216.Mackinnon SE, Novak CB, Myckatyn TM, Tung TH. Results of reinnervation of the biceps and brachialis muscles with a double fascicular transfer for elbow flexion. J Hand Surg Am 2005;30:978–985. 217.Gu YD, Zhang GM, Chen DS, Yan JG, Cheng XM, Chen L. Seventh cervical nerve root transfer from the contralateral healthy side for treatment of brachial plexus root avulsion. J Hand Surg Br 1992;17:518–521. 218.Chen L, Gu YD. An experimental study of contralateral C7 root transfer with vascularized nerve grafting to treat brachial plexus root avulsion. J Hand Surg Br 1994;19:60–66. 219.Gu YD. Distribution of the sensory endings of the C7 nerve root and its clinic significance. J Hand Surg Br 1994;19:67–68. 220.Songcharoen P, Wongtrakul S, Mahaisavariya B, Spinner RJ. Hemi-contralateral C7 transfer to median nerve in the treatment of root avulsion brachial plexus injury. J Hand Surg Am 2001;26:1058–1064. 221.Waikakul S, Orapin S, Vanadurongwan V. Clinical results of contralateral C7 root neurotization to the median nerve in brachial plexus injuries with total root avulsions. J Hand Surg Br 1999;24:556–560. 222.Terzis JK, Kokkalis ZT. Selective contralateral C7 transfer in posttraumatic brachial plexus injuries: A report of 56 SRPS • Volume 11 • Issue R7 • 2015 cases. Plast Reconstr Surg 2009;123:927–938. 223.Chen L, Gu YD, Hu SN, Xu JG, Xu L, Fu Y. Contralateral C7 transfer for the treatment of brachial plexus root avulsions in children: A report of 12 cases. J Hand Surg Am 2007;32: 96–103. 224.Beaulieu JY, Blustajn J, Teboul F, Baud P, De Schonen S, Thiebaud JB, Oberlin C. Cerebral plasticity in crossed C7 grafts of the brachial plexus: An fMRI study. Microsurgery 2006;26:303–310. 225.de Medinaceli L, Prayon M, Merle M. Percentage of nerve injuries in which primary repair can be achieved by end-to-end approximation: Review of 2,181 nerve lesions. Microsurgery 1993;14:244–246. 226.Frykman GK, Waylett J. Rehabilitation of peripheral nerve injuries. Orthop Clin North Am 1981;12:361–379. 227.Adams J, Wood VE. Tendon transfers for irreparable nerve damage in the hand. Orthop Clin North Am 1981;12:403–432. 239.Smith RJ, Hastings H II. Principles of tendon transfers to the hand. Instr Course Lect 1980;29:129–152. 240.Omer GE. Timing of tendon transfers in peripheral nerve injury. Hand Clin 1988;4:317–322. 241.Peljovich A, Ratner JA, Marino J. Update of the physiology and biomechanics of tendon transfer surgery. J Hand Surg Am 2010;35:1365-1369. 242.Brown SH, Hentzen ER, Kwan A, Ward SR, Fridén J, Lieber RL. Mechanical strength of the side-to-side versus Pulvertaft weave tendon repair. J Hand Surg Am 2010;35:540-545. 243.Grabb WC, Smith JW. Repair of peripheral nerves. In, Converse JM (ed): Reconstructive Plastic Surgery. 2nd ed, vol 6. Philadelphia: WB Saunders; 1977:3215–3227. 244.Littler JW. Neurovascular pedicle transfer of tissue in reconstructive surgery of the hand. J Bone Joint Surg Am 1956;38:917. 228.White WL. Restoration of function and balance of the wrist and hand by tendon transfers. Surg Clin North Am 1960;40:427–459. 245.Stocks GW, Cobb T, Lewis RC. Transfer of sensibility in the hand: A new method to restore sensibility in ulnar nerve palsy with use of microsurgical digital nerve translocation. J Hand Surg Am 1991;16:219–226. 229.Boyes JH. Selection of donor muscle for tendon transfer. Bull Hosp Joint Dis 1962;23:1–4. 246.Bunnell S. Opposition of the thumb. J Bone Joint Surg Am 1938;20:269–284. 230.Tubiana R. Anatomic and physiologic basis for the surgical treatment of paralyses of the hand. J Bone Joint Surg Am 1969;51:643–660. 247.Tubiana R, Valentin P. Opposition of the thumb. Surg Clin North Am 1968;48:967–977. 231.Curtis RM. Fundamental principles of tendon transfer. Orthop Clin North Am 1974;5:231–242. 232.Brand PW. Biomechanics of tendon transfer. Hand Clin 1988;4:137–154. 233.Burkhalter WE. Early tendon transfer in upper extremity peripheral nerve injury. Clin Orthop Relat Res 1974;104: 68–79. 234.Goldner JL. Tendon transfer for irreparable peripheral nerve injuries of the upper extremity. Orthop Clin North Am 1974;5:343–375. 235.Riordan DC. Rehabilitation and re-education in tendon transfers. Orthop Clin North Am 1974;5:445–449. 236.Smith RJ. Balance and kinetics of the fingers under normal and pathological conditions. Clin Orthop Relat Res 1974;104:92–111. 237.Leffert RD. Patterns of neuromuscular activity following tendon transfer in the upper limb: A preliminary study. J Hand Surg Am 1976;1:181–189. 238.Freehafer AA, Peckham PH, Keith MW. Determination of muscle-tendon unit properties during tendon transfer. J Hand Surg Am 1979;4:331–339. 248.Royle ND. An operation for paralysis of the intrinsic muscles of the thumb. JAMA 1938;111:612–613. 249.Thompson TC. A modified operation for opponens paralysis. J Bone Joint Surg Am 1942;24:632–640. 250.Littler JW. Tendon transfers and arthrodesis in combined median and ulnar nerve paralysis. J Bone Joint Surg Am 1949;31:225–234. 251.Riordan DC. Tendon transfers for nerve paralysis of the hand and wrist. Curr Pract Orthop Surg 1964;23:17–40. 252.Brand PW. The hand in leprosy. In, Pulvertaft RG (ed): Clinical Surgery: The Hand. London: Butterworths; 1966: 279–295. 253.Camitz H. Uber die behandlung der oppositionslahmung. Acta Chir Scand 1929;65:77–81. 254.Littler JW, Li CS. Primary restoration of thumb opposition with median nerve decompression. Plast Reconstr Surg 1967;39:74–75. 255.Braun RM. Palmaris longus tendon transfer for augmentation of thenar musculature in low median nerve palsy. J Hand Surg Am 1978;3:488–491. 256.Terrono AL, Rose JH, Mulroy J, Millender LH. Camitz palmaris longus abductorplasty for severe thenar atrophy 69 SRPS • Volume 11 • Issue R7 • 2015 secondary to carpal tunnel syndrome. J Hand Surg Am 1993;18:204–206. 257.Rowntree T. Anomalous innervation of the hand muscles. J Bone Joint Surg Br 1949;31:505-510. 258.Doi K, Sakai K, Ihara K, Abe Y, Kawai S, Kurafuji Y. Reinnervated free muscle transplantation for extremity reconstruction. Plast Reconstr Surg 1993;91:872–883. 259.Burkhalter WE. Median nerve palsy. In, Green DP (ed): Operative Hand Surgery. 2nd ed. vol 2. New York: Churchill Livingstone; 1988:1029–1059. 260.Ustün ME, Oğün TC, Karabulut AK, Büyükmumcu M. An alternative method for restoring opposition after median nerve injury: An anatomical feasibility study for the use of neurotisation. J Anat 2001;198:635–638. 261.Vernadakis AJ, Humphreys DB, Mackinnon SE. Distal anterior interosseous nerve in the recurrent motor branch graft for reconstruction of a median nerve neuroma-incontinuity. J Reconstr Microsurg 2004;20:7–11. 273.Zancolli EA. Claw-hand caused by paralysis of the intrinsic muscles: A simple surgical procedure for its correction. J Bone Joint Surg Am 1957;39:1076–1080. 274.Brown PW. Zancolli capsulorrhaphy for ulnar claw hand. J Bone Joint Surg Am 1970;52:868–877. 275.Leddy JP, Stark HH, Ashworth CR, Boyes JH. Capsulodesis and pulley advancement for the correction of claw-finger deformity. J Bone Joint Surg Am 1972;54: 1465–1471. 276.Bunnell S. Surgery of the intrinsic muscles of the hand other than those producing opposition of the thumb. J Bone Joint Surg Am 1942;24:1–31. 277.Parkes A. Paralytic claw fingers: A graft tenodesis operation. Hand 1973;5:192–199. 278.Riordan DC. Tendon transplantations in median-nerve and ulnar-nerve paralysis. J Bone Joint Surg Am 1953;35: 312–320. 262.Burkhalter WE, Christensen RC, Brown PW. Extensor indicis proprius opponensplasty. J Bone Joint Surg Am 1973;55:725–732. 279.Riordan DC. Surgery of the paralytic hand. In, Reynolds FC (ed): American Academy of Orthopaedic Surgeons Instructional Course Lectures. vol 16. St Louis: CV Mosby; 1959:79–90. 263.Schneider LH. Opponensplasty using the extensor digiti minimi. J Bone Joint Surg Am 1969;51:1297–1302. 280.Fowler SB. Extensor apparatus of the digits. J Bone Joint Surg Br 1949;31:477. 264.Henderson ED. Transfer of wrist extensors and brachioradialis to restore opposition of the thumb. J Bone Joint Surg Am 1962;44:513–522. 281.Brown PW. Reconstruction for pinch in ulnar intrinsic palsy. Orthop Clin North Am 1974;5:323–341. 265.Huber E. Hilfsoperation bei median Uslahmung. Dtsch Arch Klin Med 1921;136:271–275. 266.Littler JW, Cooley SG. Opposition of the thumb and its restoration by abductor digiti quinti transfer. J Bone Joint Surg Am 1963;45:1389–1396. 282.Boyes JH. Principles of tendon transfer in the upper extremity: The Sir Robert Jones Lecture 1977 of the Hospital for Joint Diseases. Bull Hosp Joint Dis 1978;39:1–11. 283.Brand PW. Tendon transfers for median and ulnar nerve paralysis. Orthop Clin North Am 1970;1:447–454. 267.Riley WB Jr, Mann RJ, Burkhalter WE. Extensor pollicis longus opponensplasty. J Hand Surg Am 1980;5:217–220. 284.Brand PW. Paralytic claw hand; with special reference to paralysis in leprosy and treatment by the sublimis transfer of Stiles and Bunnell. J Bone Joint Surg Br 1958;40B:618–632. 268.Kessler I. Transfer of extensor carpi ulnaris to tendon of extensor pollicis brevis for opponensplasty. J Bone Joint Surg Am 1969;51:1303–1308. 285.Burkhalter WE, Strait JL. Metacarpophalangeal flexor replacement for intrinsic-muscle paralysis. J Bone Joint Surg Am 1973;55:1667–1676. 269.Koh S, Buford WL II, Andersen CR, Viegas SF. Intrinsic muscle contribution to the metacarpophalangeal joint flexion moment of the middle, ring, and small fingers. J Hand Surg Am 2006;31:1111–1117. 286.Smith RJ. Extensor carpi radialis brevis tendon transfer for thumb adduction: A study of power pinch. J Hand Surg Am 1983;8:4–15. 270.Hastings H II, Davidson S. Tendon transfers for ulnar nerve palsy: Evaluation of results and practical treatment considerations. Hand Clin 1988;4:167–178. 287.Fritschi EP. Reconstructive Surgery in Leprosy. Bristol: John Wright & Sons; 1971:64. 271.Mikhail IK. Bone block operation for clawhand. Surg Gynecol Obstet 1964;118:1077–1079. 288.Littler JW. Restoration of power and stability in the partially paralyzed hand. In, Converse JM (ed): Reconstructive Plastic Surgery. 2nd ed, vol 6. Philadelphia: WB Saunders; 1977:3266–3280. 272.Bunnell S. Surgery of the Hand. Philadelphia: JB Lippincott; 1944. 289.Scuderi C. Tendon transplants for irreparable radial nerve paralysis. Surg Gynecol Obstet 1949;88:643–651. 70 SRPS • Volume 11 • Issue R7 • 2015 290.Zachary RB. Tendon transplantation for radial paralysis. Br J Surg 1946;23:350–364. upper extremity of patients with cerebral palsy. J Hand Surg Am 1979;4:424–431. 291.Boyes JH. Tendon transfers for radial palsy. Bull Hosp Joint Dis 1960;21:97–105. 307.House JH, Gwathmey FW, Lundsgaard DK. Restoration of strong grasp and lateral pinch in tetraplegia due to cervical spinal cord injury. J Hand Surg Am 1976;1:152–159. 292.Chuinard RG, Boyes JH, Stark HH, Ashworth CR. Tendon transfers for radial nerve palsy: Use of superficialis tendons for digital extension. J Hand Surg Am 1978;3:560–570. 293.Brand PW. Tendon transfers in the forearm. In, Flynn JE (ed): Hand Surgery. Baltimore: Williams & Wilkins; 1966: 331–342. 294.Green DP. Radial nerve palsy. In, Green DP (ed): Operative Hand Surgery. 3rd ed, vol 2. New York: Churchill Livingstone; 1993:1401–1417. 295.Beasley RW. Tendon transfers for radial nerve palsy. Orthop Clin North Am 1970;1:439–445. 296.Lowe JB III, Tung TR, Mackinnon SE. New surgical option for radial nerve paralysis. Plast Reconstr Surg 2002;110: 836–843. 308.Lamb DW, Landry RM. The hand in quadriplegia. Paraplegia 1972;9:204-212. 309.Lipscomb PR, Elkins EC, Henderson ED. Tendon transfers to restore function of hands in tetraplegia, especially after fracture dislocation of the sixth cervical vertebra on the seventh. J Bone Joint Surg Am 1958;40A:1071-1080. 310.Moberg E. Surgical treatment for absent single-hand grip and elbow extension in quadriplegia: Principles amd preliminary experience. J Bone Joint Surg Am 1975;57: 196–206. 311.Moberg EA, Lamb DW. Surgical rehabilitation of the upper limb in tetraplegia. Hand 1980;12:209–213. 297.Freehafer AA. Tendon transfers in tetraplegic patients: The Cleveland experience. Spinal Cord 1998;36:315–319. 312.Zancolli E. Surgery for the quadriplegic hand with active, strong wrist extension preserved: A study of 97 cases. Clin Orthop Relat Res 1975;112:101–113. 298.Freehafer AA. Gaining independence in tetraplegia: Cleveland technique. Clin Orthop Relat Res 1998;355:282– 289. 313.Toth S. Therapist’s management of tendon transfers. Hand Clin 1986;2:239–246. 299.Hamlin C. Upper extremity reconstruction in the tetraplegic patient. Tech Hand Up Extrem Surg 2001;5: 91–104. 300.Apfelberg DB, Maser MR, Lash H, Keoshian L. “I-P flexor lag” after thumb flexor reconstruction: Causes and solution. Hand 1980;12:167–172. 301.Bryan RS. The Moberg deltoid-triceps replacement and key-pinch operations in quadriplegia: Preliminary experiences. Hand 1977;9:207–214. 302.Colyer RA, Kappelman B. Flexor pollicis longus tenodesis in tetraplegia at the sixth cervical level: A prospective evaluation of functional gain. J Bone Joint Surg Am 1981;63:376–379. 303.Curtis RM. Tendon transfers in the patient with spinal cord injury. Orthop Clin North Am 1974;5:415–423. 304.Freehafer AA, William AM. Transfer of the brachioradialis to improve wrist extension in high spinal cord injury. J Bone Joint Surg Am 1967;49:648–652. 305.Hentz VR, Brown M, Keoshian LA. Upper limb reconstruction in quadriplegia: Functional assessment and proposed treatment modifications. J Hand Surg Am 1983;8:119–131. 306.Hoffer MM, Perry J, Melkonian GJ. Dynamic electromyography and decision making for surgery in the 314.Sultana SS, MacDermid JC, Grewal R, Rath S. The effectiveness of early mobilization after tendon transfers in the hand: A systematic review. J Hand Ther 2013;26:1-20. 315.Chuang DC. Brachial plexus injury: Nerve reconstruction and functioning muscle transplantation. Semin Plast Surg 2010;24:57–66. 316.Chuang DC. Neurotization and free muscle transfer for brachial plexus avulsion injury. Hand Clin 2007;23:91–104. 317.Doi K, Muramatsu K, Hattori Y, Otsuka K, Tan SH, Nanda V, Watanabe M. Restoration of prehension with the double free muscle technique following complete avulsion of the brachial plexus: Indications and long-term results. J Bone Joint Surg Am 2000;82:652–666. 318.Doi K, Hattori Y, Kuwata N, Soo-heong T, Kawakami F, Otsuka K, Watanabe M. Free muscle transfer can restore hand function after injuries of the lower brachial plexus. J Bone Joint Surg Br 1998;80:117-120. 319.Dellon AL. Patient evaluation and management considerations in nerve compression. Hand Clin 1992;8: 229–239. 320.Bianchi S, Martinoli C, Abdelwahab IF. High-frequency ultrasound examination of the wrist and hand. Skeletal Radiol 1999;28:121–129. 321.Mallouhi A, Pülzl P, Trieb T, Piza H, Bodner G. Predictors of carpal tunnel syndrome: Accuracy of gray-scale and color 71 SRPS • Volume 11 • Issue R7 • 2015 Doppler sonography. AJR Am J Roentgenol 2006;186: 1240–1245. 322.Beekman R, Visser LH. Sonography in the diagnosis of carpal tunnel syndrome: A critical review of the literature. Muscle Nerve 2003;27:26–33. 323.Bayrak IK, Bayrak AO, Tilki HE, Nural MS, Sunter T. Ultrasonography in carpal tunnel syndrome: Comparison with electrophysiological stage and motor unit estimate. Muscle Nerve 2007;35:344–348. 324.Kele H, Verheggen R, Bittermann HJ, Reimers CD. The potential value of ultrasonography in the evaluation of carpal tunnel syndrome. Neurology 2003;61:389–391. 325.Bianchi S, Montet X, Martinoli C, Bonvin F, Fasel J. Highresolution sonography of compressive neuropathies of the wrist. J Clin Ultrasound 2004;32:451–461. 326.Wiesler ER, Chloros GD, Cartwright MS, Smith BP, Rushing J, Walker FO. The use of diagnostic ultrasound in carpal tunnel syndrome. J Hand Surg Am 2006;31:726–732. 327.Yassi A. Repetitive strain injuries. Lancet 1997;349: 943–947. 328.Strömberg T, Dahlin LB, Lundborg G. Vibrotactile sense in the hand-arm vibration syndrome. Scand J Work Environ Health 1998;24:495–502. 329.Giannini F, Rossi S, Passero S, Bovenzi M, Cannavà G, Mancini R, Cioni R, Battistini N. Multifocal neural conduction impairment in forestry workers exposed and not exposed to vibration. Clin Neurophysiol 1999;110:1276–1283. 330.Cederlund R, Nordenskiöld U, Lundborg G. Handarm vibration exposure influences performance of daily activities. Disabil Rehabil 2001;23:570–577. 331.Spinner M, Spencer PS. Nerve compression lesions of the upper extremity: A clinical and experimental review. Clin Orthop Relat Res 1974;104:46–67. 332.Rempel D, Dahlin L, Lundborg G. Pathophysiology of nerve compression syndromes: Response of peripheral nerves to loading. J Bone Joint Surg Am 1999;81:1600–1610. 333.Eversmann WW Jr. Entrapment and compression neuropathies. In, Green DP (ed): Operative Hand Surgery. New York: Churchill Livingstone; 1982:957–1009. 334.Eversmann WW Jr. Compression and entrapment neuropathies of the upper extremity. J Hand Surg Am 1983;8:759–766. 335.Dawson DM. Entrapment neuropathies of the upper extremities. N Engl J Med 1993;329:2013–2018. 336.Mackinnon SE, Novak CB. Clinical commentary: Pathogenesis of cumulative trauma disorder. J Hand Surg Am 1994;19:873–883. 72 337.Osorio AM, Ames RG, Jones J, Castorina J, Rempel D, Estrin W, Thompson D. Carpal tunnel syndrome among grocery store workers. Am J Ind Med 1994;25:229–245. 338.Collins K, Storey M, Peterson K, Nutter P. Nerve injuries in athletes. Phys Sports Med 1988;16:92–96. 339.Eversmann WW Jr. Entrapment and compression neuropathies. In, Green DP (ed): Operative Hand Surgery. 3rd ed, vol 2. New York: Churchill Livingstone; 1993:1341–1385. 340.Lorei MP, Hershman EB. Peripheral nerve injuries in athletes: Treatment and prevention. Sports Med 1993;16:130–147. 341.Olave E, Del Sol M, Gabrielli C, Mandiola E, Rodrigues CF. Biometric study of the relationships between palmar neurovascular structures, the flexor retinaculum and the distal wrist crease. J Anat 2001;198:737–741. 342.Lanz U. Anatomical variations of the median nerve in the carpal tunnel. J Hand Surg Am 1977;2:44–53. 343.Amadio PC. Anatomic variations of the median nerve within the carpal tunnel. Clin Anat 1988;1:23–31. 344.Szabo RM, Pettey J. Bilateral median nerve bifurcation with an accessory compartment within the carpal tunnel. J Hand Surg Br 1994;19:22–23. 345.Green DP, Morgan JP. Correlation between muscle morphology of the transverse carpal ligament and branching pattern of the motor branch of median nerve. J Hand Surg Am 2008;33:1505–1511. 346.Kaplan EB. Functional and Surgical Anatomy of the Hand. 2nd ed. Philadelphia: JB Lippincott; 1965:265–270. 347.Eskandari MM, Yilmaz C, Oztuna V, Kuyurtar F. Topographic localization of the motor branch of the median nerve. J Hand Surg Am 2005;30:803–807. 348.Vella JC, Hartigan BJ, Stern PJ. Kaplan’s cardinal line. J Hand Surg Am 2006;31:912–918. 349.Sugimoto H, Miyaji N, Ohsawa T. Carpal tunnel syndrome: Evaluation of median nerve circulation with dynamic contrast-enhanced MR imaging. Radiology 1994;190:459–466. 350.Eriksen J. A case of carpal tunnel syndrome on the basis of an abnormally long lumbrical muscle. Acta Orthop Scand 1973;44:275–277. 351.Jabaley ME. Personal observations on the role of the lumbrical muscles in carpal tunnel syndrome. J Hand Surg Am 1978;3:82–84. 352.Desjacques P, Egloff-Baer S, Roth G. Lumbrical muscles and the carpal tunnel syndrome. Electromyogr Clin Neurophysiol 1980;20:443–449. 353.Cobb TK, An KN, Cooney WP. Effect of lumbrical muscle SRPS • Volume 11 • Issue R7 • 2015 incursion within the carpal tunnel on carpal tunnel pressure: A cadaveric study. J Hand Surg Am 1995;20:186–192. 655–661. 354.Braun RM, Davidson K, Doehr S. Provocative testing in the diagnosis of dynamic carpal tunnel syndrome. J Hand Surg Am 1989;14:195–197. 368.Richman J, Gelberman R, Rydevik BL, Hajek PC, Braun RM, Gylys-Morin VM, Berthoty D. Carpal tunnel syndrome: Morphologic changes after release of the transverse carpal ligament. J Hand Surg Am 1989;14:852–857. 355.Osamura N, Zhao C, Zobitz ME, An KN, Amadio PC. Evaluation of the material properties of the subsynovial connective tissue in carpal tunnel syndrome. Clin Biomech (Bristol, Avon) 2007;22:999–1003. 369.Silver MA, Gelberman RH, Gellman H, Rhoades CE. Carpal tunnel syndrome: Associated abnormalities in ulnar nerve function and the effect of carpal tunnel release on these abnormalities. J Hand Surg Am 1985;10:710–713. 356.Filius A, Thoreson AR, Yang TH, Vanhees M, An KN, Zhao C, Amadio PC. The effect of low- and high-velocity tendon excursion. J Orthop Res 2014;32:123-128. 370.Singh I, Khoo KM, Krishnamoorthy S. The carpal tunnel syndrome: Clinical evaluation and results of surgical decompression. Ann Acad Med Singapore 1994;23:94–97. 357.Yoshii Y, Zhao C, Henderson J, Zhao KD, An KN, Amadio PC. Shear strain and motion of the subsynovial connective tissue and median nerve during single-digit motion. J Hand Surg Am 2009;34:65–73. 371.Lindau T, Karlsson MK. Complications and outcome in open carpal tunnel release: A 6-year follow-up in 92 patients. Chir Main 1999;18:115–121. 358.Ariyan S, Watson HK. The palmar approach for the visualization and release of the carpal tunnel: An analysis of 429 cases. Plast Reconstr Surg 1977;60:539–547. 359.Heckler FR, Jabaley ME. Evolving concepts of median nerve decompression in the carpal tunnel. Hand Clin 1986;2:723–736. 360.Jakab E, Ganos D, Cook FW. Transverse carpal ligament reconstruction in surgery for carpal tunnel syndrome: A new technique. J Hand Surg Am 1991;16:202–206. 372.Kuschner SH, Brien WW, Johnson D, Gellman H. Complications associated with carpal tunnel release. Orthop Rev 1991;20:346–352. 373.Shurr DG, Blair WF, Bassett G. Electromyographic changes after carpal tunnel release. J Hand Surg Am 1986;11:876–880. 374.Goodwill CJ. The carpal tunnel syndrome: Long-term follow-up showing relation of latency measurements to response to treatment. Ann Phys Med 1965;8:12–21. 361.Serra JM, Benito JR, Monner J. Carpal tunnel release with short incision. Plast Reconstr Surg 1997;99:129–135. 375.Thomas JE, Lambert EH, Cseuz KA. Electrodiagnostic aspects of the carpal tunnel syndrome. Arch Neurol 1967;16:635–641. 362.Lee WP, Strickland JW. Safe carpal tunnel release via a limited palmar incision. Plast Reconstr Surg 1998;101: 418–424. 376.Wintman BI, Winters SC, Gelberman RH, Katz JN. Carpal tunnel release: Correlations with preoperative symptomatology. Clin Orthop Relat Res 1996;326:135–145. 363.Lee WP, Schipper BM, Goitz RJ. 13-year experience of carpal tunnel release using the Indiana Tome technique. J Hand Surg Am 2008;33:1052–1056. 377.Cartotto RC, McCabe S, Mackinnon SE. Two devastating complications of carpal tunnel surgery. Ann Plast Surg 1992;28:472–474. 364.Lowry WE Jr, Follender AB. Interfascicular neurolysis in the severe carpal tunnel syndrome: A prospective, randomized, double-blind, controlled study. Clin Orthop Relat Res 1988;227:251–254. 378.Okutsu I, Ninomiya S, Ugawa Y, Kuroshima N, Inanami H, Hiraki S. New operative procedure for carpal tunnel syndrome: Endoscopic operation and clinical results. J Jap Soc Surg Hand 1987;4:117–120. 365.Seiler JG III, Milek MA, Carpenter GK, Swiontkowski MF. Intraoperative assessment of median nerve blood flow during carpal tunnel release with laser Doppler flowmetry. J Hand Surg Am 1989;14:986–991. 379.Chow JC. Endoscopic release of the carpal ligament: A new technique for carpal tunnel syndrome. Arthroscopy 1989;5:19–24. 366.Mackinnon SE, McCabe S, Murray JF, Szalai JP, Kelly L, Novak C, Kin B, Burke GM. Internal neurolysis fails to improve the results of primary carpal tunnel decompression. J Hand Surg Am 1991;16:211–218. 380.Nagle DJ, Fischer T, Harris GD, Hastings H II, Osterman AL, Palmer AK, Viegas SF, Whipple TL, Foley M. A multicenter prospective review of 640 endoscopic carpal tunnel releases using the transbursal and extrabursal chow techniques. Arthroscopy 1996;12:139−143. 367.Blair WF, Goetz DD, Ross MA, Steyers CM, Chang P. Carpal tunnel release with and without epineurotomy: A comparative prospective trial. J Hand Surg Am 1996;21: 381.Agee JM, McCarroll HR Jr, Tortosa RD, Berry DA, Szabo RM, Peimer CA. Endoscopic release of the carpal tunnel: A randomized prospective multicenter study. J Hand Surg Am 73 SRPS • Volume 11 • Issue R7 • 2015 992;17:987–995. 2003;28:250-254. 382.Nagle DJ. Endoscopic carpal tunnel release: In favor. Clin Plast Surg 1996;23:477–486. 396.Lamberti PM, Light TR. Carpal tunnel syndrome in children. Hand Clin 2002;18:331–337. 383.Palmer AK, Toivonen DA. Complications of endoscopic and open carpal tunnel release. J Hand Surg Am 1999;24:561–565. 397.Choudry UH, Moran SL, Cooney WP. Pediatric carpal tunnel syndrome: A 30-year analysis and treatment algorithm [abstract]. Presented at the 24th Annual Adrian E. Flatt Residents and Fellow Conference in Hand Surgery, American Society for Surgery of the Hand, September 6, 2006. 384.Brown RA, Gelberman RH, Seiler JG III, Abrahamsson SO, Weiland AJ, Urbaniak JR, Schoenfeld DA, Furcolo D. Carpal tunnel release: A prospective, randomized assessment of open and endoscopic methods. J Bone Joint Surg Am 1993;75:1265–1275. 385.Murphy RX Jr, Jennings JF, Wukich DK. Major neurovascular complications of endoscopic carpal tunnel release. J Hand Surg Am 1994;19:114–118. 386.Feinstein PA. Endoscopic carpal tunnel release in a community-based series. J Hand Surg Am 1993;18:451–454. 387.Roth JH, Richards RS, MacLeod MD. Endoscopic carpal tunnel release. Can J Surg 1994;37:189–193. 388.Palmer DH, Paulson JC, Lane-Larsen CL, Peulen VK, Olson JD. Endoscopic carpal tunnel release: A comparison of two techniques with open release. Arthroscopy 1993;9:498508. 389.Jimenez DF, Gibbs SR, Clapper AT. Endoscopic treatment of carpal tunnel syndrome: A critical review. J Neurosurg 1998;88:817–826. 390.Thoma A, Wong VH, Sprague S, Duku E. A cost-utility analysis of open and endoscopic carpal tunnel release. Can J Plast Surg 2006;14:15–20. 391.Thoma A, Veltri K, Haines T, Duku E. A meta-analysis of randomized controlled trials comparing endoscopic and open carpal tunnel decompression. Plast Reconstr Surg 2004;114:1137–1146. 392.Atroshi I, Hofer M, Larsson GU, Ornstein E, Johnsson R, Ranstam J. Open compared with 2-portal endoscopic carpal tunnel release: A 5-year follow-up of a randomized controlled trial. J Hand Surg Am 2009;34:266-272. 393.Chatterjee JS, Price PE. Comparative responsiveness of the Michigan Hand Outcomes Questionnaire and the Carpal Tunnel Questionnaire after carpal tunnel release. J Hand Surg Am 2009;34:273-280. 394.Kotsis SV, Chung KC. Responsiveness of the Michigan Hand Outcomes Questionnaire and the Disabilities of the Arm, Shoulder and Hand Questionnaire in carpal tunnel surgery. J Hand Surg Am 2005;30:81-86. 395.Gay RE, Amadio PC, Johnson JC. Comparative responsiveness of the Disabilities of the Arm, Shoulder and Hand, the Carpal Tunnel Questionnaire, and the SF-36 to clinical change after carpal tunnel release. J Hand Surg Am 74 398.Hunter JM. Recurrent carpal tunnel syndrome, epineural fibrous fixation, and traction neuropathy. Hand Clin 1991;7:491–504. 399.O’Malley MJ, Evanoff M, Terrono AL, Millender LH. Factors that determine reexploration treatment of carpal tunnel syndrome. J Hand Surg Am 1992;17:638–641. 400.Rose EH, Norris MS, Kowalski TA, Lucas A, Flegler EJ. Palmaris brevis turnover flap as an adjunct to internal neurolysis of the chronically scarred median nerve in recurrent carpal tunnel syndrome. J Hand Surg Am 1991;16:191–201. 401.Botte MJ, von Schroeder HP, Abrams RA, Gellman H. Recurrent carpal tunnel syndrome. Hand Clin 1996;12: 731–743. 402.Howard FM. Compression neuropathies in the anterior forearm. Hand Clin 1986;2:737–745. 403.Olehnik WK, Manske PR, Szerzinski J. Median nerve compression in the proximal forearm. J Hand Surg Am 1994;19:121–126. 404.Ritts GD, Wood MB, Linscheid RL. Radial tunnel syndrome: A ten-year surgical experience. Clin Orthop Relat Res 1987;219:201–205. 405.Scher DL, Wolf JM, Owens BD. Lateral epicondylitis. Orthopedics 2009;32:pii. 406.Jebson PJ, Engber WD. Radial tunnel syndrome: Longterm results of surgical decompression. J Hand Surg Am 1997;22:889–896. 407.Roles NC, Maudsley RH. Radial tunnel syndrome: Resistant tennis elbow as a nerve entrapment. J Bone Joint Surg Br 1972;54:499–508. 408.Lawrence T, Mobbs P, Fortems Y, Stanley JK. Radial tunnel syndrome: A retrospective review of 30 decompressions of the radial nerve. J Hand Surg Br 1995;20:454–459. 409.Kotani H, Miki T, Senzoku F, Nakagawa Y, Ueo T. Posterior interosseous nerve paralysis with multiple constrictions. J Hand Surg Am 1995;20:15–17. 410.Leffert RD. Anterior submuscular transposition of the ulnar nerves by the Learmonth technique. J Hand Surg Am SRPS • Volume 11 • Issue R7 • 2015 1982;7:147–155. 411.Amadio PC, Beckenbaugh RD. Entrapment of the ulnar nerve by the deep flexor-pronator aponeurosis. J Hand Surg Am 1986;11:83–87. 412.Folberg CR, Weiss AP, Akelman E. Cubital tunnel syndrome: Part I. Presentation and diagnosis. Orthop Rev 1994;23:136–144. 413.Folberg CR, Weiss AP, Akelman E. Cubital tunnel syndrome: Part II. Treatment. Orthop Rev 1994;23:233–241. 414.Learmonth JR. A technique for transplanting the ulnar nerve. Surg Gynecol Obstet 1942;75:792–793. 415.Novak CB, Mackinnon SE. Selection of operative procedures for cubital tunnel syndrome. Hand (N Y) 2009;4:50–54. 416.Bartels RH, Verhagen WI, van der Wilt GJ, Meulstee J, van Rossum LG, Grotenhuis JA. Prospective randomized controlled study comparing simple decompression versus anterior subcutaneous transposition for idiopathic neuropathy of the ulnar nerve at the elbow: Part 1. Neurosurgery 2005;56:522–530. 417.Biggs M, Curtis JA. Randomized, prospective study comparing ulnar neurolysis in situ with submuscular transposition. Neurosurgery 2006;58:296–304. 418.Gervasio O, Gambardella G, Zaccone C, Branca D. Simple decompression versus anterior submuscular transposition of the ulnar nerve in severe cubital tunnel syndrome: A prospective randomized study. Neurosurgery 2005;56:108–117. 419.Nabhan A, Ahlhelm F, Kelm J, Reith W, Schwerdtfeger K, Steudel WI. Simple decompression or subcutaneous anterior transposition of the ulnar nerve for cubital tunnel syndrome. J Hand Surg Br 2005;30:521–524. 420.Bartels RH, Termeer EH, van der Wilt GJ, van Rossum LG, Meulstee J, Verhagen WI, Grotenhuis JA. Simple decompression or anterior subcutaneous transposition for ulnar neuropathy at the elbow: A cost-minimization analysis: Part 2. Neurosurgery 2005;56:531–536. 421.Mowlavi A, Andrews K, Lille S, Verhulst S, Zook EG, Milner S. The management of cubital tunnel syndrome: A meta-analysis of clinical studies. Plast Reconstr Surg 2000;106:327–334. 422.Lowe JB III, Mackinnon SE. Management of secondary cubital tunnel syndrome. Plast Reconstr Surg 2004;113: E1–E16. 423.Race CM, Saldana MJ. Anatomic course of the medial cutaneous nerves of the arm. J Hand Surg Am 1991;16:48–52. 424.Lowe JB III, Maggi SP, Mackinnon SE. The position of crossing branches of the medial antebrachial cutaneous nerve during cubital tunnel surgery in humans. Plast Reconstr Surg 2004;114:692–696. 425.Moneim MS. Ulnar nerve compression at the wrist: Ulnar tunnel syndrome. Hand Clin 1992;8:337–344. 426.König PS, Hage JJ, Bloem JJ, Prosé LP. Variations of the ulnar nerve and ulnar artery in Guyon’s canal: A cadaveric study. J Hand Surg Am 1994;19:617–622. 427.Wilgis EF, Murphy R. The significance of longitudinal excursion in peripheral nerves. Hand Clin 1986;2:761–766. 428.Jones NF, Shaw WW, Katz RG, Angeles L. Circumferential wrapping of a flap around a scarred peripheral nerve for salvage of end-stage traction neuritis. J Hand Surg Am 1997;22:527–535. 429.Chou KH, Papadimitriou NG, Sarris I, Sotereanos DG. Neovascularization and other histopathologic findings in an autogenous saphenous vein wrap used for recalcitrant carpal tunnel syndrome: A case report. J Hand Surg Am 2003;28:262–266. 430.Magill CK, Tuffaha SH, Yee A, Luciano JP, Hunter DA, Mackinnon SE, Borschel GH. The short- and long-term effects of Seprafilm on peripheral nerves: A histological and functional study. J Reconstr Microsurg 2009;25:345–354. 431.Simpson RL, Fern SA. Multiple compression neuropathies and the double-crush syndrome. Orthop Clin North Am 1996;27:381–388. 432.Upton AR, McComas AJ. The double crush in nerve entrapment syndromes. Lancet 1973;2:359–362. 433.Mackinnon SE. Double and multiple “crush” syndromes: Double and multiple entrapment neuropathies. Hand Clin 1992;8:369-390. 75 We thank the Aesthetic Surgery Journal and Plastic and Reconstructive Surgery for their support. Now Indexed in MEDLINE! Submit Your Manuscript Today Aesthetic Surgery Journal (ASJ) is a peer-reviewed international journal focusing on scientific developments and clinical techniques in aesthetic surgery. An official publication of the 2200-member American Society for Aesthetic Plastic Surgery (ASAPS), ASJ is also the official English-language journal of twelve major international societies of plastic, aesthetic, and reconstructive surgery representing South America, Central America, Europe, Asia, and the Middle East, and the official journal of The Rhinoplasty Society. Submit your manuscript! www.aestheticsurgeryjournal.com Order Today: (800) 818-7243 (US & Canada) (805) 499 9774 (Other countries) www.aestheticsurgeryjournal.com Published by This is your life. Whether you’re performing a routine cosmetic procedure or a difficult burn repair, having the right tool for unexpected complications is critical. This is your website. The new Plastic and Reconstructive Surgery Online The intuitive, all-new, next-generation electronic journals platform from Lippincott Williams & Wilkins comes loaded with personalized options for its journal subscribers, including customizable state-of-the-art capabilities, self-service options tailored to your information preferences, rich media content, improved search options, better article readability in HTML and better tools to help you manage content that’s vital to your practice and patient care. Register online at prsjournal.com Not a subscriber? Visit prsjournal.com to subscribe and find out more