* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Simulation modeling as an exotic livestock disease disaster
Sexually transmitted infection wikipedia , lookup
Hepatitis B wikipedia , lookup
Chagas disease wikipedia , lookup
Tuberculosis wikipedia , lookup
Middle East respiratory syndrome wikipedia , lookup
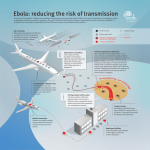
Ebola virus disease wikipedia , lookup
Oesophagostomum wikipedia , lookup
Hepatitis C wikipedia , lookup
Sarcocystis wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Bioterrorism wikipedia , lookup
Bovine spongiform encephalopathy wikipedia , lookup
Coccidioidomycosis wikipedia , lookup
Onchocerciasis wikipedia , lookup
Marburg virus disease wikipedia , lookup
Schistosomiasis wikipedia , lookup
Brucellosis wikipedia , lookup
Leishmaniasis wikipedia , lookup
Trichinosis wikipedia , lookup
African trypanosomiasis wikipedia , lookup
Fasciolosis wikipedia , lookup
[Session: Modeling and Risk Assessment] Simulation Modeling as an Exotic Livestock Disease Disaster Planning Tool DR. TIM CARPENTER UNIV. OF CALIFORNIA-DAVIS M ichael gave you the introduction so I won’t spend any time on the slide other than to say we’re at a Center for Modeling and much of the funding that we’re getting for this is coming from DHS, modeling Foot and Mouth Disease. In the presentation I’d like to do 2 things; one convince you – first of all, let me ask you, Michael gave an introduction for me, how many of you are modelers? How about food technologists? What else do we have; Colonels, at least 2 Colonels; thank you. So I’ll quickly go over some of the reasons why we as modelers do what we do. Secondly, what I want to do is illustrate this with examples of modeling disease disaster planning, some of the things that we have done and what we plan to do in the near future. So, goals of epidemic simulation modeling – why do we model; well obviously, to predict disease incidence to give us an idea of what is coming, what to anticipate, secondly, to predict spatial/category disease spread or spatial and category disease spread. So where is it going to appear next, where should be put our focus, what livestock sector for instances and what location? Thirdly – and this was what was done in the U.K. with their modeling is estimate latent infections and if we’re looking at something like footand-mouth disease, where there is a substantial period of time where an individual or a herd or flock might be latently infected, cannot be detected. This is important to know when you are doing your simulation modeling or preparation, what is really out there, not only what you see. Fourthly to evaluate alternative controls and I think this is probably the most important part of simulation modeling to – when you have time to do the evaluations, you can look at a variety of scenarios, what ifs – what if we had vaccination, what if we had limited vaccination, what if we had vaccination of certain species, what if we had different restrictions, surveillance zones or an alternative to surveillance zones. Fifthly, identify high risk and vulnerable areas. I think that’s one of the prime focuses that I’ll be talking about and what was talked about earlier too. Where do we need to put our effort, where should we put our surveillance programs, where should we focus? Sixthly, to identify program needs and specifically looking at manpower and vaccination needs. If we were to have foot-and-mouth disease, carcass disposal is going to be a major event occurring. Do we have enough people for it, how long will it take, how many people do we need, how many people do we need for trace backs? If we were to vaccinate, how many would we need for that? Do we have enough vaccine; if not, what will the impact of that be?; ultimately identify potential hot spots. So, through simulation modeling you can run a variety of scenarios and identify places of focus. To illustrate all of these just as a reflection of what did occur four years ago in the U.K., devastating situation – that’s what we’re talking about here, where approximately 10% of the population were depopulated. I think somebody mentioned earlier that most of these individuals were not infected; most of them were either welfare or dangerous contacts, they were incriminated as being infected. So a 10% loss and some of these figures here – carcass disposal between 9,000 and 80,000 animals a day disposed, 100,000 carcasses as a peak. The good news is these were primarily sheep in the U.S. and I’m going to focus on the work that we’ve done in California to-date. We’re looking at primarily cattle and it’s a lot more difficult to dispose of cattle than sheep and it’s a lot more difficult to dispose of anything in California. So applications and quickly, these are the points that I want to illustrate to show you examples of applications in modeling. First of all I want to assess herd infectiousness and detection, secondly, evaluate diagnostic delay, what is the impact of a delay? Third, compare control alternatives and fourthly, assess the system vulnerability. So the first example – let me put it in perspective. I’m going to be talking about 2 geographic areas, one within the other. The first is this 3-county area in California, the Central Valley, where we had a study about seven years ago with Tom Bates, when he was a graduate student. For his dissertation, he was looking at the impact of FMD in that 3-county area and we had population identified there and movement activities well identified. So we want to use that as a preliminary area. Secondly, I’ll show some examples where we expand to the state as a whole. A previous speaker in one of the earlier sessions was talking about the livestock industry and the cattle population. I did some calculations on a number of animals in a herd, the number of herds in the U.S. and the average herd size came out to around 70 or 75 in a dairy. California, our average herd size is around 1,000, ranging up to maybe 10,000 animals. So it’s a very different situation we are dealing with in the states, especially in this 3-county area. So one of the benefits would be to assess infectiousness and detectability within a herd and we have an intra-herd model and some of the models for footand-mouth do this, some don’t; but we think it’s a benefit for a couple of reasons at least. First of all, what’s going on within the herd; it’s infected or not, it’s infectious or not, it can be detected or not. That’s a dichotomous situation that a lot of models will assume when in reality it’s a very dynamic situation. So in this example, we’re looking at a 1,000 cow dairy; we’re looking at an adequate contact rate of about one per hour meaning if there is an animal that is infected and infectious it could infect another animal at the rate of about 1 per hour, 1 animal Proceedings of the Institute of Food Technologists’ First Annual Food Protection and Defense Conference Simulation modeling . . . per hour. In a 24-h day we’ve got about 20 to 24 animals that can be infected by one infectious individual. It’s not really that far off on an assumption for foot-and-mouth disease because it’s the most contagious disease there is. But what does this mean as far as the herd infection level? Well looking at this, the assumption was that by day 9, one-half of the herd would become infected and that’s based on expert opinion. With that information, in about a week very few of the animals are actually infectious; so if somebody walks into a herd that is infected, few animals are infectious and it’s relatively unlikely that they will become contaminated and walk into the next herd and infect animals in that herd as well. Or if you were to move animals from the infected herd to a susceptible herd early on, it’s highly unlikely that that the moved animals would be infected. So there would be no disease transmission to the new herd. But if it’s a week or 2 wk later, you’ve got populations here at 7 d, almost half the herd infected, maybe 5% of the individuals infectious. So the probability of transmission is much higher and it increases dramatically over the period of 4 or 5 d later on. We think this is important for identifying if you have a trace back, how important is that contacted herd, how much surveillance effort should be put into determining whether or not that herd is infected, is it a highrisk herd or not for contamination or infection of a new herd? Looking at this in another way, looking at the clinical picture in that herd, how long would it take to detect that infected herd? Well, the same few numbers here – well we’ve got the infected population again, nine days on average, 50% infected but after about 10 d the number of clinically infectious animals is only approximately ten, which is one percent. Ten cows in a 1,000 cow dairy where you might have animals with very small clinical lesions, oral lesions, or you might have some animals slobbering, which is extremely common in dairies and they might be missed. So it might go another day or 2 before this herd would be presumed to be infected with foot-and-mouth disease. What does this mean; well we have more infectious individuals obviously to spread the disease, either in the milk or through people contacting the dairy or with animal movement. What does it mean as far as evaluation or detection of the virus; detection of the herd being infected, this is an illustration to show days post-diagnosis assuming that we could detect the herd as being infected on day whatever it is, when there are 10 animals or 1% infected. What would happen if that is delayed is that – this is the number of infected animals. So 450 or so are actually infected – I’m sorry, clinically affected. This number increases by about 150 per day that diagnosis is delayed. So a large portion of the population becoming infected with time. The good news is that it’s relatively low here early on – down on the bottom of the screen, very few animals are infectious and it’s taken off here so again, how long would it take to detect this herd; relatively and likely early on and with every day delayed there are more and more animals becoming infectious, infected, infectious, clinically infectious, and so on. So this is just for us a good look at what would happen; what do we expect to happen within a herd that is infected. A delay of diagnosis on a population level, population of herds, this is looking at the 3-county area again where we had 2200 plus herds in that population. We estimated that based on the U.K. experience 21 d is basically what occurred in the U.K. so we assume that would be similar to the U.S. before the index herd would be diagnosed. So with that, about 46 herds would on average, we believe become infected, about 2 plus percent of the population at risk. If the diagnosis were delayed a week, we estimated that the number of infected herds would more than double up to 111, but optimistically if we could detect it earlier, a week earlier, you could reduce that number from 46 down to 12 or 1/4 of the infected herds or infected population. Very optimistically, if we could reduce it to 7 d, for instance, if there were sniffers located on these herds or at the premises or if there were some sort of detection in the milk tank that was delivered to the creamery – the milk delivered to the creamery, this might be detected very early in a 7-d period at which case on average only the index herd would be infected. This can occur; it occurred very frequently in the U.K., where there were limited numbers of infected herds in the ’60s. Some back of the envelope calculations; what does this mean? We looked at the economic impact, only looking at direct cost of this three-county area and came up with a ball park figure of about $2.5 million dollars per hour delay. If we look at indirect cost, maybe that’s $10 million dollars. If we look at trade impact, that’s enormous. If we extend it to the states it’s maybe ten times worse than what we have here. So thinking about how much money should you put in through surveillance or early detection, this is one example of an estimate that if it occurs for every hour you save it’s worth $2.5 million dollars or more. Comparing alternatives – control alternatives – and I’ve got a couple of examples here. One is vaccination is possible; if we had the traditional base-line program of once a herd is identified as being infected, it’s depopulated in a timely fashion. I think that’s a 1-d period, then it would be estimated that in California we would have over 400,000 animals affected – ergo depopulated. Most of those are cattle, dairy cattle; so over 400,000 carcasses to dispose of in a period of about 4 mo. The good news is with vaccination you could reduce that in half, only 210 animals would have to be slaughtered and the epidemic would be much shorter, 40, 50 days in the state. The bad news is that currently there is not a vaccinate-to-live strategy, its vaccinate-to-kill. So those animals might have to be killed either after they live out their productive life, if they are dairy, or not. Also the bad news is we can’t possibly dispose of carcasses on a dairy, one thousand of them, within a day. Estimates are something like 4 to 6 min per head, up to 15 min per head for slaughter and carcass disposal. Do the math, it doesn’t workout for 24 h; it could take several days to weeks. If there were a state-wide movement in place throughout the epidemic and in California, as I understand it we would have, an immediate stand still of animals, 3-d movement then, it could be extended. So if that were the case, it could be reduced to less than 200,000 infected animals; the epidemic duration decreased as well. But to have a stand still of movement for 3 mo is unrealistic. Another strategy we are looking at is there are ring strategies and regional strategies. Typically there is a ring vaccination, a surveillance ring, an infected area ring, things are done in rings because of – I don’t know. You can calculate a radius of a circle and determine what’s within that circle and GIS programs; it’s really nice and easy to do to evaluate how many animals are at risk or the population that would be vaccinated for instance. Otherwise I’m really not sure of the rationale for ring vaccination, especially with movements being as they are, noncircular. So we looked at what would be the difference between having a regional strategy, a regional surveillance strategy versus a circular surveillance strategy whereby movements would be reduced, individual contacts would be reduced as well, surveillance would be that much stronger, stricter. We found that this light blue line indicates that you could reduce the number of infections from maybe 210 to 190 or so. So regional surveillance is beneficial in reducing the number of infected herds, but in this case we are assuming that if a herd is within 20 kilometers of an infected herd, the county that the new herd is in would be under quarantine or in a surveillance area. So it seems reasonable that looking at this as far as how many herds would be affected – in California let’s assume we have about 23,000 herds with the ring surveillance, maybe only 10% of the herds would be affected in that surveillance zone. On the other hand close to 25% would be affected if there were a regional surveillance zone put in place. So it would be much more difficult to manage. Our assumptions were that they would be equally affective and this is unlikely. So it’s unlikely that regional surveillance would be of much benefit and it would be at a much, much higher cost. Proceedings of the Institute of Food Technologists’ First Annual Food Protection and Defense Conference Simulation modeling . . . The next point is to test system vulnerability and as I’ve pointed out, this is a very intensive area in the three-county area and the state as well. This is an example looking at the three-county area and we have two bars here for each of the livestock categories. We have – this represents the portion of the population that is in that category; so about a third of the population in the three-county area are beef, 1/4 dairy and about a third of them backyard, small operations. The gray bar represents the proportion of herds that would be infected in an outbreak; so about 3/4 of the population, an infected population, we believe would be dairy. It’s a much larger risk for dairies and especially backyard, which has fewer contacts and the beef population as well. So this gives us an idea of maybe where we should focus our surveillance and what industry is at greatest risk. Also, if we look at the three-county area, if we look at index cases, where are we at greatest risk of having a large epidemic? We have the random introduction, about 50, 45 to 50 on average, herds would be infected, we believe. If that were beef, it’s much lower; if it’s dairy, you can basically double that, a much higher risk. If it were in a sales yard, as expected, the impact is going to be much more extreme. So instead of going from 45 or so with a 2%, we’re estimating up to 20% of the population could be infected. Accidental introduction is one way that we could get foot-and-mouth disease or any other pathogen into the country. Another route is intentional and if there were intentional introduction it’s unlikely that it would be just in a single premises because that wouldn’t cause as much impact as multiple premises. So we looked at multiple index cases, starting with our baseline of 1, going up to 10 and simulated what the impact would be if it were – if foot-and-mouth disease were introduced into a random area in California, so not going after the big ones, just randomly in a varied population. So we go from 200 to over 800 to 900 if there were multiple introductions of the virus. It would get even worse if we had a couple of serotypes; there are 7 different serotypes and about 69 different strains. Possibly, you might recognize one serotype and vaccinate against that where there are multiple serotypes introduced that you could miss and the vaccine would not be effective. I wanted to go through a couple of things that we are working on. One is calf ranches that we have in California that is different, I think, from much of the rest of the U.S. but they are growing. We’ve got these calves that – this is a heifer calf that’s got double ear tags which means that she came from a particular farm, she’s going to go back to that farm in a few months. In the meantime she’s going to be raised in this calf ranch, which does an extremely good job of raising the heifers, very, very low mortality and morbidity. This particular example – each of these buckets represents a calf home – and on this particular premises there were probably 12,000 to 15,000 heifer calves being raised on a single location. Population in California we believe is around 200; ranging from small of a couple of thousand to large of 70 to 80,000 cows on a single location. Contacts – looking at this for a medium operation, 15,000, 10 to 15,000, they service or receive heifer calves from approximately 100 dairies in a day. So they are contacting 100 dairies; somebody is going onto the dairy, picking up a cow, putting it in a truck, going onto another dairy and doing this maybe 20 times per truck. We’ve got five or so people out there doing this for this one. The range is quite variable from 1 to 3,000 miles they get cows from New York, with a lot of employees, so a lot of indirect contact. So we need to develop a new type of modeling here and we’re looking at social network, where we have identified regular, not random contacts occurring. So this is a future direction we’re thinking of. California State Fair; the State Fair is a lovely place to go see all of these animals out there, a lot of 4H kids, a lot of FFA kids. We went out to survey, just to see what sort of security they had, where they were going and what they were going back to. So we got some goats here, some cows there and on dairy day you’ve got dairy goats and dairy cows mixed together. Very good, very clean operation; the individuals are extremely well cared for but the general public has access to all the animals in the population. These animals go back-and-forth in the walkways, if one is infected, it’s easy to expose another. We looked at a three-week period with about 1,000 entrants, surveyed over 100 of them; people are very willing to respond. We found out the important thing here is that we had 60% of the population going home to a commercial operation. So if exposed at the Fair, they would take it home with them. Conclusions – I’ve got about 4 or 5 here. We like simulation modeling because we can predict within and among herd disease spread – and we don’t have a laboratory to do this – well, we do. We have a simulation modeling lab but we’re not going to experientially infect herds to see what’s going on. We don’t have information around the world that would be similar to what’s going on in a population in the U.S. So we have to use simulation models to do these predications. We can identify systems, which I hopefully have shown you, and evaluate alternative controls for disasters ultimately. So modeling may be useful before, during and after exotic disease epidemics. Obviously, before where we have a lot of time during – where we have to respond very quickly without much time and after, again, we have a lot of time. In the U.K. they are still collecting data on the 2001 epidemic and validating their models with that. I think we need to focus on the pre-epidemic planning because that’s when we do have the time to develop these models, expand them to be responsive to the decision makers who want to evaluate different aspects of the model, different scenarios that during an epidemic we wouldn’t have time to do. The last statement is models are only as good as the data that goes into them. Obviously, the garbage-in-garbage-out, and we need these data now. If anybody has data hanging around as far as premises locations, as far as animal movements, we know they are out there, we know people have them. The modelers need this right now to create effective models, realistic models; otherwise we’ll be doing our best guesses, making it our best estimates at the populations out there. Carl Mellius, in the back, has been doing a lot of this with NASS data; he doesn’t have the data, but he’s doing a best guess. That’s what – I don’t want to say, you are stuck with right now, Carl, but it’s not as good as if we had precise data and I’m sure you’ll admit to that I think. So that’s my plea; we need the data, we need the data now, otherwise we’ll do as good as we can and hopefully that’s okay. Proceedings of the Institute of Food Technologists’ First Annual Food Protection and Defense Conference Simulation Modeling as an Exotic Livestock Disease Disaster Planning Tool Tim Carpenter School of Veterinary Medicine Center for Animal Disease Modeling and Surveillance (CADMS) University of California, Davis Presentation Outline Why model? Examples of modeling in disease disaster planning Why model? (Goals of Epidemic Simulation Modeling) 1. Predict disease incidence 2. Predict spatial and category spread 3. Estimate latent infections 4. Evaluate alternative controls Why model? (Goals of Epidemic Simulation Modeling) 5. Identify high risk and vulnerable areas 6. Identify program (e.g. manpower and vaccine) needs 7. Identify potential hotspots Slaughter/Disposal Numbers = 6-10 million livestock (“officially” 6,456,000: 10% of UK total) Daily average of animals slaughtered = 9,000 - 80,000 Peak daily disposal = 100,000 carcasses Source: Pam Hullinger, CDFA Model Applications 1. Assess Herd Infectiousness and Detection 2. Evaluate Diagnostic Delay 3. Compare Control Alternatives 4. Assess System Vulnerability Dairy Beef Swine Sheep/goat Backyard Assess Herd Infectiousness and Detection 1000-cow Dairy (k=0.91/hr.; CID50=9) 1000 Prevalence 800 Infected 600 400 Infectious 200 0 0 5 10 Time (days) 15 20 1000-cow Dairy Prevalence 1000 800 Clinical 600 Infected 400 200 Subclinical 0 0 5 10 Time (days) 15 20 Evaluate Diagnostic Delay 1000-cow Dairy Simulation Results (diagnosis based on 1% (10) clinical cases) N u m b e r i n fe c te d 1000 800 600 400 200 0 0 1 2 3 4 5 Days post diagnosis N u m b e r i n fe c ti o u s 1000 800 600 400 200 0 0 1 2 3 Days post diagnosis 4 5 Impact of delays in diagnosis of the index case (3-county area of CA) Cumulative no. inf. herds 1 12 46 111 $2.6 mill/hr. delay 0.9 0.8 0.7 % herds infected / day Diagnosis day 7 14 21 28 1-day delay 0.6 7-day delay 0.5 14-day delay 21-day delay 0.4 28-day delay 0.3 0.2 0.1 0 0 7 14 21 28 35 42 49 Days after index herd exposed 56 63 70 Compare Control Alternatives Expected Statewide Impact with 3 Alternative Controls Cumulative Carcases (1000) 500 433,000 450 400 350 300 250 210,000 200 180,000 150 Baseline 100 Statewide movement ban 50 Vaccination 0 20 40 60 80 100 Epidemic Duration (days) 120 140 Cum. inf. premises 50 100 150 Mean no. cumulative infected premises, with circular vs. regional surveillance (Index=dairy) 0 Circular SZ Regional SZ 0 50 100 150 Time (days post initial infection) 200 0 1000 Premises 2000 3000 4000 5000 Mean no. premises located in surveillance zone, with circular vs. regional surveillance (Index=dairy) 0 50 100 150 Time (days post initial infection) 200 Assess System Vulnerability Distributions of all herds and infected herds 80 73 Percent of all herds Percent of infected herds 70 Percent 60 50 40 30 35 30 26 20 10 10 5 4 5 6 5 2 0 0 Beef Dairy Swine Goat/sheep Backyard Sales yard Effect of index herd type on epidemic size 600 No. herds infected 500 Total no. herds = 2,238 400 300 200 100 0 Beef Dairy Swine Sheep/Goat Backyard Sales yard Possible FMD impact in CA given alternative no. of index cases 800 858 Total infected premises 200 400 600 630 449 205 0 1 index case 3 index cases 5 index cases 10 index cases 0 50 100 Time (days) 150 200 Calf Ranches Calf Ranches Population - approx. 200 in CA Size - Small (<2000) to Large (70,000+) Contacts (DCs) – medium (10-15K) 75 - 100 dairies/day range = 1 - 3,000 miles (IDCs) > 100 employees Modeling – need to construct/evaluate the “social network” connecting dairies, calf ranches, sales yards and feedlots. CA State Fair CA State Fair Approximately 1,000 “entered” exhibitors in a 3-week period (Aug. – Sept., 2005). Surveyed 137 (> 95% participation rate). 82 (60%) of the exhibitors returned with animals to a commercial premises. Conclusions Simulation modeling may be useful to: • predict within and among herd disease spread • evaluate the need for improved surveillance • identify system vulnerability • evaluate alternative controls • plan for disasters Additional Thoughts 1. Modeling may be useful before, during and after exotic disease epidemics occur. 2. The focus needs to be on pre-epidemic planning because there is little time to act during the actual epidemic. 3. Models are only as good as the data that goes into them; hence, data relating to premises and individual animal movement should be made available a.s.a.p.