* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chemical Kinetics
Hypervalent molecule wikipedia , lookup
Asymmetric induction wikipedia , lookup
Electrochemistry wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Process chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Marcus theory wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Chemical reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Hydroformylation wikipedia , lookup
Supramolecular catalysis wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Stoichiometry wikipedia , lookup
Click chemistry wikipedia , lookup
George S. Hammond wikipedia , lookup
Reaction progress kinetic analysis wikipedia , lookup
Rate equation wikipedia , lookup
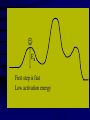
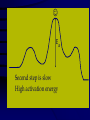
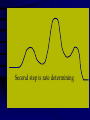
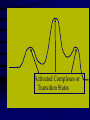
Chemical Kinetics Rates at which chemical reactions occur. Main goal is to understand the steps by which a reaction takes place: Reaction Mechanism 1 As we will see, there are several factors which affect the rate of a chemical reaction including: 1. the nature and concentrations of the reactants. 2. the temperature of the reaction system. 3. the presence of a catalyst and 4. the surface area of the reactants or catalyst. 2 Reaction Rate Change in concentration of a reactant or product per change in time. ∆[ ] = [final] – [initial] Rate = ∆[reactants] ∆t Rate = ∆[products] ∆t concentration concentration ( )Sign is used if the [ ] is decreasing Units usually molar/second 3 2NO2(g) 2NO(g) + O2(g) in a flask at 300C, measure change in concentration as it decomposes. Calculate the average rate it changes in the first 50 sec. .0079mol / L .0100mol / L 50. s 0s 4.2 x10 5 mol / L s (-)means decreasing 4 Instantaneous rates Value at a particular time: slope of the line tangent to the curve at that point. y [ NO2 ] x t [ NO2 ] Rate t Rate ( slope of tan gent ) 0.0026mol / L 110s 2.4 x10 5 mol / L s 5 Rates and Stoichiometry Relative rates (need to look at the coefficients) 2NO2(g) 2NO(g) + O2(g) Must include reference: disappearance of reactant appearance of product 6 2NO2(g) 2NO(g) + O2(g) Rate of consumption = Rate of production =2(rate of production of NO2 of NO of O2) NO 2 t = ∆[NO] ∆t 2 O2 t Rate of O2 production is half that of NO 4PH3 P4 + 6H2 7 Example: 2A + 3B C + 4D What is the rate of B, C and D in reference to one mole of A? - Δ[A] Δt 3 -Δ[B] = 1 Δ[C] = / / 2 Δt 2 Δt = 2 Δ[D] Δt 8 Rate Laws 2NO2(g) 2NO(g) + O2(g) Reactions are reversible Choose conditions where the reverse has negligible contributions. Study at a point soon after they are mixed before product builds up. Reaction rate will depend only on concentration of the reactants. 9 Rate Laws Two key points 1. The concentration of the products do not appear in the rate law because this is an initial rate. 2. The order (exponent) must be determined experimentally, can’t be obtained from the equation So, what is a Rate Law? • Algebraic expression of the relationship between concentration and the rate of a reaction at a particular temperature. • Constant of proportionality in the expression is given the symbol k and is referred to as the specific rate constant for the reaction 11 Rate Laws ( ignore reverse) Rate = k[NO2]n k = rate constant (constant of proportionality) n = rate order (must be determined experimentally not from a balanced equation) can be an integer including 0. [product] does not appear in the rate law 12 Types of Rate Laws Differential Rate Law: expresses how rate depends on concentration. (rate law) Integrated Rate Law: expresses how concentration depends on time. 13 Determining the Form of the Rate Law *how a reaction occurs* 1. Need to determine n (order) 2N2O5(aq) 4NO2(aq) + O2(g) N2O5 = 0.90M = 0.45M 5.4 x 10 -4mol/L·s 2.7 x 10-4mol/L·s ½ the concentration , ½ the rate 14 Slope of the tangent to the curve 15 This means the rate of the reaction depends on concentration of N2O5 to the first power. The differential rate law is: [ N 2 O5 ] Rate k [ N 2 O5 ]1 k [ N 2 O5 ] t ***first order reaction doubling the concentration doubles the reaction rate. 16 Reaction Rate and Concentration Click in this box to enter notes. Go to Slide Show View (press F5) to play the video or animation. (To exit, press Esc.) This media requires PowerPoint® 2000 (or newer) and the Macromedia Flash Player (7 or higher). [To delete this message, click inside the box, click the border of the box, and then press delete.] Copyright © Houghton Mifflin Company. All rights reserved. Method of Initial Rates (experimentally determining the form of the rate law) Initial Rate: the “instantaneous rate” just after the reaction begins before the [initial] of reactants have changed significantly. Just after t=0 1. Run the reaction several times 2. Vary initial concentrations 3. Measure reaction rate just after reaction is mixed 18 NH4+(aq) + NO2-(aq) →N2(g) + 2H2O(l) Double the NO2 ; double the initial rate Rate k[ NH4 ]n [ NO2 ]m 19 Rate 2 = 2.7 x 10-7 = (.0100)m = (2.0)m Rate 1 1.35 x 10-7 (.0050)m =2.00 = (2.0)m In terms of NO2- Value of m=1 First order reaction in terms of NO2-. 20 Rate 3 = 5.4 x 10-7 = (.200)n = (2.0)n Rate 2 2.7 x 10-7 (.100)n =2.00 = (2.0)n In terms of NH4+ Value of n = 1 and the value of m=1 First order in both NH4 and NO2 Rate law is : Rate = k[NH4+][NO2-] 21 Overall Reaction Order Sum of the order of each component in the rate law. rate = k[NH4+][NO2-] The overall reaction order is 1 + 1=2. 22 Calculate k 1.35x10-7=k(0.100mol/L) (0.0050mol/L) k=2.7 x 10-4L/mol.s 23 Sample 12.1 The reaction between bromate ions and bromide ions in acidic aqueous solution is given by the equation BrO3-(aq) + 5Br-(aq) + 6H+(aq) →3Br2(l)+ 3H2O(l) Copyright©2000 by Houghton Mifflin Company. All rights reserved. 24 Determine the order for all three reactants, the overall reaction Order, and the value of the rate constant. Rate = k[BrO3-]n[Br-]m[H+]p Determine the values of n, m, and p by comparing the rates from the various experiments. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 25 n= 1 m= 1 p= 2 The rate is 1st order in BrO3- and br-, and 2nd order in H+. The overall reaction order is n + m + p = 4 Rate law is written as; Rate = k[BrO3-][Br-][H+]2 K= 8.0 L3/mol3•s Copyright©2000 by Houghton Mifflin Company. All rights reserved. 26 Integrated Rate Laws So far looked at rate as a funx of the [reactant]. Also useful to express the [reactant] as a funx of time, give the (differential) rate law for the reaction. 27 2N2O5(soln) → 4NO2(soln) + O2(g) [ N 2 O5 ] Rate k [ N 2 O5 ] t Since the rate depends on the [N2O5] to the 1st power, 1st order reaction This means if the [N2O5] doubled, the rate would double. Put into different form using calculus 28 First-Order Rate Law For aA Products in a 1st-order reaction, A Rate = k A t Integrated first-order rate law is ln[A] = kt + ln[A]o **Concentration as a function of time** 29 Integrated first-order rate law is ln[A] = kt + ln[A]o 1. The equation shows how [A] depends on time. 2. Is in the form y=mx + b, where a plot of y vs. x is a straight line with slope m and intercept b. y=ln[A] x=t m=-k b=ln[A]0 30 For the reaction: aA→products The reaction is first order in A if a plot of ln[A] versus t is a straight line. If it is not a straight line it is not 1st order. 3. This integrated rate law for a first-order reaction also can be expressed in terms of ratio of [A] and [A]0 as follows; [ A]0 ln kt [ A] 31 2N2O5(soln) → 4NO2(soln) + O2(g) Using data, verify that the rate law is first order in [N2O5], And calculate the value of the rate constant. [N2O5] (mol/L) .1000 0.707 0.0500 0.0250 0.0125 0.00625 Time (s) 0 50 100 200 300 400 32 Figure 12.4: A plot of ln[N2O5] versus time. This verifies that the reaction is 1st order. 33 ln[N2O5] = -kt + ln[N2O5]0 y = mx + b Since the reaction is 1st order, the slope of the line equals -k, where change y (ln[ N 2 O5 ]) Slope change x t 1 5075 . (2.303) 2772 3 slope 6.93x10 s 400s 0s 400 k ( slope) 6.39 x10 3 s 1 34 Using the data given, calculate [N2O5] at 150s after the start of the reaction. [N2O5] (mol/L) .1000 0.707 0.0500 0.0250 0.0125 0.00625 Time (s) ln[N2O5] = -kt + ln[N2O5]0 k =6.39x10-3s- 0 50 100 200 300 400 ln([ N 2 O5 ]) t 150 (6.93x10 3 s 1 )(150s) ln(010 . ) 1040 . 2.303 3.343 [ N 2 O5 ]t 150 anti log( 3.343) 0.0353mol / L 35 Half-Life of a First-Order Reaction Time required to reach [1/2] t1/2 0.693 k t1/2 = half-life of the reaction k = rate constant For a first-order reaction, the half-life does not depend on concentration. 36 Figure 12.5: A plot of [N2O5] versus time for the decomposition reaction of N2O5. t1/2=100s Copyright©2000 by Houghton Mifflin Company. All rights reserved. 37 Formula for t1/2 is derived from integrated rate law: aA→ product If the reaction is 1st order in [A] [ A]0 ln kt [ A] [ A]0 [ A] 2 By definition when t=t1/2, [ A]0 kt1/ 2 ln [ A]0 / 2 ln(2) kt1/ 2 t1/ 2 0.693 k 38 Half-life first order reaction A certain first-order reaction has a half-life of 20.0min. a. Calculate the rate constant for this reaction. b. how much time is required for this reaction to be 75% complete. 39 1. 0.693 k 3.47 x10 2 min 20.0 min 2. Using the integrated rate law in the form [ A]0 ln kt [ A] If the reaction is 75% complete, 75% of reactant has been consumed, leaving 25% in the original form. [ A] x100% 25% [ A0 ] This means that [ A]0 [ A] 1 .25 or 4.0 [ A]0 [ A] .25 40 [ A] 347 . x10 2 ln 4.0 kt then ln t min [ A]0 and ln 4.0 t 2 40.min 3.47 x10 min Copyright©2000 by Houghton Mifflin Company. All rights reserved. 41 Another way to solve.. • t1/2 is at 50% completion • If the [1mol/L]0, after one half-life the [0.50mol/L]. • One more half-life produces [0.25mol/L]. • 25% of reactant is left. • Two half-lives = 2(20.0min)=40.0min 42 Second-Order Rate Law For aA products in a second-order reaction, A Rate = k A t Integrated rate law is 2 *Doubling the concentration quadruples the reaction rate. 1 1 kt + A Ao 43 1 1 kt + A Ao 1. A plot of 1/[A] versus t will produce a straight line w/slope = k 2. Shows how [A] depends on time and can be used to calculate [A] at any time t, provided k and [A]0 are known. 44 Half-Life of a Second-Order Reaction t1/2 1 kA o t1/2 = half-life of the reaction k = rate constant Ao = initial concentration of A The half-life is dependent upon the initial concentration. 45 Half life of second order reaction At t=t1/2, [ A]0 [ A] 2 Then the equation becomes 1 1 kt 1/ 2 + A 0 A o 2 2 1 kt 1/ 2 [ A]0 Ao 1 kt 1/ 2 A o Solve for t1/ 2 1 k [ A]0 46 Butadiene reacts to form its dimer according to the equation: 2C4H6(g)→C8H12(g) (two identical molecules combine) The following data were collected for this reaction at a given temp : [C4H6](mol/L) Time (+,- s) 0.01000 0.00625 0.00476 0.00370 0.00313 0.00270 0.00241 0.00208 0 1000 1800 2800 3600 4400 5200 6200 a. Is the reaction 1st or 2nd order? b. What is the value of the rate constant for the reaction? c. What is the half-life for the reaction under the conditions of this experiment? 47 Figure 12.6: (a) A plot of ln[C4H6] versus t. (b) A plot of 1n[C4H6] versus t. 48 a. Is it first or second order? Write the rate law. Rate = 2nd C 4 H6 t k C H 4 2 6 49 b. What is the rate constant? 2nd order: plot 1/[C4H6] vs. t and get a straight line with slope of k 1 ( ) change y C4 H6 Slope change x t 1 (481 100) L / mol 381 slope 614 . x10 2 s 6200s 0s 6200 50 c. Half-life? t1/2 1 kA o t1/2=1/(6.14x10-2L/mol.s)(1.000x10-2mol/L) = 1.63x103s 51 Difference between ½-life of a 1st order and 2nd order reaction 2nd order •Depends on both k and [A]0 •Each ½ life is double of the preceding one 1st order •Depends only on k •Constant time for each ½-life 52 Zero order reaction Rate =k[A]0=k(1)=k The rate is constant (does not change with concentration) Integrated rate law : [A]=-kt+[A]0 Plot of [A] versus t gives a straight line of slope -k 53 Half-life [A]=[A]0/2 when t=t1/2 , so [ A]0 kt1/ 2 [ A]0 2 [ A]0 t1/ 2 2k 54 Figure 12.7: A plot of [A] versus t for a zeroorder reaction. Zero order Metal surface or an enzyme is required for the reaction to occur. 56 The decomposition reaction 2N2O(g) → 2N2(g) + O2(g) takes place on a platinum surface. Concentration has no effect on the rate 57 More Complicated Reactions • BrO3- + 5 Br- + 6H+ 3Br2 + 3 H2O • For this reaction we found the rate law to be • Rate = k[BrO3-][Br-][H+]2 • To investigate this reaction rate we need to control the conditions Rate = k[BrO3-][Br-][H+]2 • We set up the experiment so that two of the reactants are in large excess. • [BrO3-]0= 1.0 x 10-3 M • [Br-]0 = 1.0 M • [H+]0 = 1.0 M • As the reaction proceeds [BrO3-] changes noticeably • [Br-] and [H+] don’t Rate = k[BrO3-][Br-][H+]2 • Assume that through the reaction: [Br-] = [Br-]0 [H+] = [H+]0 Therefore the rate law can be written: 60 Rate = k[BrO3-][Br-][H+]2 • Rate = k[BrO3-][Br-]0[H+]02 • Rate = k[BrO3-][Br-]0[H+]02 k’ = k[Br-]0[H+]02 Rate = k’[BrO3-] This is called a pseudo first order rate law. k’ k= [Br-] [H+] 2 A Summary 1. Simplification: Conditions are set such that only forward reaction is important. 2. Two types: 3. Which type? Depends on the type of data collected - differential and integrated forms can be interconverted. differential rate law integrated rate law 62 A Summary (continued) 4. Most common: method of initial rates. 5. Concentration v. time: used to determine integrated rate law, often graphically. 6. For several reactants: choose conditions under which only one reactant varies significantly (pseudo first-order conditions). 63 Copyright©2000 by Houghton Mifflin Company. All rights reserved. 64 Reaction Mechanism The series of steps by which a chemical reaction occurs. (bond making and bond breaking) Must be determined by experiment! Must agree with overall stoichiometry AND the experimentally determined rate law. A chemical equation does not tell us how reactants become products - it is a summary of the overall process. 65 Elementary steps: the steps in the reaction mechanism that add up to the balance equation Molecularity: how the molecules collide The rate for a reaction can be written from its molecularity . 66 Reaction Mechanisms 2NO2 + F2 2NO2F Rate = k[NO2][F2] The proposed mechanism is: Elementary steps NO2 + F2 NO2F + F (slow) F + NO2 NO2F (fast) 2NO2 + F2 2NO2F F is called an intermediate it is formed then consumed in the reaction Formed in reversible reactions 2 NO + O2 2 NO2 Mechanism 2 NO N2O2 (fast and reversible) N2O2 + O2 2 NO2 (slow) Determining! rate = k[N2O2][O2] No intermediates Substitute [NO]2 for [N2O2] What is the rate determining step in this video clip Collision Model Key Idea: Molecules must collide to react. However, only a small fraction of collisions produces a reaction. Why? Arrhenius: An activation energy must be overcome. Mechanism Sim Copyright©2000 by Houghton Mifflin Company. All rights reserved. 69 Factors that Affect Reaction Rate 1. Concentrations of reactants • More reactants mean more collisions if enough energy is present 2. Temperature • Collision Theory: When two chemicals react, their molecules have to collide with each other with sufficient energy for the reaction to take place. • Kinetic Theory: Increasing temperature means the molecules move faster. Collision Theory • • • • • • Particles have to collide to react. Have to hit hard enough Things that increase this increase rate High temp – faster reaction High concentration – faster reaction Small particles = greater surface area means faster reaction Rates and collisions However… • Has been found that the rate of a reaction is much smaller than the calculated collision frequency in a collection of gas particles. • Therefore only a small fraction of the collisions produces a reaction. WHY? Copyright©2000 by Houghton Mifflin Company. All rights reserved. 72 Arrhenius Equation Collisions must have enough energy to produce the reaction (must equal or exceed the activation energy (threshold energy)). Orientation of reactants must allow formation of new bonds. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 73 Figure 12.11: (a) The change in potential energy as a function of reaction progress for the reaction 2BrNO 2NO + Br2. The activation energy Ea represents the energy needed to disrupt the BrNO molecules so that they can form products. The quantity ∆E represents the net change in energy in going from reactant to products. (b) A molecular representation of the reaction. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 74 Figure 12.12: Plot showing the number of collisions with a particular energy at T1 and T2, where T2 > T1. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 75 Temperature Dependence of the Rate Constant **10 increase will double the rate** k = A • exp( -Ea/RT ) (Arrhenius equation) Ea is the activation energy (J/mol) R is the gas constant (8.314 J/K•mol) T is the absolute temperature A is the frequency factor -Ea 1 Ln k = + lnA R T 13.4 ln(k) = -Ea/RT + ln(A) Catalysts • Speed up a reaction without being used up in the reaction. • Enzymes are biological catalysts. • Homogenous Catalysts are in the same phase as the reactants. • Heterogeneous Catalysts are in a different phase as the reactants. Ways to speed up a reaction: 1. Catalysts Speed up reactions by lowering activation energy 2. Surface area of a solid reactant Bread and Butter theory: more area for reactants to be in contact 3. Pressure of gaseous reactants or products Increased number of collisions 79 Catalysts allow reactions to proceed by a different mechanism - a new pathway with a lower activation energy. More molecules will have this activation energy. Does not affect ∆E Show up as a reactant in one step and a product in a later step 80 Figure 12.16: Effect of a catalyst on the number of reaction-producing collisions. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 81 Heterogeneous Catalysis (gas on a solid surface) Steps: 1. Adsorption and activation of the reactants. 2. Migration of the adsorbed reactants on the surface. 3. Reaction of the adsorbed substances. 4. Escape, or desorption, of the products. 82 Heterogenous Catalysts H H • Hydrogen bonds to surface of metal. • Break H-H bonds H H H H H H Pt surface Hydrogenation of unsaturated fats Heterogenous Catalysts H H H C C H H H H H Pt surface Heterogenous Catalysts • The double bond breaks and bonds to the catalyst. H H H C H C H H Pt surface H H Heterogenous Catalysts • The hydrogen atoms bond with the carbon H H H C H C H H Pt surface H H Heterogenous Catalysts H H H H C C H H H Pt surface catalyst sim H Homogenous Catalysts (same phase) • Chlorofluorocarbons (CFCs) catalyze the decomposition of ozone. • Enzymes regulating the body processes. (Protein catalysts) Ostwald Process Pt catalyst 4NH3 (g) + 5O2 (g) 4NO (g) + 6H2O (g) 2NO (g) + O2 (g) 2NO2 (g) 2NO2 (g) + H2O (l) HNO2 (aq) + HNO3 (aq) Pt-Rh catalysts used in Ostwald process Hot Pt wire over NH3 solution 13.6 Catalytic Converters CO + Unburned Hydrocarbons + O2 2NO + 2NO2 catalytic converter catalytic converter CO2 + H2O 2N2 + 3O2 13.6 Enzyme Catalysis 13.6 Catalysts and rate • Catalysts will speed up a reaction but only to a certain point. • Past a certain point adding more reactants won’t change the rate. • Zero Order Catalysts and rate. R a t e • Rate increases until the active sites of catalyst are filled. • Then rate is independent of concentration Concentration of reactants • This reaction takes place in three steps Ea First step is fast Low activation energy Ea Second step is slow High activation energy Ea Third step is fast Low activation energy Second step is rate determining Intermediates are present Activated Complexes or Transition States Catalysts • Speed up a reaction without being used up in the reaction. • Enzymes are biological catalysts. • Homogenous Catalysts are in the same phase as the reactants. • Heterogeneous Catalysts are in a different phase as the reactants. How Catalysts Work • Catalysts allow reactions to proceed by a different mechanism - a new pathway. • New pathway has a lower activation energy. • More molecules will have this activation energy. • Does not change E • Show up as a reactant in one step and a product in a later step Figure 12.15: Energy plots for a catalyzed and an uncatalyzed pathway for a given reaction. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 103 Chapter 12(b) Chemical Kinetics (cont’d) Copyright©2000 by Houghton Mifflin Company. All rights reserved. 104 Copyright©2000 by Houghton Mifflin Company. All rights reserved. 105 Figure 12.9: A molecular representation of the elementary steps in the reaction of NO2 and CO. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 106 12.10: A plot showing the exponential dependence of the rate constant on absolute temperature. orientations for a collision between two BrNO molecules. Orientations (a) and (b) can lead to a reaction, but orientation (c) cannot. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 108 for the reaction 2N2O5(g) 4NO2(g) + O2(g). The value of the activation energy for this reaction can be obtained 12.17: Heterogeneo us catalysis of the hydrogenatio n of ethylene. Figure 12.18: The exhaust gases from an automobile engine are passed through a catalytic converter to minimize environmental damage. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 111 removal of the end amino acid from a protein by reaction with a molecule of water. The products are an amino acid and a enzyme carboxypeptidase-A, which contains 307 amino acids. The zinc ion is shown above as a black sphere in the center. (b) Carboxy-peptidase-A with a substrate (pink) in place. Copyright©2000 by Houghton Mifflin Company. All rights reserved. 113 interaction. The substrate is shown in black and red, with the red representing the terminal amino acid. Blue indicates side chains from the enzyme that help bind the substrate.