* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Introduction to gauge theory wikipedia , lookup
Gibbs free energy wikipedia , lookup
Old quantum theory wikipedia , lookup
Conservation of energy wikipedia , lookup
Thermal conduction wikipedia , lookup
Photon polarization wikipedia , lookup
Condensed matter physics wikipedia , lookup
Nuclear physics wikipedia , lookup
Hydrogen atom wikipedia , lookup
Thermal conductivity wikipedia , lookup
Density of states wikipedia , lookup
Electron mobility wikipedia , lookup
Electrical resistivity and conductivity wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Condensed Matter Physics
J. Ellis (10 Lectures)
Periodic Systems: Overview of crystal structures, the reciprocal lattice.
Phonons: Phonons as normal modes – classical and quantum picture. 1D
monatomic chain, 1D diatomic chain, examples of phonons in 3D. Debye
theory of heat capacity, thermal conductivity of insulators.
Electrons in solids:
Free electron model: Fermi-Dirac statistics, concept of Fermi level,
electronic contribution to heat capacity. Bulk modulus of a nearly free
electron metal. Electrical and thermal conductivity. Wiedemann-Franz law.
Hall effect.
Nearly free electron model: Derivation of band structure by considering
effect of periodic lattice on 1-D free electron model. Bloch’s theorem.
Concept of effective mass. The difference between conductors,
semiconductors and insulators explained by considering the band gap in
2D. Hole and electron conduction.
Doping of semiconductors, p and n types, pn junctions – diodes, LEDs and
solar cells.
Books
In general the course follows the treatment in Solid State Physics, J.R.
Hook and H.E. Hall (2nd edition, Wiley, 1991).
Introduction to Solid State Physics, Charles Kittel (8th edition, Wiley,
2005) is highly recommended. (need not be the latest edition)
Another book, generally available in College libraries and may usefully be
consulted is The Solid State, Rosenberg H M (3rd edn OUP 1988)
Webpage http://www-sp.phy.cam.ac.uk/~je102/
1
Condensed Matter Physics:
Periodic Structures
• Course deals with crystalline materials – can be
extended later to amorphous materials.
• Crystalline structure characterised by set of lattice
points – each in equivalent environment, but not
necessarily at the position of an atom.
• Each lattice point will have associated with it one or
more atoms - the ‘basis’. e.g. NaCl
Structure
Lattice
=
Basis
*
• (Mathematically, the lattice would be represented by
an array of delta functions, and the crystal described
by a convolution of the lattice with a function that
described e.g. the electron density associated with
the basis.)
2
Condensed Matter Physics:
Unit Cells
• Lattice described by a unit cell – which may have
one lattice point per unit cell (a ‘primitive’ unit cell) or
more than one (‘non-primitive). e.g. for cubic:
• Primitive Cubic
• Face Centred Cubic (fcc)
Non primitive unit cell, 4
lattice points per unit cell
Primitive unit cell, 1
lattice point per unit cell
• Body Centred Cubic
(bcc)
Non primitive, 2 lattice
points per unit cell
• How many lattice points per unit cell? Either count
those at corners and face centres with weight 1/8
and ½ respectively, or move whole cell so that no
lattice points are on the sides/corners, and count
lattice points inside the cell.
3
Bravais Lattices
In 3D there are
14 different
lattices – know
as ‘Bravais’
lattices.
P=primitive
I= body centred
F=Face centred
on all faces
A,B,C = centred
on a single face
Need to
remember the
P,I, and F forms
of the cubic unit
cells
4
Directions
• Unit cells characterised by the 3 ‘lattice vectors’
(a,b, and c) that define their edges.
e.g. for a face centred cubic (fcc) lattice
Unit Cell
Lattice Vectors
c
Non primitive
b
a
Primitive
c
b
a
• Directions given in terms of basis vectors – a
direction:
r ua vb wc
would be writen as [u,v,w].
_
_
_
• In a cubic lattice [100], [010], [001], [1 00], [01 0], [001]
are all related by symmetry. They are together
denoted by 100 .
5
Planes
• The notation describing a set of uniformly spaced
planes within a crystal is defined as follows:
• Assume one of the planes passes through the origin
• Look at where the next plane cuts the three axes that
are defined by the three lattice vectors.
• If the plane cuts the three axes at a/h , b/k , c/l , then
the set of planes is described by the Miller indices
(h,k,l), and {h,k,l} indicates all planes related to (h,k,l)
by symmetry.
• If h,k, or l is zero it indicates that the plane is parallel
to the respective axis.
e.g. (showing only the plane next to one that contains the origin)
(111)
z
c/
(120)
z
l
y
y
b/
b/
l
a/
l
x
2
a/
l
x
6
Fourier Transforms and The
Reciprocal Lattice
•
1D periodic functions
•
A 1D periodic function, f(x)=f(a+x), can be represented as a
Fourier series:
ik x
Ch e
f ( x)
h
h
where : k h 2h
a
The wave vectors used, kh are a uniformly separated set of points
in 1D wave vector (k) space.
•
To illustrate how a 3D Fourier series is built up consider the
orthorhombic case (a ≠ b ≠ c, 90° between axes)
•
In 2D, the coefficients Ch vary with y:
Ch y eikh x
f ( x, y )
•
h
But the function is periodic in y, so represent Ch(y) as a Fourier
series:
Ch ( y )
•
C
k
hk
eikk y where : k k 2k
In 3D the Chk vary with z: f ( x, y, z )
b
ikh x k k y
C
z
e
hk
h , k
•
And again since the function is periodic in z, Chk (z) can be
written as a Fourier series:
Chk ( z )
•
C
l
Hence the 3D series is:
hkl
eikl z where : kl 2l
f ( x, y, z )
C
h , k ,l
•
hkl
c
e i ( k h x k k y kl z )
k vectors, (kh,kk,kl), needed for the Fourier transform form a
lattice in 3D reciprocal space known as the RECIPROCAL
LATTICE.
7
The Reciprocal Lattice: the
General Case
• For a periodic function in 3D with a lattice described by
lattice vectors a, b and c, all the wavevectors you need in
3D k space for a 3D Fourier transform representation are:
G hkl hA kB lC
(integer h, k , l )
Where :
bc
ca
ab
, B 2π
, C 2π
abc
a bc
a bc
and : A a 2 , b 0, A c 0 etc : hence ' reciprocal '
A 2π
(Always use primitive unit cells.)
• The set of G vectors given by all possible integer values of
h,k, and l is known as the reciprocal lattice. The G vectors
are know as reciprocal lattice vectors.
• A periodic function f(r) can then be expressed as the 3D
Fourier series.
iG hkl r
f (r)
C
h , k ,l ,
hkl
e
• Since the dot product of a lattice vector (ua+vb+wc) with a
reciprocal lattice vector Ghkl is 2(uh+vk+wl) – an integer
multiple of 2 - if you move by a lattice vector the phase of
the exponentials remains unchanged giving the same
value for f(r) and the correct periodicity in real space.
8
The Reciprocal Lattice. An
Orthorhombic Example
• Orthorhombic: a ≠ b ≠ c, 90° between axes, a, b, c form a
right handed set.
• Reciprocal lattice vectors:
bc
bc 2
2
a
a,
a bc
abc
a
2π
2π
B
b,
C
c
b
c
A 2π
• View structure down ‘c’ axis:
Reciprocal
Space Lattice
Real Space Lattice
b
a
B=2/b
A=2/a
• If the angles between the a,b, and c axes are not 90° then
a axis in real space will not necessarily be parallel to the A
axis in reciprocal space. (See hexagonal example later.)
9
The Reciprocal Lattice and
Miller Index Planes
• The first plane (after the plane going through the origin)
with a Miller index (h, k, l) goes through the points:
a , b , c
h
k
l
• The normal to this plane is parallel to the cross product of
two vectors in this plane, and hence to Ghkl :
z
c/
Ghkl
l
a h b k a h c l hkl1 hb c kc a la b
a bc
hA kB lC
hkl
G hkl
y
b/
k
a/
h
x
• For a plane wave, wavevector Ghkl, the difference in phase
between a point on the plane that goes through the origin,
and a point in the plane shown in the diagram above is:
G hkl a hA kB lC a 2
h
h
(The phase difference between two points separation r is
k.r. Ghkl is perpendicular to the planes and so any vector, r
joining a pair of points, one in each plane, will do.)
• Thus the set of planes with Miller indices (h,k,l) are
perpendicular Ghkl, and the phase of a wave, wavevector
Ghkl changes by 2 between one plane and the next. The
set of planes have the same spacing, therefore, as
wavefronts of the wave with wavevector Ghkl
10
Reciprocal Lattice and Miller Index
Planes: Orthorhombic Example
• Examples of Miller indices and G vectors
Real Space
3rd index undefined as
we are looking in 2D
Reciprocal
Space
(01•) planes
G01•
b
B
a
A
G000
Reciprocal
Space
Real Space
(02•) planes
G02•
b
B
a
A
11
Reciprocal Lattice: Orthorhombic
and Hexagonal Examples
Reciprocal
Space
Real Space
(12•) planes
G12•
b
B
a
Real Space
A
Reciprocal
Space
(10•) planes
G000
B
b
A
a
G10•
Note:[1] G vector perpendicular to planes, and of length
inversely proportional to the plane spacing.
[2] If the lines (=planes in 3D) drawn in the figure were ‘wave
crests’ then the wavevector of that wave would be the
associated G vector
12
X-ray and Neutron Diffraction
• The diffraction of x-rays and neutrons from a solid is used
to study structure.
• The phase of a wave changes by k.r over distance r
• The condition for diffraction from a crystal relates to the
scattering wave vector ks, which is the difference:
ks = kf - ki between the wavevectors of the outgoing (kf)
and incoming (ki) beams.
Scattering objects
Extra phase ki.r
incoming wave
wavevector ki
phase difference
(kf –ki).r=ks.r
r
Extra phase kf.r
• If the scattering wavevector is equal to a reciprocal lattice
vector then since the product of a lattice vector with a
reciprocal lattice vector is an integer multiple of 2, all
equivalent points within the crystal (e.g. all identically
located atoms) will scatter in phase and give a strong
outgoing beam. i.e. the diffraction condition is:
and:
ks = Ghkl
kf = ki + Ghkl
13
X-ray and Neutron Diffraction:
Energy Conservation
• Conservation of energy requires that the incoming and
outgoing wavevectors (once the scattering particle is free
of the crystal) must be of equal magnitude.
• Condition for diffraction neatly represented by Ewald’s
sphere construction: both ki and kf must lie on the surface
of a sphere, and be separated by a G vector.
Ewald’s
Construction
kf
G
ki
• It is clear that it is quite possible that for a particular
incident condition there is no diffraction from a crystal –
the Ewald construction is quite specific on ki and kf .
• Diffraction from powders overcomes this by having a large
number of crystals in different orientations.
14
Strong Scattering of Waves in
Crystals
•
Neutrons (provided the sample is typically thinner than 1cm) and
x-rays are scattered at most once as they pass through a crystal.
•
If you try to send a beam of electrons through a crystal it is very
strongly scattered – the mean free path depends on energy, but
takes a minimum at 50-100eV of about 6Å in a typical metal.
•
If you imagine starting a beam of electrons inside a crystal with a
particular wave vector k, it will quickly be scattered into a set of
waves travelling with wavevectors k + Ghkl .
•
There will then be more scattering, but now, since diffraction
simply adds a G vector to the intial wave vector, it will be from
one of these new set of waves to another – indeed some may be
scattered back into the original wave with wave vector k.
•
After a while a sort of equilibrium is reached with the rate of
scattering out of a particular set of waves equalling the rate of
scattering into it. Once this has happened, no further effect of the
scattering can be seen, and this explains why despite the large
scattering cross sections, as we shall see later, electrons can
behave as if they move through a crystal unimpeded.
•
We shall see later that because electrons moving through a
crystal with a certain wave vector (k) can in fact have some of
their ‘probability amplitude’ in whole set of associated waves
(wavevectors k + Ghkl ), one may have to allow for this by making
a correction to the ‘effective mass’ that they seem to have.
15
Condensed Matter Physics:
Phonons
• Aims:
• Lattice vibrations ‘normal modes’/’phonons’
• Establish concepts by considering modes of a 1dimensional, harmonic chain, both monatomic and
diatomic.
• Examples of phonons in a 3D lattice.
• Debye theory of heat capacity
• 3kB/atom at ‘high’ temperatures.
a T3 at low temperatures.
• What are high/low temperatures,
concept of Debye temperature.
• Thermal Conductivity of insulating crystals
a T3 at low temperatures.
a T-1 at high temperatures.
• Strong effect of defects and
specimen dimensions at low
temperatures.
Thermal Conductivity of Ge v.
Temperature
Thermal
Conductivity/W/cmK
100
10
1
1
10
100
1000
0.1
Temperature/K
16
Atomic Motion in a Lattice
• In a solid, the motion of every atom is coupled to that of its
neighbours – so cannot describe motion atom by atom –
use ‘normal mode’ approach instead.
• The motion of a ‘harmonic’ system (objects connected by
‘Hook’s law’ springs), can be described as a sum of
independent ‘normal modes’ in which the coordinates all
oscillate at same frequency and maintain fixed ratios to
each other.
• Motion of atoms must be described quantum
mechanically, but we will use the results that:
• The displacement patterns of the classical normal modes are
the same as the ratios of the coordinates in the quantum
mechanical ones.
• The energy of the quantum mechanical modes is expressed
in terms of the frequency (w) of the classical mode:
1
E w n
2
• In a solid these quantised normal modes are called
phonons.
17
Normal Modes - Classical
View: 2 Coordinate Example
k
k
k
m
m
x1
x2
Spring tensions, left to right : F1 kx1 , F2 k x2 x1 , F3 kx2
Equations of motion : mx1 F2 F1 2kx1 kx2
mx2 F3 F2 kx1 2kx2
(1)
(2)
mx1 x2 k x1 x2 mu1 ku1
Subtacting (1) (2) : mx1 x2 3k x1 x2 mu2 3ku2
Adding (1) (2) :
1/√2 for normalisation
u1 1
x1 x2 and
u2 1
x1 x2 are the normal coordinate s
2
2
with independen t equations of motion, solutions :
u1 A1 cosw1t 1 , u2 A2 cosw2t 2
where w1 k
, w2 3k and A1, A2 are independen t amplitudes .
m
m
In mode 1, u2 0 so x1 x2 .
• Mode 1:
In mode 2, u1 0 so x1 x2 .
Mode 2:
x1
1 1
1 1
u1
u2
x
1
2
2
1
2
1 1
1 1
cosw1t 1 A2
cosw2t 2
A1
1
2
2 1
18
• General solution.
Normal Modes - Quantum
View: 2 Coordinate Example
Schroeding er' s equation :
2 2 x1 , x2 2 2 x1 , x2
V x1 , x2 x1 , x2 E x1 , x2
2
2
2m
2
m
x1
x2
where : V x1 , x2
1 2 1
1 2
2
kx1 k x2 x1 kx2
2
2
2
however V x1 , x2 can be written as V u1 , u2
1 2 1
2
ku1 3ku2
2
2
i.e. Schroeding er' s equation separates in u1 , u2 coordinate s :
2 2 u1 , u2 1 2
2 2 u1 , u2 1
2
ku
u
,
u
3
ku
u1 , u2
1
1
2
2
2
2
2m
2
2m
2
u1
u2
and we can write u1 , u2 1 u1 2 u2 and :
1 2 2 1 u1 1 2
ku
u
1
1 1
1 u1 2m u12
2
E u1 , u2
Each term has only one variable
but their sum is constant, so each
must be constant giving two
Independent equations
1 2 2 2 u2 1
2
3
ku
u
2
2
2 E1 E2 E
2
2 u2 2m u2
2
Where the standard SHO solution applies and :
1
1
E1 w1 n1 and E2 w2 n2
2
2
• The frequencies and amplitude ratios are the same as
for the classical case, but the energy is quantized
19
Lattice vibrations
• 1-D harmonic chain
• Take identical masses, m, separation a connected by
springs (spring constant, a):
• This is a model limited to “nearest-neighbour” interactions.
Equation of motion for the nth atom is:
mun a un 1 un un un 1
mun a un 1 un 1 2un
• We have N coupled equations (for N atoms).
• Take cyclic boundary conditions – N+1th atom equivalent
to first (will be discussed later).
• All masses equivalent – so the normal mode solutions
must reflect this symmetry and all have the same
amplitude (u0) and phase relation to their neighbours, i.e.
un1 un exp i , un1 un exp i
20
Lattice Vibrations: Frequency
of Modes
• Look for normal mode solutions
• Each coordinate has time dependence: exp iwt
• Substitute into equation of motion:
mw 2un exp iwt a ei e i 2un exp iwt
mw 2 a 2 2 cos 4a sin 2
2
w
4a
sin
m
2
• Phase, , only has unique meaning for a range of
2: makes most sense to consider w as a function
of over the range – to , giving:
-
Phase
21
Lattice Vibrations: Nature of
Modes
• Can write the amplitude of the nth atom as:`
un u0 exp in wt
• Can write the phase difference between successive atoms
in terms of a wavevector, conventionally written as q for
phonons:
qa
• Now it is clear that the modes are waves travelling along
the chain of atoms:
na is the distance x along the chain
un u0 exp iqna wt u0 exp iqx wt
• The dispersion relation for these waves is:
4a
qa
sin
m
2
w q
• Since the phase, , only has unique meaning for the range
–/2 to /2, q only has a unique value over the range:
a
q
a
1
E
w
n
• Energy stored in mode is
2
i.e.
a ground state of energy ħw/2
plus: n phonons each of energy ħw.
• Momentum of a phonon turns out to be ħq .
• Velocity = w/q (If you can see the wave move you must
have formed a wavepacket, so velocity is group velocity)
22
The Meaning of phonon
wavevector q.
• The wavevector q gives the phase shift between
successive unit cells.
• q is defined on the range
a
q
a
i.e.
G
G
q
2
2
where G =2/a is the smallest reciprocal lattice vector.
• q has no meaning between lattice points, so is
equivalent to q+G.
q+G
Amplitude
0.8
q
0.3
0
1
2
3
4
5
6
7
8
-0.2
-0.7
-1.2
n
Phase shift between
lattice points is
meaningless
• Remember: a wave vector that is a reciprocal lattice
vector gives a phase shift of 2n between two points
separated by a lattice vector.
• Free space is uniform, so a phase shift along a wave
given by f=kr works for any r. In a crystal, space is not
uniform - equivalent points are separated by a lattice
vector, and f=kr only has meaning if r is a lattice vector.
23
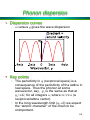
Phonon dispersion
• Dispersion curves
w versus q gives the wave dispersion
• Key points
• The periodicity in q (reciprocal space) is a
consequence of the periodicity of the lattice in
real space. Thus the phonon at some
wavevector, say, q1 is the same as that at
q1+nG, for all integers n, where G=2/a (a
reciprocal lattice vector).
• In the long wavelength limit (q→0) we expect
the “atomic character” of the chain to be
unimportant.
24
Limiting behaviour
• Long wavelength limit
• dispersion formula
4a
qa
w
sin
m
2
q→0
sin qa 2 qa 2
• leads to the continuum
result (see IB waves course)
w q
q 0
aa
ma
;
w
q
Continuum result
Y - Young’s modulus
- density
Y
• These are conventional sound-waves.
• Short wavelength limit
• “Atomic character” is evident as the wavelength
approaches atomic dimensions q→/a. l=2a is
the shortest, possible wavelength.
• Here we have a standing wave w/q=0
wmax 4a m
25
Momentum of a Phonon: ħq
•
Need to extend our concept of momentum to something that
works for phonons – a so called ‘crystal momentum’.
•
If, for example, a neutron hits a crystal and creates a phonon, we
want a definition of phonon momentum such that momentum will
be conserved in the scattering/phonon creation process.
•
For a static lattice we simply have diffraction, and to get a large
scattered intensity all the scatterers have to scatter in phase, i.e.
(kf - ki).r = 2n (r is a lattice vector: separation of identical atoms)
and for this to be true kf - ki = G.
•
If the lattice is now distorted by a phonon, the way each atom
scatters will be modified by an extra phase term, q.r, so, if the
scattered amplitudes are all to add up, the scattering wavevector
will have to give an extra phase difference between lattice
positions of q.r. i.e:
(kf - ki).r = 2n + q.r and
kf - ki = G + q .
•
This means that on scattering the crystal changes momentum by
ħ(G + q). ħG is the momentum transfer due to diffraction from the
lattice causing the whole crystal to recoil, and so it is sensible to
define the momentum of the phonon as ħq – after scattering
either you have created a phonon momentum -ħq or you have
annihilated one of momentum ħq.
•
But you say, you can’t just define momentum anyhow you like –
surely it is something that exists and we have to measure it. Not
at all. Momentum and energy entered physics as constructions
created to make the maths of doing physics easy. Consider
potential energy – to what measurable ‘real’ quantity can you add
an arbitrary offset and everything is still ok? Why is energy
conserved ?– because we carefully define all forms of energy so
that it is, at least that is how the idea started.
26
Momentum of a Phonon: ħq,but is it
reasonable? (non examinable)
•
The problem is that if you really do have a infinite uniform wave in
an infinite lattice, there are as many atoms going forwards as
backwards and it carries no momentum.
•
The total (classical) momentum is carried somehow by all the
atoms in the crystal – what we are trying to do is divide it
notionally between momentum carried by the phonon and that
associated with motion of the centre of mass – so its complicated.
•
However, if you make a wavepacket out of a small spread of
wavevectors, then if you give the wavepacket an energy ħw and
add up the momentum associated with all the vibrating particles
they don’t quite cancel out, but do indeed give ħq.
•
Effectively, after the neutron scatters the whole crystal starts to
move, carrying momentum ħG, and inside the crystal is a
wavepacket of vibrations travelling through the crystal that carries
a net extra momentum of ħq.
•
If you are considering scattering of a neutron, you are not
considering an infinite crystal, so one can reconcile normal
momentum with crystal momentum.
•
To understand the infinite case (non quantum mechanically) –
you have to take the limit of the wavepacket going to infinite
length – which is approaching infinitiy in a different way from
saying that we have uniform oscillations throughout the crystal
and let the crystal size go to infinity, so you get a different result
for the momentum when you go to the limit in a different way.
• If an inifinte lattice has to supply ħG or ħq of momentum it
does not change the state of motion of the lattice (i.e. the
lattice does not start to move) because it has infinite mass.
27
1st Brillouin Zone
• Periodicity: All the physically distinguishable modes
lie within a single span of 2/a.
• First Brillouin Zone (BZ)
• we chose the range of q to lie
within |q| /a. This is the 1st BZ.
1st Brillouin zone
(shaded)
• Number of modes must equal the number of atoms, N,
in the chain and for finite N the allowed q values are
discrete, separation 2/Na (see ‘waves in a box’ later).
• To Summarise:
Each mode (at particular q) is a quantised, simpleharmonic oscillator, E= ħw(n+1/2). Phonons have
particle character – bosons: each mode can have
any number of phonons in it with:
Energy=ħw, Mom.= ħq, Velocity = w/q.
The unique modes lie within the first B.Z..
28
Measurement of Phonons
• Basic principle:
• Need a probe with a momentum and energy
comparable to that of the phonons e.g. thermal
energy neutrons for bulk, and He atoms at surfaces.
X-rays can have correct wavelength, but the energy
is so high it is hard to resolve the small changes
induced by phonon interactions. (At l=1Å, energy is
12.4keV – typical phonon energies are up to 40meV)
• Particle hits the lattice and creates/annihilates
phonons.
• Illuminate sample with a monochromatic beam –
incident wavevector ki
• Energy analyse scattered signal – peaks in signal
correspond to single phonon creation/annihilation
occurring at a particular kf.
• Use of conservation laws
• Energy of phonon (+ = creation, - = annihilation):
2 2
w
ki k 2f
2m
• Crystal momentum conservation for phonon creation:
ki k f q G
• Crystal momentum conservation for phonon
annihilation:
ki q G k f
29
Measurement of Phonons II
• To measure energy of probe can use time of flight
techniques – e.g. helium atom scattering (HAS):
Rotating Disk
Chopper
• Time flight of individual atoms through apparatus – to
determine energy transfer on scattering.
HAS ToF data for Cu(100) surface
Relative Intensity
Single phonon
creation peaks
Elastic peak
Phys. Rev. B
48, 4917, (1993)
Energy Transfer/meV
30
Diatomic lattice
• Technically a lattice with a basis
mA
mB
• proceeding as before. Equations of motion are:
m Au2 n a u2 n 1 u2 n 1 2u2 n
mB u2 n 1 a u2 n 2 u2 n 2u2 n 1
Trial solutions:
u2 n U1 exp i2nqa w t
u2 n 1 U 2 exp i 2n 1 qa w t
substituting gives
m
2
w
2a U1 2a cos qa U 2 0
A
2a cos qa U1 mB w 2 2a U 2 0
homogeneous equations require determinant to
be zero giving a quadratic equation for w2.
w
a
2
m A mB
m
mA mB
mB 4m A mB sin qa
2
A
Two solutions
for each q
2
12
31
Acoustic and Optic modes
• Solutions
• q→0:
• Optic mode (higher frequency)
w
a 2m A mB
m A mB
2a
Effective
mass
• Acoustic mode (lower frequency)
w2
a
m A mB
m A mB
12
m A mB
2
m A mB 1 4
qa
2
m A m B
2aa 2
w
q
m A mB
w2a/mB
w2a/mA
Periodic: all
distinguishable
modes lie in
|q|</2a
32
Displacement patterns
•
Displacements shown as transverse to ease visualisation.
• Acoustic modes: Neighbouring atoms in phase
• Optical modes: Neighbouring atoms out of phase
• Zone-boundary modes
• q=/2a; l=2/q=4a (standing waves)
• Higher energy mode – only light atoms move
• Lower energy mode – only heavier atoms move
33
Origin of
optic and acoustic branches
• Effect of periodicity
• The modes of the diatomic chain can be seen
to arise from those of a monatomic chain.
Diagrammatically:
Monatomic chain,
period a
period in q is /a
for diatomic chain
Modes with q outside new BZ period
‘backfolded’ into new BZ
by adding ± G=/a
Energy
of optical
Acoustic
and and
acoustic
modes
optical
modes
split if alternating
masses different
34
Diatomic chain:
summary
• Acoustic modes:
• correspond to sound-waves in the longwavelength limit. Hence the name.
w→0 as q→0
• Optical modes:
• In the long-wavelength limit, optical modes
interact strongly with electromagnetic radiation
in polar crystals. Hence the name.
• Strong optical absorption is observed.
(Photons annihilated, phonons created.)
w→finite value as q→0
• Optical modes arise from folding back the
dispersion curve as the lattice periodicity is
doubled (halved in q-space).
• Zone boundary:
• All modes are standing waves at the zone
boundary, w/q = 0: a necessary consequence
of the lattice periodicity.
• In a diatomic chain, the frequency-gap
between the acoustic and optical branches
depends on the mass difference. In the limit of
identical masses the gap tends to zero.
35
Phonons in 3-D crystals:
Monatomic lattice
• Example: Neon, an f.c.c. solid:
• Inelastic neutron scattering results in different
crystallographic directions
(00)
()
( 0)
Phys. Rev. B
11, 1681, (1975)
• Many features are explained by our 1-D model:
• Dispersion is sinusoidal (nearest neighbour.
interactions).
• All modes are acoustic (monatomic system)
36
Neon:
a monatomic, f.c.c. solid
• Notes: (continued)
• There are two distinct types of mode:
• Longitudinal (L), with displacements parallel to
the propagation direction,
• These generally have higher energy
• Transverse (T), with displacements
perpendicular to the propagation direction
• These generally have lower energy
• They are often degenerate in high
symmetry directions (not along (0))
• Minor point (demonstrating that real systems are
subtle and interesting, but also complicated):
• L mode along (0) has 2 Fourier components,
suggesting next-n.n. interactions (see Q 3, sheet
1). In fact there are only n.n. interactions
• The effect is due to the
(110)
fcc structure. Nearestneighbour interactions from
atom, A (in plane I) join to
atom C (in plane II) and to
B
C
atom B (in plane III) thus
linking nearest- and
next-nearest-planes.
A
C
I
II
III
37
Phonons in 3-D crystals:
Diatomic lattice
• Example: NaCl, has sodium chloride structure!
• Two interpenetrating f.c.c. lattices
• Main points:
Phys. Rev. 178
1496, (1969)
• The 1-D model gives several insights, as
before. There are:
• Optical and acoustic modes (labels O and A);
• Longitudinal and transverse modes (L and T).
• Dispersion along () is simplest and most like
our 1-D model
• () planes contain, alternately, Na atoms and
Cl atoms (other directions have Na and Cl mixed)
38
NaCl phonons
• Notes, continued…
• Note the energy scale. The highest energy
optical modes are ~8 THz (i.e. approximately
30 meV). Higher phonon energies than in
Neon. The strong, polar bonds in the alkali
halides are stronger and stiffer than the weak,
van-der-Waals bonding in Neon.
• Minor point:
• Modes with same symmetry cannot cross,
hence the avoided crossing between acoustic
and optical modes in (00) and (0) directions.
• Ignore the detail for present purposes
39
Conservation Laws and
Symmetry
•
•
Lagrangian Mechanics
•
Newton 2 normally considered in Cartesian coordinates: ‘F=ma’
•
Can generalise to non-Cartesian coordinates, but now write
equations of motion in terms of derivatives of the Lagrangian:
L=K.E. – P.E. . Field of ‘analytical dynamics’ based on this idea.
Conservation Laws
•
A key result of analytical dynamics is Noether’s theorem – for
every symmetry in the Lagrangian (i.e. in the system), there is an
associated conservation law.
e.g. it turns out that:
• If the system’s behaviour is independent of the time you set it going,
energy is conserved.
• If the system’s behaviour is independent of where it is in space ,
momentum is conserved.
• If the system’s behaviour is independent of the its angular orientation,
angular momentum is conserved.
•
In a crystal – space is no longer uniform but has a new symmetry
– its periodic, so the law of conservation of momentum is
replaced by a new law – the conservation of ‘crystal momentum’
in which momentum is conserved to within a factor of ħG.
E.g.
• Diffraction: wavevector allowed to change by factors of G
• Phonon creation:
k f ki G
ki k f q G
• Adding a G vector to a phonon’s wavevector does not change its
properties, but its crystal momentum changes by ħG
40
A mathematical aside
(for interest - non examinable).
• L=K.E.-P.E
(e.g. S.H.O. L
1 2
mx kx2 )
2
• Equations of motion:
Euler-Lagrange equations:
SHO:
d
mx kx 0
dt
mx kx
d L L
0
dt qi qi
• Conjugate momentum: pi
L
qi
(SHO:
d
pi L 0
dt
qi
L
mx
qi
)
Noether’s
theorem
• If L is independent of qi:
dpi
L
0 and hence
0 and pi does not vary w ith time.
qi
dt
• Energy conservation? Under many circumstances the
Hamiltonian H (defined as
Nf
H qi
L
L
qi
) is the
energy, and since dH L , if L does not have explicit
dt
t
time dependence , H and hence energy is conserved.
i 1
41
The Use of Conservation Laws
• What do conservation laws tell you?
• Conservation laws tell you what is allowed to happen –
it is not possible to have an outcome of an event that
violates a valid conservation law.
• Unless conservation laws permit only one outcome,
they do not tell you what will actually happen, nor how
fast it will happen.
e.g. conservation of crystal momentum inside a
periodic solid tells you what possible outgoing
momenta a diffracted particle may have, (k f k i G )
but they do not tell you how intense the outgoing
beams will be.
42
Thermal Properties of Insulating
Crystals: Heat Capacity
• Thermal energy is stored in the phonons
• Need to know how much energy is stored in
each mode.
• Need to know how many phonon modes there
are.
• Need to sum the thermal energy over all
modes
• Heat capacity is then the derivative of the
thermal energy.
• Energy stored in a phonon ‘normal mode’
• Each mode has an energy E=ħw(n+ ½) where
n is the number of phonons in the mode.
• The factor of ½ is the ‘zero point’ energy – it
cannot be removed. Since thermal energies
are taken to be zero in the ground state, it will
be ignored in this treatment.
• It the solid is in thermal contact with some fixed
temperature ‘reservoir’ then the probability of
the mode having n phonons relative to the
chance of it having none is given by a
Boltzmann factor: Pn= exp(-n ħw/kBT)
43
Energy/normal mode,
continued
• Calculate average energy stored in a particular normal
mode (ith ) by averaging over all possible values of n (0 to
∞).
Ei
nw
n 0
i
exp(
exp(
nw i
nw i
n 0
k BT
k BT
)
nw
n 0
)
i
exp( nw i )
exp( nw )
n 0
where β
1
k BT
i
Denominato r is a geometric series ratio exp( w i ) :
exp( nwi )
n 0
Numerator is
1
1 exp( w i )
of denominato r :
w exp( w i )
n
w
exp(
n
w
)
exp(
n
w
)
i
i
i
2
n 0
n 0
1 exp( w i )
Hence average energy stored in a particular mode is :
Ei
w i exp( w i )
w i
1 exp( w i ) exp( w i
) 1
k BT
Planck’s formula
for a single
oscillator
44
Heat Capacity at High
Temperatures
• Low temperature (kBT<<ħw) limit of energy:
w i
Ei
exp(
w i
k BT
) 1
w i exp(
w i
k BT
)
for small T
• High temperature limit of energy (1>>ħw/ kBT)
w i
Ei
exp(
w i
k BT
) 1
w i
1
w i
k BT
1
k BT
• How many phonon modes?
• If a crystal contains N atoms, you need 3N
coordinates to describe position of all N atoms and so
there will be 3N normal modes.
• Thermal behaviour of whole crystal at high
temperatures:
• Since each mode stores kBT of energy at ‘high’
temperatures, and there are 3N modes, then the total
energy stored at high temperatures is 3NkBT and the
heat capacity for kBT >>ħwi is 3NkB. (Basis of Dulong
and Petit’s law, 1819 – heat capactiy/atomic weight
constant.)
45
Debye Theory: The Aim
• Thermal behaviour of whole crystal at intermediate
and low temperatures.
• At ‘non-high’ temperatures, Ei depends on ħwi so we
need a way of summing the contribution all the modes:
N
w i
Etotal
i 1 exp( w i
) 1
k BT
• The first step is to convert the sum to an integral:
Etotal
w
w
0 exp(
k BT
) 1
g w dw
where g(w)=dN/dw is the ‘density of states and g(w)w
gives the number of phonon states N with energies
lying between w and w+w.
• The actual g(w) is complicated – the Debye theory of
heat capacity works by producing a simplified model for
g(w) so that the integral for Etotal can be performed.
46
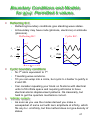
Boundary Conditions and Models
for g(w): Permitted k values.
•
Reflecting B.C.
•
•
Reflecting boundary conditions give standing wave states.
At boundary may have node (photons, electrons) or antinode
(phonons).
Cyclic B.C.
Reflecting B.C.
n=3
n=3
2A
n
n
kn
A
A
n
2n
kn
A
ln
ln
•
n=1
n=2
n=1
A
Cyclic boundary conditions
•
•
•
•
•
n=2
N+1th atom equivalent to 1st.
Travelling wave solutions.
1D you can wrap into a circle, but cyclic b.c.harder to justify in
2 and 3D.
Can consider repeating your block on N atoms with identical
units to fill infinite space and requiring all blocks to have
identical atomic displacement patterns. Ok classically, but
hard to get the quantum mechanics correct.
‘Infinite’ extent:
•
As soon as you use the modes derived you make a
wavepacket of some sort with zero amplitude at infinity, which
fits any b.c. at infinity, but this method does not give density of
states.
47
Debye Model: g(w) for ‘Waves
in a Box’
• For small values of k (long wavelengths) phonons look
like sound waves – with a linear dispersion relation
w=vsk where vs is a mean speed of sound (see
discussion later).
• The Debye model assumes this is true for all
wavelengths – not just long ones – i.e. it ignores the
structure in g(w) due to the atomic nature of the
material.
• g(w) is calculated by assuming that the crystal is a
rectangular box of side lengths A,B,C. We use reflecting
b.c., though cyclic b.c. give same results
• In each dimension the there must be a whole number of
half wavelengths across the box so as to fit the
boundary conditions, i.e. in each direction A=nl/2 and
k=n/A. The total wavevector of the phonon must be:
n
k x
A
n y
B
nz
C
• Volume per state in k space is 3/(ABC) i.e. 3/V where
V is the volume of the box.
•
(k not q is used in this derivation because the idea of waves in a box
applies to many problems in physics – including black body radiation and
the free electron model of a solid, and by convention k is used in these
derivation.)
48
g(w) for ‘Waves in a Box’ II
All states in the shell
have same |k|
• First, work out g(k) from no.
of states, N, that have a
wavevector of magnitude
between k and k +k.These
states lie within the positive
octant of a spherical shell of
radius k and thickness k. (k
+ve for standing waves.)
States uniformly
distributed in
k-space
• For each phonon mode
there are two transverse and
one longitudinal polarisation,
i.e. 3 modes per point in k
space.
3 Polarisations/k state
Volume of shell
1 state “occupies a
volume” (3/ABC)
Vol. of one state
4k 2
N g k k 3
k
8
3
. g k 3 ABCk 2 2 2
ABC
Since N g k k g w w,
g w g k dk
dw
2
3
V
w
w
dk
For a sound wave k
so
1 and g w
3
vs
dw
vs
2 2 vs
(V ABC is the volume of the box)
• For cyclic BC, states 2x as far apart –vol/state = 83/V but
require full shell, not just +ve octant, – net result same g(k).
• Can show (Wigner) results independent of shape of box.
49
Internal Energy in Debye
Model
• Heat capacity follows from differentiating the internal
energy (as usual).
• For the present we will ignore the zero point motion.
• Need to make sure you integrate over the correct number
of modes – use the fact that if there are N atoms in the
crystal (volume V) then there are 3N modes. Debye
suggested simply stopping the integral (at the ‘Debye
frequency’, wD) once 3N modes have been covered, i.e.
wD
wD
Vw D3
3Vw 2
3N g w dw 2 3 dw
2 2 v s3
0
0 2 v s
• Internal energy
U
wD
0
w
exp w kT 1
w D3 6 2 v s3 N / V
g w dw
No. of phonons
in dw at w
Energy per phonon
(Planck formula)
• Hence:
U
wD
0
3Vw 2
1
w
dw
2 3
2 vs
exp w kT 1
3V w D 3
1
2 3 w
dw
0
2 vs
exp w kT 1
(We can now see that the appropriate mean velocity is
1 1 1
2
where vL and vT are the longitudinal and
v s3
3 v L3
vT3
transverse sound wave velocities)
50
Heat Capacity Within the
Debye Model
• Differential U to get heat capacity C:
U
C
3V
2 2 v s3
wD
0
1
dw
exp w k B T 1
w3
U
3V
T 2 2 v s3
wD
0
w
exp w k B T
2
3 kT
w
dw
2
exp w k BT 1
1
9 Nk B
w D3
w D
3 k T
B
0
1
V
k BT
3
2
3
6 Nvs
w
w
exp w k B T
d
2
k B T exp w k B T 1 k B T
4
3
T D T 4 e x
9 Nk B
x
dx
2
0
x
e 1
D
where the Debye temperatu re D is given by w D k B D
C/3NkB
T/D
51
Debye Temperatures
Element
Li
Na
D/K
344 158 91
Element
C
Si
D/K
2230
645 374 200 105
V
K
Ge
Cr
Rb
Cs
56
38
Sn
Mn
Pb
Element
Sc
Ti
Fe
Co
Ni
Cu
Zn
D/K
360
420 380 630 410 470 445 450 343 437
• Debye frequency controlled by mass of atoms and
stiffness of lattice.
• As you go down a group in the periodic table (e.g. Li to
Cs or C to Pb) the mass increases and the atoms
become bigger and more deformable –so the rigidity
goes down.
• Transition metals tend to lie between 200-600K – so
Dulong and Petit’s law works roughly at room
temperature.
• Note very high value for carbon – a light atom and a
very rigid lattice.
52
Heat Capacity at
Low Temperature
• Check high temperature behaviour:
T
C 9 Nk B
D
3
D
T
x
0
for T D
e
T
C 9 Nk B
D
3
ex
1
x
D
0
T
2
4
e
ex
x
1
2
dx
1
1 x 1
2
1
x2
and :
x 2 dx 3 Nk B
• Limiting behaviour as T → 0.
• At low temperature the higher frequency modes are
not excited. Thus contributions to the integral for
large w (>wD) can be ignored and wD replaced by
.
3
T 4 ex
C 9 Nk B x
dx
2
0
x
e 1
D
4/15
3
Integral
=
4
4
T
12
C
Nk B
5
D
C T3
Debye, T3 Law
• Note similarity to heat capacity of a vacuum –
photons in black body radiation.
53
Measured Density of States
• Example: Aluminium (shows common features)
• Measured density of states compared with
Debye approximation.
• Both measured and Debye density of states
are similar at low w, as expected (w q).
• Debye frequency chosen to give same total
number of modes (i.e. equal area under both
curves)
• Largest deviations where phonon modes
approach zone boundary.
• Measured curve is complex because the 3-D
zone has a relatively complicated shape, and
the transverse and longitudinal modes have
different dispersions (as we have seen earlier).
54
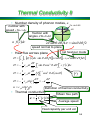
Thermal Conductivity
• Phonons and thermal conductivity
• Phonons are travelling waves that carry energy
and can therefore conduct heat.
• Kinetic theory gives the thermal conductivity
l
z
z = -l cos
• Excess temperature of phonons crossing plane
T
dT
dT
z l cos
dz
dz
• Excess energy in each phonon mode
heat capacity of
a phonon mode
c ph T c ph
dT
l cos
dz
• l = mean free path – phonons assumed to
thermalise at each collision.
55
Thermal Conductivity II
• Number density of phonon modes, n
number with
speed c to c+dc
fraction with
angles to +d
n f c dc
2 sin d
d
2 sin d 4 sin d 2
speed normal to plane
net heat per mode
• Heat flux across plane
H nf c dc sin d 2c cos c phl cos dT dz
0
0
1
dT
2
H c ph nl
sin
cos
d
c f c dc
0
0
2
dz
1
1
dT
H c ph nl
c cos 2 d cos
c
1
2
dz
1
dT
dT
H c ph nl c
3
dz
dz
c ph nl c 3
Definition of thermal conductivity
• Thermal conductivity
1
C cl
3
Mean free path
Average speed
Heat capacity per unit vol
56
Mean Free Path for Phonons.
• Mean free path – limited by scattering processes
• With many scattering processes add scattering rates:
c l c l1 c l2 1 l 1 l1 1 l2
Thus, the shortest mean free path dominates.
• “Geometric” scattering:
• Sample boundaries (only significant for purest samples
at low temperatures).
• Impurities/grain boundaries: l independent of T.
• Phonon-phonon scattering:
• True normal modes do not interact with each other.
• However, in an anharmonic lattice, phonons can scatter.
As a phonon extends/compresses the bonds it changes
the spring constants and one phonon can diffract from
the grating of variable elastic properties produced by
another phonon.
LennardJones 6-12
potential
V(r)
Spring
Const.
r/r0
Mean phonon amplitude
at 20K in Ne is 1% of
mean nearest neighbour
distance and gives
significant changes in
the spring constant.
57
Temperature Dependence of
Thermal Conductivity of Insulators
• Low temperatures:
- few phonons,
geometric scattering
dominates, l constant.
-C and hence a T3.
• High temperatures:
-C constant (3NkB)
- no of phonons a T, so
l a 1/T and a 1/T
Thermal
Conductivity/W/cmK
• In insulators there are no contributions from free electrons.
• In pure crystalline form the conductivity can be very high
(larger than metals e.g. at 70K diamond has
=12000W/m/K, at 300K Copper has =380W/m/K)
• Non-crystalline systems have much lower conductivity l ~
local order e.g. at 300K for glass has l ~ 3Å, and rubber
has l ~ 10-20Å.
• Thermal conductivity shows strong temperature
dependence.
1
Thermal Conductivity of Ge v.
C c l
3
Temperature
100
aT3
10
a1/T
1
1
10
100
1000
0.1
Temperature/K
• Intermediate temperatures – expect conductivity to
be below 1/T and T3 asymptotes, (heat capacity is
dropping and there is both phonon and geometric
scattering) but actually get a large rise for pure,
crystalline samples.
58
Thermal Conductivity at Intermediate
Temperatures:Phonon-Phonon
Scattering and Umklapp Processes.
• A typical phonon-phonon collision process is coalescence:
q3
q2
q1
• However, if the resulting phonon has a momentum that is
simply the addition of the two colliding phonons – the
collision is very ineffective as far as thermal conductivity
goes – you still have the same energy going in roughly the
same direction.
• To really change the direction energy is flowing – the sum of
the momenta of the incoming phonons must be outside the
first Brillouin zone – so that you get an ‘umklapp’ (german
for ‘fold around’) i.e. the new phonon momentum has a G
vector subtracted from the sum of the old ones, which
changes significantly the direction of travel.
• As the temperature drops
fewer phonons have enough
energy to have enough
momentum to give a resulting
phonon that is produced by an
umklapp process and so the
mean free path and hence
rise dramatically
G
q3
Umkapp
q2
q1
Process
59
Umklapp Processes – a 1D
example
1st Brillouin Zone
q3= q1+ q2
q3-G
L
T
q2
q1
-G/2
Direction of travel
(group velocity)
G/2
Phonons with enough q for
umklapp poorly excited at
intermediate temperatures
• Direction of travel determined by group velocity dw/dq
• To change direction you need to have enough momentum
in the incoming phonons, q1+q2 so that q3 is outside the
first Brillouin zone, so that its group velocity has a direction
very different from q1 and q2.
• By convention if q3 is outside the first B.Z. a G vector is
subtracted to show it in the first B.Z.- hence the ‘umklapp’.
60
Condensed Matter Physics:
Free Electron Model
• Aims:
• Assumptions of the free electron model
• What states do the electrons occupy – the
controlling fact is that electrons are fermions.
• Success of free electron model:
• Heat capacity of a free electron gas
• Bulk modulus
• Electrical and thermal conductivity
‘In a theory which has given results like these, there must
certainly be a great deal of truth’
H.A. Lorentz
• The limits of the free electron model.
• Hall Effect - evidence for conduction by
positively charged ‘holes’.
• Does not explain differences between
conductors/semiconductors/insulators, let alone
properties of semiconductors.
61
Assumptions of Free Electron
Model
• All ‘valence’ electrons are free to move.
• Atomic structure consists of full ‘shells’ or atoms +
more loosely bound shells partially occupied by
‘valence’ electrons – so called because they
participate in chemical bonding.
• The FE theory assumes these valence electrons are
free to move and explain the electrical conductivity
of a metal.
• Sodium, magnesium and aluminium assumed to have
1,2 and 3 free electrons/atom respectively
• Positively charged ion cores are left behind.
• The charge from the positive ions is represented
as a uniform positive background.
• Electron-Electron repulsion ignored.
• The electrons are treated as independent particles.
• There are a range of rather subtle reasons why
these assumptions work so well…
• Cyclic boundary conditions.
• Best examples: Alkali metals (Li, Na, K, …); also
noble metals (Cu, Ag, Au)
62
What States Do the Electrons
Occupy?
• Cyclic boundary conditions used as we want to consider
travelling electrons.
• Basic density of states in k space as before, but electrons
have 2 spin states giving two states per permitted point in
k space.
Volume of shell
Degeneracy 2
Spin up or down
Vol. of one state [=(2/A)3]
8 3
. g k Vk 2 2
N g k k 2 4k k
V
2
• Since thermodynamics is dominated by the energy of
the states – we will need g().
dN dN dk
dk
g
g k
d
dk d
d
2k 2
d 2 k
Since
,
2m
dk
m
3
m
Vkm
V 2m 2 1 2
g 2 . 2 2 2
k
2 2 2
Vk
2
g
1
2
63
Concept of Fermi Level:
Electron Distribution at T=0
• Electrons are fermions and obey Pauli exclusion
principal – only one electron allowed per state. (2 spin
states per k state – already included in g().)
• At T = 0 electrons occupy lowest possible states and
states fill up to the Fermi energy, F. (Na: 3.2eV, Cu: 7eV)
Fermi energy
•
All states within a sphere of radius kF, the Fermi
wavevector, are filled.
• Determine kF and F from the requirement that we have
N electrons (i.e. N filled states)
2 spin states/k state
4 3
k f
N 2. 3
Volume of sphere
Volume/k state
8 / V
3
k 3f 3 2 N 3 2 n
V
2
2 2 2
F
kF
3 2 n 3
2m
2m
where n N
V
64
The Fermi-Dirac Distribution: a
Thermodynamical Aside (non
examinable, but must know result)
• What is distribution of electrons for T>0?
• First law of thermodynamics:
dU dq dW
• Use state functions and add particle exchange:
dU TdS PdV dN
U
N
S ,V
chemical
potential
• At const. V (keeps energy levels and g() the same).
dS dU T dN T
• Consider a state in equilibrium with a reservoir of particles
and heat. In the ground state (electron state unoccupied)
entropy of the reservoir is S0=kBln(W0) where W0 is the
number of different reservoir configurations.
• To occupy the electron state, 1 electron and energy must
be transferred from the reservoir, whose entropy now is:
S 0 dS S 0 T T k B ln W
ln W ln W 0 ln W W 0 k B T
• Rel. chance of state being occupied: W/W0=exp[ -(-)/kBT]
• Normalise to obtain probability of state being occupied:
exp k BT
1
p
1 exp k BT exp k BT 1
Fermi-Dirac Distribution
65
p
1
exp k B T 1
• At T = 0.
• Low temperature limit of the Fermi-Dirac distribution
(also known as the Fermi function) is a step function:
• States fill up
to energy
starting at
= 0. All
higher energy
states are empty.
• For T ≈ 0
• At low T and for < , (-)/kBT is large and negative,
so exp[(-)/kBT] is v. small and FD distribution 1.
• At low T for > , (-)/kBT is large and positive, so
exp[(-)/kBT] is v. large and FD distribution 0.
• In thermodynamics, the value of (T) at T = 0K is
known as the Fermi energy, F.
• In semiconductor textbooks the value of (T) at any
temperature is called the Fermi energy.
• For T > 0
66
Chemical Potential,
(T), for T>0
• Occupied states (F.D. dist. x g() )
• Chemical potential (T) for T>0K.
• Require total number of electrons to be N:
g
N
d
0 exp k T 1
B
~ constant
an implicit equation for (T)
• Solve numerically:
key point – at low T, g()
changes very little within
kBT of , so (T) ≈ (0)
at low T. (True for most
practical situations – see
Q2 on 2nd question sheet.
67
Thermal properties of metals
• Thermal capacity
• Electronic contribution to the heat capacity
follows from differentiating the electronic
energy w.r.t. T.
g
U el
d Result from a straight0 exp k T 1
B
C el
U el
T
Nk B
T
2
TF
2
forward, if lengthy,
manipulation. See
Ashcroft and
Mermin p42-7
(TF = F/kB = ‘Fermi temperature’)
• A similar result follows from a qualitative
argument – emphasising the essential physics
• Compare occupied states at T=0K with those at
T0K
68
Electronic Contribution to the
Heat Capacity of a Metal
• cont…..
• only electrons within ~kBT are active thermally.
• There are nex g F kT such electrons
• If we treat these excited electrons like classical electrons.
That is, having kinetic energy 3kBT/2 per electron.
U el nex 3k BT 2 g F 3k B2 T 2 2
A
Cel
• Recall:
U el
g F 3k B2 T
T
g F
3
V 2m 2 1 2
F
2 2
2
at T 0K N
B
1F 2
εF
εF
0
0
g d K
12
K 1F 2
2 32
d K F
3
g F 3N 2 F 3N 2k BTF
• Combining A and B gives
C el
3N
T
3k B2T 4.5 Nk B
2kTF
TF
Note T
dependence
• Even more simply: fraction of electrons excited T/TF,
energy gained per electron 3/2 kT, change in U
2/3kT2/TF, so Cel 3kT/TF
69
Electronic Heat Capacity
cont…..
• Absolute magnitude is much less than the
classical result (3NkB/2). T/TF~10-2 at room
temperature for typical metals. (The original freeelectron model (Drude model) assumed the
electrons were a classical perfect gas - one of its
many failures was that the heat capacity was far too
large.)
• Observed values are in good agreement. E.g.
Sodium (Na) a “classic”, free-electron metal:
(Cel)meas = 1.5T mJ mol-1 K-1
(Cel)f.e. = 1.1T mJ mol-1 K-1
• T dependence arises because, as the Fermiedge broadens, more electrons get excited.
• N.B. the electronic contribution to the heat
capacity of a metal is masked by a much
larger contribution from vibration at all but the
lowest temperatures. (See Q3 on second
question sheet.)
70
Deviations from Theoretical
Heat Capacity: Effective Mass
2
2
T 2
2
2
3
Cel
NkB
NkB T
3 n NTmn 3
2mkB
2
TF
2
2
(Valid at low T, ≈ arises from assumption that isT indep.)
• Seen only at low T, otherwise masked by phonons.
• At low temperatures: C = gT+T3, so plot:
C/T v. T2 for Potassium
• For K: gTheoretical =1.67mJmol-1K-2 gexpt =2.08mJmol-1K-2.
• In free electron model, C el NTmn 2 3. Discrepancy arises
because electrons move with an ‘effective mass’ m*.
• For ‘nearly free electron metals’ main contribution: ions
around an electron pushed around by moving electron,
(‘electron-phonon coupling’) and the other electrons tend
to avoid the first electron, => extra contribution to the K.E.
of the system included in the FE model by increasing the
effective mass of the moving electron. For ‘nearly free
electron metals’ m*/m somewhat greater than 1: e.g.
K: m*/m=1.25
Mg: m*/m=1.3:
Al m*/m=1.48.
• Other types of deviation:
• Strong effects of the periodic potential of the ion lattice can
get larger deviations, or e.g. for Zn and Cd m*/m <1
• Strong electron-electron correlations can give huge values
(many 1000’s) for m*/m.
71
Pressure Due to Electron Gas
• Consider the energy levels in a
box under compression. When
n
the box is compressed the
4
single-particle wavelengths
shorten and the K.E. rises.
3
2
Hence there must be an
1
outwards pressure.
n
3
2
1
a = l/2
a=l
• At T = 0, compressing the gas, we have ΔU = PΔV and hence: P U so we need U.
V
• Calculate <U>, the mean energy of an electron
F
F
U g d
g d
0
F
32
0
d
2 52
F
5
A
F
0
1 2 d
2 32 3
F F
3
5
F N V 2 3
• for N electrons: U N U 3N F 5
P
0
U F
3 2
N F
F V
5 3V
P 2n F 5 N V
53
72
Bulk modulus of metals
• Electron pressure in metals
• The pressure of the degenerate electron gas also
contributes to the mechanical properties of
metals.
• Isothermal bulk modulus, KT is defined as
p
F N V 2 3
KT V
V T
Eq. A, previous slide
2N
p
2 N F
P
F
5V
V
3 V2
Electron K.E. contribution
2 N F
KT
to the bulk modulus
3 V
• Calculated values are of the right magnitude. We
have neglected the attractive forces, due to the
ion cores. Attractive forces make the metal more
compressible (Experimental bulk modulus, Kexp, is
usually smaller than KT, above).
n=(N/V)/m-3
EF/eV
Kexp/KT
Li
4.6E+28
4.7
0.63
Na
2.5E+28
3.1
0.83
K
1.3E+28
2.0
1.03
Metal
73
Transport Properties of Electrons:
Representation as Wavepackets
• When considering a particle you usually want to
consider a object that is located to some degree
in space – combine fractions of basic k states to
make a wave packet.
• What about Pauli exclusion principle?
• Start with N k states, and just as with normal
modes, you can make N new states out of them.
• The quantum mechanics does not change – its just
a matter of which functions you chose a basis set to
visualise/analyse what is going one, and some
pictures are more helpful.
Separation
2/A
Start with N normal 1D k states
Make a new single electron state
as a wavepacket of k states
You can make N new states, each
by Pauli E.P. taking 1 electron
74
Dynamics of Wavepackets
•
dw
v
Electron velocity given by group velocity:
dk
• Its just as well that we are not using the phase velocity since
in Q-mech, w has an arbitrary offset making phase velocity
w/k meaningless.(only changes in w have meaning).
• q of a phonon has a similar arbitrary offset- q is equivalent to
q+G.
• Effect of a force on a wavepacket?
Work done by a force f in a time t :
fvt
If the electrons energy increases by its momenum also
changes and k can be worked out from :
d
dw
k
k vk
dk
dk
So
fvt vk
dk
f
dt
• For an electron in a particular ‘k’ state, the force causes k
to increase continuously with time – not easy to visualise
with discrete k states – it would have to jump every now
and again from one state to another-so consider the
electron as a wave packet in k space. It moves
continuously across the array of available k states:
can be written :
k k
t 0
ft
75
Effective Mass, m*, of a
Wavepacket
• The velocity of a wavepacket is determined by the
dispersion relation w(k): v
dw
dk
Rate of change of velocity time :
dv d dw d 2w dk 1 d 2 dk
2
dt dt dk dk dt dk 2 dt
A
But we would like to think of this in terms of an effective mass :
dv
m*
f
dt
dk
Since :
f
dt
dv
dk
B
we have
*
and equating A and B gives
dt m dt
dk 1 d 2 dk
m * dt dk 2 dt
2
m*
d 2
dk 2
• The motion of the electron wavepacket obeys the
classical equations of motion for a particle of mass m* makes considering the motion mathematically much
easier. (The caveat is that the wavepacket must be
significantly larger than the lattice spacing)
76
Equation of Motion for the
Electrons
• If no collisions occur then the force on the electron is the
Lorentz force and:
dv
f m*
eE ev B
dt
• Need to allow for collisions – use idea that if the mean
time to the next collision is t, then the probability of the
electron surviving a time t without a collision is exp(-t/ t).
After the collision the velocity is randomised, so its
average value is 0 and hence after a time t the average
velocity is:
<v>=v. exp(-t/t) +0.(1- exp(-t/t) ) = v. exp(-t/t)
i.e. the collisions induce an exponential decay of the mean
velocity and their effect may be written:
d v
dt
v
1
v exp t t
t
t
• Since we are looking at the movement of wavepackets,
any velocity we use is an average velocity and so the
effect of collisions looks like a velocity dependent friction
and may be included in the equation of motion by adding
in an extra term to the wavepacket acceleration:
dv
1
v
* eE ev B
dt m
t
dv v
77
m*
eE ev B
dt t
Electrical Conductivity
• All the electrons obey the same equation of motion,
and so we can work out their average velocity, the drift
velocity, in the presence of a constant electric field:
v
m* drift eE
τ
• Electron mobility, , is defined as:
vdrift
eτ
E
m*
• Since the current density (j) is given by the product of
the number of electrons per unit volume (n), their
charge and drift velocity we have:
j n e v drift
ne 2t
*
E neE
m
• Hence Ohm’s law, with conductivity:
ne 2t
m
*
ne
• As for phonons, collisions can occur either with defects
in the crystal, or with phonons, and since we add
collision rates:
1
1
1
t
t phonon
t defect
• Electron-electron ‘collisions’ have to be handled by a
much more sophisticated theory that deals with
electron-electron correlated behaviour.
78
Electron Scattering and
Resistivity
• When an electric field is applied, all the electrons in the
Fermi sphere start to move continuously though k space in
the –E direction at a rate:
dk
1
eE
dt
i.e. the whole Fermi sphere moves continuously together
in k space.
• Phonons have a momentum (wavevector) comparable to
that of electrons, but their energies are typically up to
40meV, tiny compared to F, so phonon scattering can
change the direction of an electrons wavevector strongly,
but only change slightly the magnitude of the wavevector.
• Similarly scattering from defects does not produce large
changes in energy since again only phonons could pick up
energy changes.
• There must be an appropriate empty state available for
scattering to occur. Since there are no free states within
the Fermi sphere, only electrons near the Fermi surface
can be scattered.
79
Electron Scattering and Drift
Velocity
• The majority of electrons that can be scattered lie at
the ‘front’ of the moving Fermi sphere, and the
scattering process results in them being transferred to
the ‘back’ of the moving sphere and so equilibrium is
reached with the Fermi Sphere shifted in k space – i.e.
with the electrons having a net drift velocity.
• The scattering rate, t, used on overheads 77 and 78 is
therefore an average rate – when the electrons are in
the middle of the Fermi sphere they are not scattered
at all, and are then scattered strongly when the arrive a
the ‘front’ of the moving sphere.
Electrons ‘thermalise’
back to Fermi sphere
by more phonon
collisions
ky
phonon
scattering
kx
Fermi sphere shifted
*
by k m v drift
E
Blurred edge of
Fermi sphere
80
Temperature Dependence of
Resistivity
1
t
1
t phonon
1
t defect
• Scattering from defects is temperature independent
• At high temperatures, all phonon states are populated
and the number density of phonons is proportional to
temperature. Since tphonon << tdefect, 1/t and hence
the resistivity are proportional to temperature => use of
resistance thermometers (usually Pt).
e.g. for a ‘Pt100’ resistance thermometer:
Temp/K 136.5
273
546
Res./W 54.8
100
202.4
• Different samples have different defect densities, so
have resistivity v. temperature curves related to each
other by a fixed offset – Matthiessen’s rule.
Resistivity ->
Sample A
Offset temp.
independent
Sample B
81
Temperature ->
82
Thermal Conductivity
1
C c l
3
• Same basic formula as for phonons. Use values for heat
capacity of the free electrons, and since only electrons
near the Fermi level are scattered, use <c>=vF and
l = vFt,
12
T
12
T
nk B
vF l
nk B * 2
3 2
TF
3 2
m vF
v t
2
F
2 nk B2Tt
3m *
2k B
• Normal pure metals have a thermal conductivity about
100x that of insulators at room temperature – electron
conductivity swamps phonon conductivity.
• At room temperature and above (‘high’ temperatures)
phonons dominate scattering, and so t a 1/T and
thermal conductivity roughly constant with temperature.
ky
Phonon
scattering to
thermalise
distribution
kx
Electrons in +ve direction
hotter than in –ve direction –
so Fermi sphere more
blurred on right hand side
83
Wiedemann-Franz Law
• Experimental Law
• ‘For metals at not too low temperatures the ratio of the
thermal to electrical conductivities is directly proportional
to temperature with the same constant of proportionality
for all metals’.
• Points to the same basic mechanism for the two
conductivities.
• For free electron model:
2
2 k B2Tnt 3m* 2 k B
T
2
*
3 e
ne t m
• The Lorenz number, L, is defined as L
T
• Theoretical value for L is 2.45x10-8 WW/K2.
• Experimental values:
Element
Ag
Au
Cd
Cu
Ir
Mo
Pb
Pt
Sn
W
L/
10-8WW/K2
2.31
2.35
2.42
2.23
2.49
2.61
2.47
2.51
2.52
3.04
• Good agreement between experimental values and
theoretical values except at low temperatures where the
the different phonons required for scattering in the two
conductivities have different probabilities of excitation.
• Good agreement gives strong support to free electron
model.
84
Hall Effect: Derivation
• B field applied perpendicular to a conductor – electrons
pushed sideways until charges build up on opposite faces
that produce an electric field that stops any further lateral
Build up of positive
motion.
charge on top side
y
+
EH
B
z
+
+
+ + +
- -
+
+
+
+
v
- - -
j
x
-ev x B
Build up of negative
charge on lower side
Balanced by force
from Hall voltage
• Lorentz force zero at equilibrium:
dv
f m* drift eE H ev drift B 0
dt
• Since j n ev drift then E B v drift
• Hall Coefficient:
RH
1
B j RH B j
ne
1
ne
85
Hall Effect: Experimental
Values
• Hall Coefficient: R H
(n = no. density for electrons)
1
ne
• Look at ratio of measured Hall coefficients to value
expected for 1 electron /atom, i.e.
n
1
natom
natom eRH
where natom is the number of atoms/unit volume.
• Some values of n/natom look reasonable (ish)…
Na: 0.9 K: 1.1 Mg: 1.5 Al:3.5
• and some certainly do not…..
Be: -0.2
Cd: -2.2
!!!
THE CHARGE CARRIERS HAVE
THE WRONG CHARGE SIGN
86
Nearly Free Electron Theory:
Bloch’s Theorem
• Now let’s consider electrons in a periodic environment.
• The translational symmetry of the lattice means that the
electron probability density |2| must have the same
periodicity as the lattice – i.e. if you move a lattice
vector it must remain unchanged.
• The amplitude of the wave function can therefore only
change by a phase factor . e.g. in 1D, for a unit cell
length a we have.
i
( x a) x e
and :
( x na) x ein
• As before with phonon’s we can write = ka where k
only has distinct values within the first Brillouin zone,
and we have:
ika
( x a) x e
• Given that
x has this form, we can write :
x uk ( x)eikx
where uk x is now a function of periodicit y a.
• Hence Bloch’s theorem:
The solutions of the Schrödinger equation in a periodic
potential must be of the form:
k r uk (r)eik.r
where uk(r) has the periodicity of the potential.
87
Matrix Representation (Q.mech
H09, pg 345-363)
•
(x) can be represented as a sum of contributions from each of a
set of basis functions (e.g. as a Fourier transform). The idea is
similar to the ‘normal mode’ approach where instead of using the
displacement of each atom, it turns out to be easier to work with
normal modes.
•
In quantum mechanics – the eigenfunctions of any operator form
a complete set of functions that can be used as a basis set to
represent the wavefunction when you are considering the effect
of that operator.
•
This approach is particularly useful when the wavefunction is
dominated by only a couple of the basis functions.
•
With this representation of the wavefunction, matrix methods can
be used to represent, for example, the Schrödinger equation.
•
e.g. in the present case (and now working in 1D to illustrate
theory) uk(x) is periodic and can be written:
uk ( x)
n -
1 inG1x
e
A
(where G1
2π
)
a
A = length of crystal
and k(x):
k x u k ( x )e
Ck,n
ikx
Ck,n
n -
1 i ( k nG1 ) x
e
Ck,n fk ,n
A
n -
88
Matrix Representation of the
Schrödinger Equation
•
The time independent Schrödinger equation is then:
Hˆ x x
Hˆ
Ck,m fk ,m
Ck,mHˆ fk ,m
m -
Ck,p fk , p
m -
p -
Ck,p fk , p
p -
premultipl ying by fk ,n gives :
Ck,m fk ,n Hˆ fk ,m
m -
Inp
Ck,p fk ,n fk , p
p -
Hnm
Which can be written as an eigenvecto r equation :
H nmCk,m Ck,n
i.e.
HC C
m -
H I C 0
and the eigenvecto rs of H give the coefficien ts Ck,n
of the basis functions in solutions of the Schoedinge r
89
equation.
Nearly Free Electon Model in
1D
• Nearly free electron model:
• Ions represented by a periodic potential.
• If we assume that the potential is weak we will see that we
only need include states of comparable energy.
• Electron-electron interaction/correlations/exchange all
ignored.
• To illustrate the phenomenology, consider the 1D case.
Take origin of potential at an ion location:
a
V(x)
V x V p cos pG1 x where : G1
p
2
a
90
Values of Matrix Elements
• Look at values of matrix elements:
A
fk ,n Hˆ fk ,m
0
1 i mG k x
1 i nG1 k x 2 2
1
e
x
V
e
A
2m x 2
A
e
1 inG1x 2 mG1 k 2 imG1x
dx
e
e
2me
A0
A
A
1 i nG1 k x V p ipG1x ipG1x i mG1 k x
dx
e
e
e
e
2
A0
p
Vp
2 mG1 k 2
n,m p n,m p
n ,m p
2
2me
• For simplicity taking only first term in V(x), the matrix eqn is:
.
.
. E k ,- 2
0 V / 2
1
0
0
0
0
0
0
0
0
Ck , 2
Ck , 1
Ck ,0 k
Ck ,1
Ck , 2
.
Ck , 2
Ck ,1
Ck ,0
Ck ,1
Ck , 2
.
K.E. of a free electron of wavevecto r nG1 k
91
0
V1 / 2
Ek , 1
V1 / 2
0
0
0
where : Ek ,n
0
0
0
0
0
V1 / 2
0
0
0
Ek ,0 V1 / 2
0
0
V1 / 2 Ek ,1 V1 / 2 0
0 V1 / 2 Ek , 2 .
0
0
.
.
0
0
0.
.
.
2 nG1 k 2
2me
Choose Significant States
• So:
• If we are looking for a solution to the S.E. that has a
wavevector k, from Bloch’s theorem it must be in the form:
k x u k ( x )e
ikx
C
n -
k,n
1 i ( k nG1 ) x
e
C k,n fk ,n
A
n -
• If we take the free electron case, V(x)=0, then the solution
is simply the n=0 term: 1 ikx
A
e
fk , 0
• If we then add in a small V(x), then the n=0 term will
remain dominant, but now there will be small contributions
(coefficients Ck,n ) from the other basis states.
• The larger the energy difference between the nth state and
the main, n=0 state, the smaller Ck,n (See Qmech handout
9, p357-9). Consider n=1 row of the matrix eqn. on OH 91:
V1
V1
Ck , 0 Ek ,1Ck ,1 Ck , 2 k Ck ,1
2
2
V1
V
Ck , 0 Ek ,1 k Ck ,1 1 Ck , 2 0
2
2
Small
Ignore
Large
V1
Ck , 0
Ck ,1 2
k Ek ,1
Small
• For small potentials, we need only consider the
contribution form the basis state with the energy closest to
that of the main, n=0, component state.
92
Choose Significant States
• K values of possible states iin 1D:
Ek
Free electron
dispersion curve
k
G1
A
k
• For small potentials, only states |fk,n> with energy close to
|fk,0 > will have significant contributions to the overall
wavefunction (See Qmech. handouts p357-9).
• Include state with the nearest energy (A).
Ek
G1
A
k
k
• For small k this state is also very different in energy – so
contributes very little – have a plane wave state k with an
energy very close to free particle energy.
93
Behaviour Near Zone
Boundary I
• Two states now
comparable in energy –
significant contribution
from both
G1
Ek
k
• At k=G1/2, both states
equivalent , and have
equal contribution,
giving two standing
wave contributions:
k
2
φG1
2
φG1 2
x
1
φG1
2
x
1
φG1
2
2
cos a x
sin x
a
x
a
x cos 2
2
Low energy
2
x sin 2
a
High energy
x
V(x)
a
94
Behaviour Near Zone
Boundary II
• Two states of comparable energy – significant contribution from
both: Ek , 1 V1 / 2 Ck ,1
C
k k , 1
V /2 E C
C
k ,0 k ,0
1
k ,0
V1 / 2 Ck ,1
Ek , 1 k
C 0
V
/
2
E
1
k ,0
k k ,0
2
V
Ek , 1 εk Ek ,0 εk 1 0
2
k2
V
Ek , 1 Ek ,0 εk Ek , 1Ek ,0 1
2
2
Ek ,1 Ek ,0
εk Ek , 1 Ek ,0 2
Ek ,1 Ek ,0 2
V
4 1
2
k
εk Ek , 1 Ek ,0 2
V
4 Ek , 1Ek ,0 1
2
V1
2
2
Stationary
States at
zone
boundary
Free
electron
Calculate in this region,
use symmetry for rest
2
G1/2 k/G1
95
Nearly Free Electron Model:
Effect of Potential
• Higher Fourier components of potential: V x
Vn cosnG1x
n
link states at higher energy and give gaps of Vn at k=nG1/2
• Potential ‘folds back’ space – k only meaningful in 1st
Brillouin Zone.
Reduced Zone
Scheme
Extended Zone Scheme
V3
k
3rd Band
V2
2nd Band
V1
1st Band
-1.5
-1
-0.5
0
0.5
1
1.5
k/G1
-0.5
0.5
1st Brillouin
Zone
96
Metals, and Insulators: 1D
• The material can only conduct if there are empty states
accessible to the electrons – otherwise no change of state
is possible
• Separation of states in k space 2/A. But, A=na, so there
are n k states, and 2n electron states in the first Brillouin
Zone.
• 1 electron per atom?
First band ½ full → conductor.
• 2 electrons per atom? First band full → insulator.
• 3 electons per atom?
2nd band ½ full → conductor
• But – get divalent metals – need full 3D picture.
1 electron
2 electrons
3 electrons
/atom
/atom
/atom
k
V2
F
V1
F
F
-0.5
0.5
-0.5
0.5
k/G1
-0.5
0.5
97
Metals, and Insulators: 2D
Model of a Divalent Element
Fermi
Contour
k
kx/G1
Free electron
model – parabolic
dependence of k
on |k|
ky/G1
Empty states
in 1st BZ
Filled
states
Weak potential:
energy of 2nd band
overlaps with
energy of 1st, Fermi
contour crosses
into 2nd B.Z. –
empty states left in
corners of 1st B.Z.
→ conductor
All states in
1st B.Z. full
Strong potential:
Gap so large
enough for there to
be no overlap
between 1st and 2nd
band – so 1st band
full → insulator 98
Conduction/Resistance
k
No Electric field
– electrons in
ground state
k
-G0/2
G0/2
In presence of electric field all states
move, but are scattered back
towards ground state
f eE
dk
dt
Scattering
Thermalisation
k
99
Bloch Oscillations
If the scattering of the electrons is weak, then a constant
electric field causes the occupied states to move
continuously through k space – but the group velocity,
and hence the position of the electrons, oscillate.
dk
f eE
dt
Group
Velocity
k
Increasing
Time
k
k
1st Brillouin Zone
100
Observation of Bloch
Oscillations
• Observation in solid state: reduce scattering rates (very
cold sample, 4K), and create huge unit cell in 1 dimension
(‘super lattice’ with 34 layers GaAs and 6 layers
Al0.3Ga0.7As) giving a very small first Brillouin zone in k
space – reducing time for the electron states to cross the
zone. Submillimeter- wave radiation (THz) emitted at a
frequency dependent on electric field [Phys.Rev. Lett 70,3319
(1993)
• Observed for ultra cold Cs atoms held in a lattice made by
optical standing wave, use lasers to push atoms with a
constant force – observe following time dependence of
atoms momentum [Phys. Rev. Lett 76, 4508 (1996)].
101
Conduction of a Nearly Full
Band
No electric field – a
few empty states at
the top of the band
E
f eE dk dt
All states move
in k space
vg
Full band – no net
motion of charge
A few positively
charged states
moving in direction of
field. HOLES
102
Hole Bands: definition
Consider energy required
to create a hole: The
lower the starting state
the more energy you put
in/gets stored in the
system
Measure energy
of empty state e
w.r.t. top of band.
Total energy of band= - e
Total momentum of band=-ke
Define: h = - e
kh = - ke
i.e. so ‘hole’ carries momentum and
energy due to the empty state.
Measure k of
empty state, ke,
w.r.t. top of band.
Hole band:
h = ħ2ke2/2mh
Corresponding hole:
h , k h
Missing electron:
e , k e
103
Hole Bands: illustration of
rational I
Electron Behaviour (i.e. of
the electron in a full band that is not
‘cancelled out’ by empty state)_
Equivalent Hole
Behaviour
Momentum:
Net momentum due to electron in
state on opposite side to hole, and k
of band (=momentum/ ) on opposite
side to k of unoccupied state
K of hole gives correct k and
momentum due to presence of
unoccupied state in band
Current:
Net current due to electron moving
in opposite direction to group
velocity at hole position
Net current due to positive hole
moving as expected from group
velocity
I
I
vg
vg
Net current carried by to electron
in state on opposite side to hole
104
Hole Bands: illustration of
rational II
Electron Behaviour
Equivalent Hole
Behaviour
vg
Effect of electric field:
Before acceleration
vg
E
After acceleration:
Missing negative charge has
accelerated in direction of field –
WRONG WAY – negative mass
Positively charged hole has
accelerated in direction of field as
expected – so positive mass
vg
vg
105
Status of ‘Hole Model’
2
m
*
• Effective mass (overhead 76:
d 2
dk 2
• At top of a band – negative curvature, and negative mass,
and electrons accelerate in opposite direction to force – all
rather confusing.
• Much better to consider motion of holes, with positive
charge and positive mass, that move in the expected way:
E
Hole band
looks just like
a free particle
band.
vg
k
• As ever – all we want is a model/picture that helps us
‘understand’ what is going on and enables us with a
minimum of mental gymnastics and calculation predict
how a solid will behave.
106
A 2D Model of a Divalent Metal
Extended zone
scheme
Reduced zone
scheme
Holes
Filled states
in 1st B.Z
Filled states
in 2nd B.Z
Back to extended zone.
Pockets of holes that
look like 2D free particles
with same m* in x and y
directions.
Pockets of electrons with
different m* in x and y
107
directions.
Observation of holes: Hall
Effect
• Hall effect
• Holes and electrons both conduct electricity:
e
ne e 2t e
me*
,
Current density :
nh e 2t h
h
mh
j e h E
• If te=th and ne=nh as is the case for a divalent metal,
then, since the curvature of the bottom of the 2nd band
is greater than that of the top of the first, m*e>mh and
hole conduction dominates, giving a Hall coefficient
characteristic of positively charged carriers.
• (For an evaluation of the Hall coefficient, see, for
example, Hook and Hall, pg 154.)
108
Measurement of Effective
Mass: Cyclotron Resonance
• Cyclotron resonance
• In the presence of a B field, charged particles perform
circular orbits around the lines of B of frequency w=eB/m.
B
Holes
F=ev x B=mwv
Electrons
kh moves
around a circle
in k space
• To see a resonance, charged particle must at least travel
1 radian, so wt>1. To achieve this w must be high
(typically ≈ a few 10’s of GHz) – so large magnetic fields
are needed (i.e. a few Tesla) (use a superconducting
solenoid), and very pure samples a very low tempatures
(typically 4K) to increase t.
• From observed resonant frequency (i.e. a strong
adsorption of radio waves), m*e or mh can be determined.
•
dw
Remember its the group velocity dk that is important -so
where the origin (k=0) is does not matter.
109
Doping of Semiconductors
• So far we have considered only holes and electrons
intrinsic to the material.
• For e.g. a divalent metal these exist at absolute zero.
• For a semiconductor the lower (valence) band is full at
absolute zero and there are no conductors. Thermal
excitation at non zero temperatures produces pairs of holes
and electrons.
• Can add impurities (‘dopants’) that replace a few of the
semiconductor atoms.
• e.g. adding a group V atom (e.g. P or As) leaves a spare
electron after all the normal bonding (valence band) states
are full, and so it must go to the conduction band.
However, if the electron moves away from the group V
atom, it leaves the atom positively charged – and so the
electron forms hydrogen like ‘donor’ states out of the
available free levels at the base of the conduction band,
which will have an energy below the bottom of the
conduction band and mean radius given by:
En
me*e 4
2 n 40
2 2 2
2
rn
n 2 2
me*e 2
40
H atom formulae
but using and m*
• E.g. for germanium me*=0.2me and the dielectric constant
=15.6 giving:
me*
13.6eV 0.01eV
E1
2
m
e
me
40A
0.53A
r1
m*
e
Radius much larger than inter110
atomic spacing – justifies ‘effective
mass’ approach – see OH76
H-Atom Like Impurity States
• Before looking further at dopant states in a semiconductor
– look at analogue in free space: the hydrogen atom.
• The electronic wavefunction in hydrogen can be written (in
Fourier transform form) as a sum of plane waves.
• Before a hydrogen nucleus is introduced these plane
waves can be represented as a free electron band:
Free electron ‘band’
0
k
H atom energy level
(only one shown)
• You can consider the H atom wavefunctions to be made
out of a sum of these free atom states – the presence of
the electrostatic potential makes a state with an binding
energy – but no k dependence since it is an isolated
object, not a periodic one – so the state is represented as
a flat line on an v. k diagram.
• For every defect state you create – you loose one state
from the band of states that it is created from.
111
n type Doping: Donor States
• Replace a silicon atom by e.g. a phosphorous atom that has
its core shells full and the same number of valence electrons
as Si. The system has the same no of electrons as pure Si,
but the P atom carries a single + charge.
Si
Si
Si
Si
+
P
Si
Si
Si
Si
Conduction
band:
empty
Valence
band: full
• All states in valence band full – a log jam, no flexibility to make
new states, but the energy of all the states is slightly changed.
• The spare electron goes to the conduction band and is either:
‘Bound’
‘Ionised’
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
+
-P
Si
Si
Si
+
P
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
e in H atom like
Si
Si
e-
e- far
away+free
or:
e
state made of C.
band states but
lower in energy
because e- feels
potential of P+
112
p Type Doping:Acceptor States
• Now replace a silicon atom by e.g. a Al atom that has its core
shells full and the same number of valence electrons as Si.
They system has the same no of electrons as pure Si, but the
Al atom carries a single - charge.
• The electron has to come from somewhere – takes an
electron from the valence band leaving a hole that either is:
‘Bound’
V. Band lacks an
electron – leaves
a state to be
made into a
localised state
(no k dep.)
or:
Bound state
occupied by
e-
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
h+
Si
Si
Si
Al
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
-
The empty electron state and full valence
band are upside down to the ‘full hole
state and empty hole band’ picture.
‘Ionised’
h+
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Al
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
Si
-
hole far
away + free
113
p/n Type Impurity States in Si
K Space
Picture
Conduction
band
EDonor: 0.045eV
below C. band
edge for P in Si
Gap energy:
1.08eV for Si
Valence
band
EAcceptor: 0.057eV
above V. band edge
for Al in Si
T=0K
Real Space
Picture
Conduction
band: empty
Acceptor/donor
states
Valence
band: full
Distance in
Space
114
Chemical Potential of Doped
Semiconductor
p
• n type
1
exp k BT 1
T=0K
Conduction
band: empty
Donor states full
1
when
2
To see where will lie,
ask where half occupied
states would be
T>0K
Donor State
Ionised
Valence
band: full
• p type
T=0K
Conduction
band: empty
Holes left by
thermal excitation
moves towards
middle of gap as T
increases. At higher T
no. of thermally
excited e- increases
and presence of donor
states becomes less
important.
Valence band: full
Acceptor states
empty (=full of holes)
T>0K
Acceptor State Ionised
= occupied by e-
Holes left by
thermal excitation
115
pn Junctions
• Basis of many electronic devices.
• Chemical potential, , is the effective ‘filling level’ of
the states: before p and n type material contact the
n type is filled to a higher level.
n-type
T>0K
p-type
e- diffusion
Hole
diffusion
• When p and n type contact, electrons diffuse from
n (high concentration) to p (low) (i.e. from high to
low ) and holes diffuse the other way.
• This charge separation produces an electric field at
the junction that stops further diffusion and
produces an electrostatic potential difference that
evens out the difference in . (within the p or n type
material fixed by requirement of charge neutrality,
‘contact potential’ raises energy of whole of p type
region w.r.t n type region)
116
pn Junctions
p-type
E field in
junction stops
carrier
diffusion
n-type
Ej
ef
Contact
potential
Width of
depletion layer
such that
contact
potential
evens out
Depletion
layer
ndonor
nacceptor
Carrier Density
Charge Density
E field in
junction
end
ena
f
Electrostatic
potential f
0
dE x x
dx
0
E
Ej
117
Position of junction
pn Junctions:
Diode: Basic idea
Forward Bias Junction:
Apply a field (potential difference) that forces
majority carriers towards junction => conducts
p-type
n-type
‘minority’ carrier
generated by
thermal excitation
V
‘majority’ carriers
due to doping
Applied
potential
EBias
V
Forward bias acts mainly
across junction and reduces
contact potential
Reverse Bias Junction (not shown):
Apply a field that forces majority carriers away from
junction and increases width of depletion layer
=> conducts v. poorly since there are very
few minority carriers.
118
pn Junctions:
Diode: Generation and
Recombination Currents
n-type
Electrons in this
region can still
cross barrier
e- Recombination Current
e(fV
p-type
e- Generation Current
V
h+ Generation Current
h+ Recombination Current
• Carriers diffuse towards junction.
• Majority carrier diffusion creates a ‘recombination
current’ – when they diffuse across the junction
they will need to recombine with holes as (for a
forward bias) there will more present than the
equilibrium (F.D. distribution) number.
• Minority carrier diffusion creates a ‘generation
current’ – when they diffuse across the junction
they enter a region depleted by diffusion the other
way, and so are needed to maintain the F.D.
distribution of carriers.
119
pn Junctions:
Diode: Current Characteristics
Virtually no electrons
at top of band
Occupation probability
Increased by expV k BT
I0,e
I0,e exp[V/kBT]
V
No bias
exp k BT
exp k BT 1
So if increases to μ V then p becomes :
pV exp V k BT exp k BT exp V k BT
Far above : p0
1
p0 exp V k BT
For no bias the recombination current ( ) balances the generation
current (
). The bias does not does not change the generation
current I0,e. Number density of electrons in the n type valence band
and hence the recombination current is increased by exp[V/kBT], so
net current due to electrons is:
.
I e I 0,e exp V k BT 1
An identical argument applies to the holes, so the total current through
the diode is given by:
I I 0 exp V k BT 1
120
(Note: we are assuming that the density of state g() is roughly constant within the bands.)
Breakdown Under Reverse
Bias: Zener Diodes
• pn diode I/V:
I I 0 exp V k BT 1
I
Tiny reverse current
due to thermally excited
minority carriers
V
• A Zener Breakdown
• Caused by tunnelling at large reverse bias (typically
above 3V)
• Used as a reference voltage in circuits
Tunnelling
through barrier
I
p-type
V
n-type
121
Breakdown Under Reverse
Bias: Avalanche Breakdown
• Avalanche Breakdown
Field from reverse bias so strong
that thermally excited carriers within
junction gain enough energy
between collisions to create
electron hole pairs that then create
more pairs etc
p-type
n-type
122
Light Emitting Diodes
p-type
n-type
Electrons and holes
recombine in depletion layer
– if ‘direct band gap’ then
light emission can be most
favourable process– get
light emitting diode with
colour determined by band
gap
Direct Band Gap:
Indirect Band Gap:
Photon emission easy
(e.g. GaAs)
Photon emission requires
momentum from phonon –
slow (e.g. Si)
q
k
Photon k =w/c so very small –
almost vertical transition
123
p-n Junction Lasers
Basic Laser Principle
n2
Occupation
probability
Electrons driven up
and down by light
n1
Photon energy matches
energy between levels
If n2>n1 (‘population inversion’) then more light
emitted than adsorbed – get Light Amplification
by Stimulated Emission of Radiation
Diode Laser
Heavily doped semi-conductor chemical potential in conduction/
valence bands. For strong forward bias, get large no of carriers
injected into other side of junction – hence population inversion and
possibility of laser action.
p-type
n-type
Carrier injection
Below , so n2>0.5
Above , so n1<0.5
124
pn Junctions as Solar Cells
Ej
p-type
n-type
Carriers pairs created
by photon adsorption
are swept out of the
junction by the electric
field, creating a photo
current IL.
Equivalent
Circuit
IL
Current
source
Output
Impedance
ID
IS
Shunt
Resistor
I
Diode
Characteristics- key
ID to V/I of cell
IL-IS-I
I = IL-ID-IS
Open circuit voltage drops as I increases
V
125
126
Appendix:Tight Binding Model
(non examinable)
• Nearly Free Electron Model
• Derived as a deviation from free electron model.
• Basic view: electrons free to move through a metal
as plane waves, but perturbed by a modest potential
caused by the ion cores.
• Reasonable starting point model for many metals,
where outer most electrons relatively weakly bonded
to the atoms.
• The structure of the ion lattice is dominated by
packing of the ions and a trade off between potential
energy gain (the closer the ions the stronger the
average potential experienced by the electrons) and
kinetic energy of the electrons – some metals
(potassium, iron at room temperature) are b.c.c. not
the close packed f.c.c. .
• Tight Binding Model
• For many materials the bonding is clearly covalent
not metallic – e.g. Si, Ge, organic conductors.
• Bonding and structure dominated by overlap/
alignment of atomic orbitals.
• An expectation that the electrons are localised in
covalent bonds.
127
(This presentation of the TBM is from a previous course by Dr. W. Allison.)
Spatial Extent of Orbitals
• Spatial extent of the wavefunctions is determined by
their l-value.
• For example: hydrogen:
• Spatial extent increases with n, an obvious
consequence of orthogonality.
• Higher l-values are more compact than lower l, for a
given n. This too is a consequence of orthogonality.
The angular variation in higher l-values allows
orthogonality with the lower n-states without the
need to expand in r.
• The spatial extent of an orbital determines the
overlap with neighbouring atoms
128
Tight Binding Model: the basis
states.
• Tight binding model
• The simplest and most intuitive model
describing the electronic structure of a solid
comes from tight-binding theory.
• Atomic orbits are used to construct Bloch
states.
• First write the wavefunction, for a Bloch state,
labelled, q, as a sum of functions on lattice
sites, j.
q r exp iq.r j r r j
j
• The expression defines f(r), known as a
Wannier function.
• It is easy to show this has the correct, Bloch
form
q r R exp iq.r j r R r j
j
exp iq.R exp iq. r j R r r j R
j
exp iq.R k r
simply another
lattice vector, rm, say
129
Tight-binding model
f(r) can be represented as a sum over atomic states
r cnlmnlm r o r
nlm
• The approximation, above, uses a single atomic
o
state r . It is the simplest and is useful provided
the state is well localised.
• Then the energy of the state, k is
q q H q exp iq.r j rm mo H oj
j
m
• For simplicity take a 1-D solid
with atoms at r = ma.
state, f o, at site m
• We can neglect all terms except the nearest
neighbours j=m, m±1, since the matrix elements
decrease rapidly with the separation of the
wavefunction centres. Hence we get 3 terms
q a exp iq.0 exp iqa exp iqa
a mo H mo
overlap integral
mo H oj
130
Energy bands
• The energy band becomes
number of nearest neighbours
q a 2 cosqa
• Width of band directly proportional to the orbital
overlap, , and the bandwidth = 4.
• Note the factor 2 arises because we have two
neighbours contributing. The analysis can easily
be extended to 3-D. Assuming a cubic crystal we
will have 6 neighbours (2 in x-direction, 2 in y and
2 in z). The band structure is
q a 2 cosq x a cosq y a cosqz a
• The bandwidth is now = 12.
• We note that the other factor in determining the
bandwidth is the number of neighbours
(coordination number).
131
Shells and bands
• Valence band
• For s-states < 0; p-states > 0. (N.B. in H,
V<0)
• Spatially extended atomic states gives overlap
and hence broad bands.
• s-p bands typically 10-20eV
• d bands usually narrower since d -states are
more compact (less overlap)
• Core states
• Highly compact with negligible overlap
• bandwidth is negligible and the states are
essentially the same as atomic states.
132
Scattering and Coalescence I
• A typical phonon-phonon collision process is coalescence:
Real Space
q3
Outgoing
phonon
q2
q1
Lattice distorted
by a phonon
Incoming phonon
• Anharmonicity allows the incoming phonon q1 to
diffracts off another phonon, q2 producing an outgoing
phonon q3.
• Usual diffraction condition obeyed: q3 = q1 + q2
133
A1
(c.f. kf = ki + G )
Scattering and Coalescence II
• The diffraction condition means that the outgoing phonon
picks up momentum both from the incoming phonon and
the phonon from which in incoming phonon is diffracted:
q3
q3
q2
q3 q1 q 2
q1
q1
q2
so one quantum has been taken from the incoming (1)
and the scattering (2) phonon state and combined in the
process of coalescence into the outgoing phonon (3),
that will also have to take an energy that is the sum of
the energies of the incoming and scattering phonon
quanta:
w3 w1 w2
• If a diffraction process occurs that diffracts the orignal
phonon in the direction –q2, the momentum equation
becomes:
q3 q1 q 2 q1 q 2 q3
and the energy:
w3 w1 w2
w1 w2 w3
i.e. the incoming phonon has decomposed into two
outgoing phonons.
q2
q3
q2
q1
q1
q3
• Remember the zero point motion – even if there are no
quanta of energy in a mode – the zero point motion 134
A2
still distorts the lattice and so can induce diffraction.