* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Appropriate Glove Use
Destruction of Syria's chemical weapons wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Soil contamination wikipedia , lookup
History of chemistry wikipedia , lookup
Acid–base reaction wikipedia , lookup
Chemical biology wikipedia , lookup
Green chemistry wikipedia , lookup
Biochemistry wikipedia , lookup
Al-Shifa pharmaceutical factory wikipedia , lookup
Chemical potential wikipedia , lookup
Chemical weapon proliferation wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Chemical weapon wikipedia , lookup
Chemical Corps wikipedia , lookup
Chemical plant wikipedia , lookup
Fine chemical wikipedia , lookup
Right to know wikipedia , lookup
Drug discovery wikipedia , lookup
Triclocarban wikipedia , lookup
Organic chemistry wikipedia , lookup
Safety data sheet wikipedia , lookup
Registration, Evaluation, Authorisation and Restriction of Chemicals wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
California Green Chemistry Initiative wikipedia , lookup
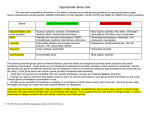
Appropriate Glove Use The chemical compatibility information in this table is intended to provide general guidelines for appropriate glove usage. Glove manufacturers provide specific, detailed information on their websites. Contact DRS at 333-2755 if you have questions. Glove Good Chemical Resistance Natural Rubber Latex (w/wo powder)* Nitrile Poor Chemical Resistance Laminate (BarrierTM, Silvershield ) Neoprene Aqueous solutions, acetone, formaldehyde, alcohols, dilute mineral acids and bases Aliphatic and aromatic hydrocarbons, DMSO, formaldehyde, alcohols, most amines, ethyl ether Chlorinated aliphatic and aromatic solvents, ketones, THF, isocyanates, DMSO Acids, alcohols, acetonitrile Many organic solvents, ethyl ether, chlorinated hydrocarbons, Perchloric acid, isocyanates Chlorinated hydrocarbons, aldehydes, nitro compounds, isocyanates Diethylamine, dimethylethylamine, 70% HF Acid Resistant Concentrated acids Most organic solvents TM Chlorinated solvents Thin barrier protective gloves (such as Natural Rubber Latex and Nitrile) are designed to provide barrier protection and tactile sensitivity to the wearer. These gloves are not designed for applications including prolonged, direct exposure to chemicals. Double gloving with two gloves of different materials may be necessary if there is no chemical resistance data available for the chemicals you are using. A thin glove used over heavier gloves provides increased tactile sensitivity. • Verify that your gloves are compatible with your specific application, process and materials before using them. • Change your gloves whenever they appear soiled or worn; be alert for permeation of your gloves by the chemicals you are using. Change gloves immediately if permeation occurs. • Be alert to any sensation of numbness, or tingling of your fingers or hands when working with chemicals. Remove gloves, wash thoroughly with soap and water and seek medical attention for assistance. *Latex gloves have been implicated in the exacerbation of latex allergies; powdered latex gloves have been shown to contain larger amounts of the latex protein allergen than non-powdered latex gloves. If you suspect that you may have an allergy to latex, or have a propensity for allergic reactions and skin rashes, avoid the use of latex gloves. S:\CSWMS\Chemical Safety\Glove Selection - chemical resistance guides\2009-3-28 Appropriate Glove Use.doc