* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Compounds Containing A Single Bond To A
Asymmetric induction wikipedia , lookup
Aromatization wikipedia , lookup
Aromaticity wikipedia , lookup
Homoaromaticity wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Hydroformylation wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
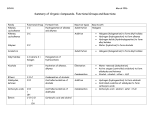
ORGANIC CHEMISTRY Dr. Serkan SAYINER [email protected] Compounds Containing A Single Bond To A Heteroatom Alkyl Halides, Alcohols, Ethers, Amines, Thiols, Sulfides Compounds Containing a Single Bond to a Heteroatom Several types of functional groups contain a carbon atom singly bonded to a heteroatom. • These are alkyl halides, alcohols, ethers, amines, thiols and sulfides. Molecules containing these functional groups may be simple or very complex. • It doesn’t matter what else is present in other parts of the molecule. Bromomethane Methanol Dimethyl ether Methylamine Methanethiol Dimethyl sulfide Alkyl Halides Introduction, Nomeclature (IUPAC system and common names), Physical Properties, Interesting Alkyl Halides, The Polar Carbon–Halogen Bond (substitution and elimination reactions) Introduction Alkyl halides are organic molecules containing a halogen atom X bonded to an sp3 hybridized carbon atom. • Halogens are any of the elements fluorine, chlorine, bromine, iodine, and astatine, occupying group VIIA (17) of the periodic table. They are reactive nonmetallic elements that form strongly acidic compounds with hydrogen, from which simple salts can be made. • Denoting lamps and radiant heat sources use a filament surrounded by the vapor of iodine or another halogen. Introduction Alkyl halides are classified as primary (1°), secondary (2°), or tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen. • Whether an alkyl halide is 1°, 2°, or 3° is the most important factor in determining the course of its chemical reactions. • Alkyl halides have the general molecular formula CnH2n+1X, and are formally derived from an alkane by replacing a hydrogen atom with a halogen. Introduction There are four types of organic halides having the halogen atom in close proximity to a π bond. 1. Vinyl halides have a halogen atom bonded to a carbon–carbon double bond 2. Aryl halides have a halogen atom bonded to a benzene ring. 3. Allylic halides have X bonded to the carbon atom adjacent to a carbon–carbon double bond 4. Benzylic halides have X bonded to the carbon atom adjacent to a benzene ring. Nomenclature The systematic (IUPAC) method for naming alkyl halides follows from the basic rules described in hydrocarbons. They have common names too, because many low molecular weight alkyl halides are often referred to by their common names. According to IUPAC System; • An alkyl halide is named as an alkane with a halogen substituent— that is, as a halo alkane. To name a halogen substituent, change the -ine ending of the name of the halogen to the suffix –o (chlorine → chloro). Name the parent chain as an alkane, with the halogen as a substituent bonded to the longest chain. Begin at the end nearest the first substituent, either alkyl or halogen. ANSWER: 2-chloro-5-methylheptane Nomeclature Common Names • Common names for alkyl halides are used only for simple alkyl halides. To assign a common name: • Name all the carbon atoms of the molecule as a single alkyl group. • Name the halogen bonded to the alkyl group. To name the halogen, change the -ine ending of the halogen name to the suffix -ide; for example, bromine bromide. • Combine the names of the alkyl group and halide, separating the words with a space. Physical Properties Alkyl halides are weakly polar molecules. They exhibit dipole–dipole interactions because of their polar C—X bond, but because the rest of the molecule contains only C—C and C—H bonds they are incapable of intermolecular hydrogen bonding. Alkyl halides have higher boiling points and melting points than alkanes having the same number of carbons. • CH3CH3 bp= -89º C iken CH3CH3Br bp= 39º C Physical Properties Boiling points and melting points increase as the size of R increases. • Larger surface area, higher boiling and melting point. Boiling points and melting points increase as the size of X increases. • More polarizable halogen, higer melting and boiling point. RX is soluble in organic solvents, but not in water. Interesting Alkyl Halides Many simple alkyl halides make excellent solvents because they are not fl ammable and dissolve a wide variety of organic compounds. Compounds in this category include CHCl3 (chloroform or trichloromethane) and CCl4 (carbon tetrachloride or tetrachloromethane). Large quantities of these solvents are produced industrially each year, but like many chlorinated organic compounds, both chloroform and carbon tetrachloride are toxic if inhaled or ingested. • Chloromethane (CH3Cl) is produced by giant kelp and algae and also found in emissions from volcanoes such as Hawaii’s Kilauea. • Dichloromethane (or methylene chloride, CH2Cl2) is an important solvent, once used to decaffeinate coffee. Coffee is now decaffeinated by using supercritical CO2 (liquid state) due to concerns over the possible ill effects of trace amounts of residual CH2Cl2 in the coffee. • Halothane (CF3CHClBr) is a safe general anesthetic that has now replaced other organic anesthetics such as CHCl3 (chloroform), which causes liver and kidney damage, and CH3CH2OCH2CH3 (diethyl ether), which is very flammable. Interesting Alkyl Halides Synthetic organic halides are also used in insulating materials, plastic wrap, and coatings. Two such compounds are Teflon and poly(vinyl chloride) (PVC). Interesting Alkyl Halides Although the beneficial effects of many organic halides are undisputed, certain synthetic chlorinated organics such as the chlorofluorocarbons and the pesticide DDT have caused lasting harm to the environment. • Chlorofluorocarbons having been extensively used as a refrigerant and an aerosol propellant. • Sunlight catalyzes their decomposition, a process that contributes to the destruction of the ozone layer. Interesting Alkyl Halides • DDT is an organic molecule with valuable short-term effects that has caused long-term problems. DDT kills insects that spread diseases such as malaria and typhus, and in controlling insect populations, DDT has saved millions of lives worldwide. • DDT is a weakly polar organic compound that persists in the environment for years. Because DDT is soluble in organic media, it accumulates in fatty tissues. • DDT is acutely toxic to many types of marine life (crayfish, sea shrimp, and some fish), but the long-term effect on humans is not known. The Polar Carbon–Halogen Bond The properties of alkyl halides dictate their reactivity. The electronegative halogen X creates a polar C– X bond, making the carbon atom electron deficient. The chemistry of alkyl halides is determined by this polar C—X bond. The characteristic reactions of alkyl halides are substitution and elimination. Because alkyl halides contain an electrophilic carbon, they react with electron-rich reagents; Lewis bases (nucleophiles) and Brønsted–Lowry bases. The Polar Carbon–Halogen Bond A nucleophile is a reactant that provides a pair of electrons to form a new covalent bond. In other words, nucleophiles are Lewis bases. When the nucleophile donates a pair of electrons to a proton, it’s called a Brønsted base, or simply, “base”. • Nucleophiles and bases are structurally similar: both have a lone pair or a π bond. They differ in what they attack. • Bases attack protons. Nucleophiles attack other electron- deficient atoms (usually carbons). The Polar Carbon–Halogen Bond Alkyl halides undergo substitution reactions with nucleophiles. • In a substitution reaction of RX, the halogen X is replaced by an electron-rich nucleophile :Nu–. The C—X σ bond is broken and the C—Nu σ bond is formed. The Polar Carbon–Halogen Bond Alkyl halides undergo elimination reactions with Brønsted–Lowry bases. • In an elimination reaction of RX, the elements of HX are removed by a Brønsted–Lowry base :B. The Polar Carbon–Halogen Bond All elimination reactions involve loss of elements from the starting material to form a new π bond in the product. Alkyl halides undergo elimination reactions with Brønsted–Lowry bases. The elements of HX are lost and an alkene is formed. THE POLAR CARBON–HALOGEN BOND Removal of the elements of HX, called dehydrohalogenation, is one of the most common methods to introduce a π bond and prepare an alkene. Alcohols and Ethers Introduction, Nomenclature, Physical Properties, Interesting Alcohols, Ethers, and Epoxides, Preparation of Alcohols and Ethers, Reactions of Alcohols, Oxidation and Blood Alcohol Screening, The Metabolism of Ethanol, Reactions of Ethers Introduction Alcohols and ethers are two functional groups that contain carbon–oxygen σ bonds. • Although alcohols and ethers share many characteristics, each functional group has its own distinct reactivity, making each unique and different from the alkyl halides. Introduction Alcohols (ROH) contain a hydroxy group (OH group) bonded to an sp3 hybridized carbon atom. Alcohols are classified as primary (1°), secondary (2°), or tertiary (3°) based on the number of carbon atoms bonded to the carbon with the OH group. Compounds having a hydroxy group on an sp2 hybridized carbon atom —enols and phenols— undergo different reactions than alcohols. • Enols have an OH group on a carbon of a C=C double bond. • Phenols have an OH group on a benzene ring. Introduction Ethers (ROR) have two alkyl groups bonded to an oxygen atom. An ether is symmetrical if the two alkyl groups are the same, and unsymmetrical if they are different. Introduction Both alcohols and ethers are organic derivatives of H2O, formed by replacing one or both of the hydrogens on the oxygen atom by R groups, respectively. Epoxides are ethers having the oxygen atom in a three-membered ring. Epoxides are also called oxiranes. Nomenclature To name an alcohol or ether using the IUPAC system, we must learn how to name the functional group either as a substituent or by using a suffix added to the parent name. Naming Alcohols • In the IUPAC system, alcohols are identified by the suffix -ol. Change the -e ending of the parent alkane to the suffix -ol. Step [2] Number the carbon chain to give the OH group the lower number, and apply all other rules of nomenclature. Answer: 5-methyl-3-hexanol Nomenclature When an OH group is bonded to a ring, the ring is numbered beginning with the OH group. Because the functional group is always at C1, the “1” is usually omitted from the name. The ring is then numbered in a clockwise or counterclockwise fashion to give the next substituent the lower number. Nomenclature Common names are often used for simple alcohols. To assign a common name: • Name all the carbon atoms of the molecule as a single alkyl group. • Add the word alcohol, separating the words with a space. Nomenclature Compounds with two hydroxy groups are called diols (using the IUPAC system) or glycols. Compounds with three hydroxy groups are called triols, and so forth... Poliols. To name a diol, for example, the suffix -diol is added to the name of the parent alkane, and numbers are used in the prefix to indicate the location of the two OH groups. Nomenclature Naming Ethers • Simple ethers are usually assigned common names. • To do so, name both alkyl groups bonded to the oxygen, arrange these names alphabetically, and add the word ether. • For symmetrical ethers, name the alkyl group and add the prefix di-. Nomenclature • More complex ethers are named using the IUPAC system. One alkyl group is named as a hydrocarbon chain, and the other is named as part of a substituent bonded to that chain. • Name the simpler alkyl group + O atom as an alkoxy substituent by changing the -yl ending of the alkyl group to -oxy. • Name the remaining alkyl group as an alkane, with the alkoxy group as a substituent bonded to this chain. Answer: 4-ethoxyoctane Physical Properties Alcohols, ethers, and epoxides exhibit dipole–dipole interactions because they have a bent structure with two polar bonds. Alcohols are also capable of intermolecular hydrogen bonding, because they possess a hydrogen atom on an oxygen, making alcohols much more polar than ethers and epoxides. Physical Properties For compounds of comparable molecular weight, the stronger the intermolecular forces, the higher the boiling point or melting point. • Alcohols and Ethers have higher boiling points and melting points than hydrocarbons of comparable size and shape. • Ethers have lower melting points and boiling points than alcohols of comparable size and shape. • Boiling point increase as the extent of hydrogen bonding increases. Physical Properties Solubility • Alcohols, ethers, and epoxides having ≤ 5 C’s are H2O soluble because they each have an oxygen atom capable of hydrogen bonding to H2O. • Alcohols, ethers, and epoxides having > 5 C’s are H2O insoluble because the nonpolar alkyl portion is too large to dissolve in H2O. • Alcohols, ethers, and epoxides of any size are soluble in organic solvents. Interesting Alcohols Ethanol (CH3CH2OH), formed by the fermentation of the carbohydrates in grains, grapes, and potatoes, is the alcohol present in alcoholic beverages. • It is perhaps the first organic compound synthesized by humans, because alcohol production has been known for at least 4000 years. • Ethanol depresses the central nervous system, increases the production of stomach acid, and dilates blood vessels, producing a flushed appearance. • Ethanol is also a common laboratory solvent, which is sometimes made unfit to ingest by adding small amounts of benzene or methanol (both of which are toxic). • Ethanol is a common gasoline additive, widely touted as an environmentally friendly fuel source. Methanol (CH3OH) is also called wood alcohol, because it can be obtained by heating wood at high temperatures in the absence of air. Methanol is extremely toxic because of the oxidation products formed when it is metabolized in the liver. Ingestion of as little as 15 mL causes blindness, and 100 mL causes death. 2-Propanol [(CH3)2CHOH] is the major component of rubbing alcohol. When rubbed on the skin it evaporates readily, producing a pleasant cooling sensation. Because it has weak antibacterial properties, 2-propanol is used to clean skin before minor surgery and to sterilize medical instruments. Ethylene glycol (HOCH2CH2OH) is the major component of antifreeze. It is readily prepared from ethylene oxide. It is sweet tasting but toxic. Glycerol [(HOCH2)2CHOH] is used in lotions, liquid soap, and shaving cream. Since it is sweet tasting and nontoxic, it is also an additive in candy and some prepared foods. Its three OH groups readily hydrogen bond to water, so it helps to retain moisture in these products. Interesting Ethers The discovery that diethyl ether (CH3CH2OCH2CH3) is a general anesthetic revolutionized surgery in the nineteenth century. • Diethyl ether is an imperfect anesthetic, but considering the alternatives in the nineteenth century, it was a miracle drug. • It is safe, easy to administer, and causes little patient mortality, but it is highly flammable and causes nausea in many patients. • For these reasons, alternatives to diethyl ether are now widely used. Many of these newer general anesthetics, which cause little patient discomfort, are also ethers. These include isoflurane, enflurane, and methoxyflurane. Replacing some of the hydrogen atoms in the ether by halogens results in compounds with similar anesthetic properties but decreased flammability. Interesting Epoxides Although epoxides occur less widely in natural products than alcohols or ethers, interesting and useful epoxides are also known. As an example, two recently introduced drugs that contain an epoxide are eplerenone and tiotropium bromide. • Eplerenone is prescribed to reduce cardiovascular risk in patients who have already had a heart attack. • Tiotropium bromide is a long-acting bronchodilator used to treat the chronic obstructive pulmonary disease of smokers and those routinely exposed to secondhand smoke. Preparation of Alcohols, Ethers, and Epoxides Alcohols and ethers are both common products of nucleophilic substitution. They are synthesized from alkyl halides. Reactions of Alcohols Alcohols undergo two useful reactions; dehydration and oxidation. Dehydration • When an alcohol is treated with a strong acid such as H2SO4, the elements of water are lost and an alkene is formed as product. • Loss of H2O from a starting material is called dehydration. • Dehydration takes place by breaking bonds on two adjacent atoms; the C—OH bond and an adjacent C—H bond. • Dehydration is an example of a general type of organic reaction called an elimination reaction. • Elimination is a reaction in which elements of the starting material are “lost” and a new multiple bond is formed. Ethanol Reactions of Alcohols Oxidation • To determine if an organic compound has been oxidized, we compare the relative number of C—H and C—O bonds in the starting material and product. • Oxidation results in an increase in the number of C—O bonds or a decrease in the number of C—H bonds. • An organic compound like CH4 can be oxidized by replacing C— H bonds with C—O bonds. Organic chemists use the symbol [O] to indicate oxidation. Reactions of Alcohols • Alcohols can be oxidized to a variety of compounds, depending on the type of alcohol and the reagent. • Oxidation occurs by replacing the C—H bonds on the carbon bearing the OH group by C—O bonds. • All oxidation products from alcohol starting materials contain a C=O, a carbonyl group. • Primary (1°) alcohols are first oxidized to aldehydes (RCHO), which are further oxidized to carboxylic acids (RCOOH) by replacing one and then two C—H bonds by C—O bonds. • Secondary (2°) alcohols are oxidized to ketones (R2CO), by replacing one C—H bond by one C—O bond. • Tertiary 3° alcohols have no H atoms on the carbon with the OH group, so they are not oxidized. Oxidation and Blood Alcohol Screening A common reagent for alcohol oxidation is potassium dichromate, K2Cr2O7, a red-orange solid. Oxidation with this chromium reagent is characterized by a color change, as the red-orange reagent is reduced to a green Cr3+ product. The first devices used to measure blood alcohol content in individuals suspected of “driving under the influence,” made use of this color change. Oxidation and Blood Alcohol Screening The oxidation of CH3CH2OH with K2Cr2O7 to form CH3COOH and Cr3+ was the first available method for the routine testing of alcohol concentration in exhaled air. Some consumer products for alcohol screening are still based on this technology. A driver is considered “under the influence” in most countries with a blood alcohol concentration of 0.08%. • Values may vary. The Metabolism of Ethanol When ethanol is consumed, it is quickly absorbed in the stomach and small intestines and then rapidly transported in the bloodstream to other organs. Ethanol is metabolized in the liver, by a two-step oxidation sequence. High molecular weight enzymes, alcohol dehydrogenase and aldehyde dehydrogenase, and a small molecule called a coenzyme carry out these oxidations. The Metabolism of Ethanol The products of the biological oxidation of ethanol are the same as the products formed in the laboratory. When ethanol (CH3CH2OH, a 1° alcohol) is ingested, it is oxidized in the liver first to CH3CHO (acetaldehyde), and then to CH3COOH (acetic acid). If more ethanol is ingested than can be metabolized in a given time period, the concentration of acetaldehyde accumulates. This toxic compound is responsible for the feelings associated with a hangover. Reactions of Ethers Ethers have a poor leaving group, so they cannot undergo nucleophilic substitution or elimination reactions directly. Instead, they must first be converted to a good leaving group by reaction with strong acids. Only HBr and HI can be used, though, because they are strong acids that are also sources of good nucleophiles (Br– and I–, respectively). When ethers react with HBr or HI, both C—O bonds are cleaved and two alkyl halides are formed as products. Amines and Amids Amines; Introduction, Structure and Classification, Nomenclature, Physical Properties, Amines as Bases, Reaction of Amines with Acids, Ammonium Salts, Ammonium Salts as Useful Drugs Amides; Naming an Amide, Physical Properties, Amide hydrolysis, Interesting Amines and Amids, Epinephrine and related compounds, Penicillin Introduction Amines are organic nitrogen compounds, formed by replacing one or more hydrogen atoms of ammonia (NH3) with alkyl groups. Like ammonia, the amine nitrogen atom has a nonbonded electron pair, making it both a base and a nucleophile. As a result, amines react with electrophiles to form ammonium salts; compounds with four bonds to nitrogen. Structure and Classification Amines are classified as 1°, 2°, or 3° by the number of alkyl groups bonded to the nitrogen atom. • A primary (1°) amine has one C—N bond and the general structure RNH2. • A secondary (2°) amine has two C—N bonds and the general structure R2NH. • A tertiary (3°) amine has three C—N bonds and the general structure R3N. Like ammonia, the amine nitrogen atom has a lone pair of electrons, which is generally omitted in condensed structures. An amine nitrogen atom is surrounded by three atoms and one nonbonded electron pair, making it trigonal pyramidal in shape, with bond angles of approximately 109.5°. Structure and Classification The amine nitrogen can also be part of a ring. • Aliphatic and aromatic amines. The structures of morphine and atropine, two alkaloids; naturally occurring amines derived from plant sources. Each alkaloid contains a nitrogen atom in a ring. • The analgesic and narcotic effects of opium are due largely to the alkaloid morphine. • Atropine is isolated from Atropa belladonna, the deadly nightshade plant. Atropine dilates pupils, increases heart rate, and relaxes smooth muscles. Nomenclature To name a primary (1°) amine, name the alkyl group bonded to the nitrogen atom and add the suffix -amine, forming a single word. For 2° and 3° amines with different alkyl groups, alphabetize the names of the alkyl groups. 2° and 3° amines having identical alkyl groups are named by using the prefix di- or tri- with the name of the primary amine. Answer: Pentylamine Answer: ethylmethylamine Aromatic amines, amines having a nitrogen atom bonded directly to a benzene ring, are named as derivatives of aniline. Use the prefix N- before any alkyl group bonded to the amine nitrogen. Physical Properties Many low molecular weight amines have very foul odors. • Trimethylamine [(CH3)3N], formed when enzymes break down certain fish proteins, has the characteristic odor of rotting fish. • Cadaverine (NH2CH2CH2CH2CH2CH2NH2) is a poisonous diamine with a putrid odor also present in rotting fish, and partly responsible for the odor of semen, urine, and bad breath. Physical Properties Because nitrogen is much more electronegative than carbon or hydrogen, amines contain polar C—N and N—H bonds. Primary (1°) and 2° amines are also capable of intermolecular hydrogen bonding, because they contain N—H bonds. In comparing compounds of similar size, 1° and 2° amines have higher boiling points than compounds incapable of hydrogen bonding, but lower boiling points than alcohols that have stronger intermolecular hydrogen bonds. Tertiary (3°) amines have lower boiling points than 1° and 2° amines of comparable size, because they have no N—H bonds. Physical Properties Amines are soluble in organic solvents regardless of size. Amines with fewer than six carbons are water soluble since they can hydrogen bond with water. Larger amines are water insoluble since the nonpolar alkyl portion is too large to dissolve in the polar water solvent. Amines as Bases Like ammonia (NH3), amines are bases; that is, they are proton acceptors. • When an amine is dissolved in water, an acid–base reaction occurs: the amine accepts a proton from H2O, forming its conjugate acid, an ammonium ion, and water loses a proton, forming hydroxide, –OH. • While amines are more basic than other families of organic compounds, they are weak bases compared to inorganic bases like NaOH. Reaction of Amines with Acids Amines also react with acids such as HCl to form water-soluble salts. The lone pair of electrons from the amine nitrogen atom is always used to form a new bond to a proton from the acid. The amine [(CH3)3N] gains a proton to form its conjugate acid, an ammonium cation [(CH3)3NH+]. A proton is removed from the acid (HCl) to form its conjugate base, the chloride anion (Cl–). In an acid–base reaction of an amine, the amine nitrogen always forms a new bond to a proton forming an ammonium ion. Ammonium Salts When an amine reacts with an acid, the product is an ammonium salt: the amine forms a positively charged ammonium ion and the acid forms an anion. • To name an ammonium salt, change the suffix -amine of the parent amine from which the salt is formed to the suffix ammonium. Then add the name of the anion. Name the ammonium salt: (CH3)3NH+ CH3COO− Answer: trimethylammonium acetate Ammonium Salts Ammonium salts are ionic compounds, and as a result: • Ammonium salts are water-soluble solids. • In this way, the solubility properties of an amine can be changed by treatment with acid. • For example, octylamine has eight carbons, making it water insoluble. • Reaction with HCl forms octylammonium chloride. This ionic solid is now soluble in water. Ammonium Salts as Useful Drugs Many amines with useful medicinal properties are sold as their ammonium salts. Since the ammonium salts are more water soluble than the parent amine, they are easily transported through the body in the aqueous medium of the blood. • Diphenhydramine is a 3° amine that is sold as its ammonium salt. Its salt is formed by treating diphenhydramine with HCl (diphenhydramine hydrochloride), is an over-the-counter antihistamine that is used to relieve the itch and irritation of skin rashes and hives. Amides Amides contain a carbonyl group bonded to a nitrogen atom. The N atom of an amide may be bonded to other hydrogen atoms or alkyl groups. Amides are classified as 1°, 2°, or 3° depending on the number of carbon atoms bonded directly to the nitrogen atom. A primary (1°) amide contains one C—N bond. A 1° amide has the structure RCONH2. A secondary (2°) amide contains two C—N bonds. A 2° amide has the structure RCONHR'. A tertiary (3°) amide contains three C—N bonds. A 3° amide has the structure RCONR'2. Naming an Amide In the IUPAC system, amides are identified by the suffix -amide. Naming an Amide A 2° or 3° amide has two parts to its structure: the RCO– group that contains the carbonyl and one or two alkyl groups bonded to the nitrogen atom. To name a 2° or 3° amide, • Name the alkyl group (or groups) bonded to the N atom of the amide. Use the prefix “N-” preceding the name of each alkyl group. • Name the RCO– group with the suffix -amide. Name the following amide: HCONHCH2CH3. Answer: N-ethylformamide Physical Properties 1° and 2° amides have higher boiling points and melting points than esters and 3° amides of comparable size. Amides having fewer than six carbons are soluble in water. Higher molecular weight amides are insoluble in water because the nonpolar portion of the molecule, the C—C and C—H bonds, gets larger than the polar carbonyl group. Amide Hydrolysis Treatment of an amide (RCONHR') with water in the presence of an acid catalyst (HCl) forms a carboxylic acid (RCOOH) and an ammonium salt (R'NH3+Cl–). Amide Hydrolysis Amides are also hydrolyzed in aqueous base to form carboxylate anions and a molecule of ammonia (NH3) or amine. Interesting Amines and Amides Caffeine and nicotine are widely used stimulants of the central nervous system that contain nitrogen atoms in rings. Caffeine and nicotine, like the amines, are alkaloids, naturally occurring amines derived from plant sources. Epinephrine and Related Compounds Epinephrine, or adrenaline as it is commonly called, is an amine synthesized in the adrenal glands from norepinephrine (noradrenaline). Epinephrine and Related Compounds When an individual senses danger or is confronted by stress, the hypothalamus region of the brain signals the adrenal glands to synthesize and release epinephrine, which enters the bloodstream and then stimulates a response in many organs. • Epinephrine synthesis occurs in the interior of the adrenal gland. Epinephrine release causes; increase in heart rate, increase in blood pressure, increase in glucose synthesis, dilation of lung passages. Epinephrine and Related Compounds The search for drugs that were structurally related to epinephrine but exhibited only some components of its wide range of biological activities led to the discovery of some useful medications. Both albuterol and salmeterol dilate lung passages; that is, they are bronchodilators. They do not, however, stimulate the heart. This makes both compounds useful for the treatment of asthma. Penicillin The antibiotic properties of penicillin were first discovered in 1928 by Sir Alexander Fleming, who noticed that a mold of the genus Penicillium inhibited the growth of certain bacteria. The penicillins are a group of related antibiotics. All penicillins contain two amide units. One amide is part of a four-membered ring called a 𝛃-lactam. The second amide is bonded to the four-membered ring. Penicillin The first penicillin to be discovered was penicillin G. Amoxicillin is another example in common use today. Penicillin interferes with the synthesis of the bacterial cell wall. Thiols and Sulfides Introduction, Reactions of Thiols, Sulfides, Dimethyl Sulfoxide Introduction Thiols contain a sulfhydryl group (SH – mercapto group) bonded to a tetrahedral carbon. The sulfur atom in thiols have two lone pairs of electrons, so each heteroatom is surrounded by eight electrons. Thiols differ from alcohols in one important way. They contain no O—H bonds, so they are incapable of intermolecular hydrogen bonding. This gives thiols lower boiling points and melting points compared to alcohols having the same size and shape. The most obvious physical property of thiols is their distinctive foul odor. 3-Methyl-1-butanethiol [(CH3)2CHCH2CH2SH] is one of the main components of the defensive spray of skunks. Reactions of Thiols Thiols undergo one important reaction: thiols are oxidized to disulfides, compounds that contain a sulfur–sulfur bond. This is an oxidation reaction because two hydrogen atoms are removed in forming the disulfide. Reactions of Thiols Disulfides can also be converted back to thiols with a reducing agent. The symbol for a general reducing agent is [H], since hydrogen atoms are often added to a molecule during reduction. Reactions of Thiols The chemistry of thiols and disulfides plays an important role in determining the properties and shape of some proteins. For example, α-keratin, the protein in hair, contains many disulfide bonds. Straight hair can be made curly by cleaving the disulfide bonds in α-keratin, then rearranging and re-forming them. To make straight hair curly, the disulfide bonds holding the protein chains together are reduced. This forms free SH groups. The hair is turned around curlers and then an oxidizing agent is applied. This reforms the disulfide bonds to the hair, now giving it a curly appearance. Sulfides In organic chemistry, "sulfide" usually refers to the linkage C—S—C, although the term thioether is less ambiguous. For example, the thioether dimethyl sulfide is CH3—S—CH3. Methionine, an amino acid that also participates in the building of the proteins is the most important thioether. Methionine (Met or M) Sulfides A disulfide bond is a single covalent bond derived from the coupling of thiol groups. The linkage is also called an SS-bond or disulfide bridge. The overall connectivity is therefore C— S—S—C. Acid compounds containing (SOH) groups are distinguished by the number of oxygen atoms within that group as follows: • RSOH selfenic acid • RSO2H sulfinic acid • RSO3H sulfonic acid Sulfides A sulfenic acid is a sulfur compound and oxoacid with the general formula RSOH. Sulfenic acids are generally unstable. Sulfinic acids are oxoacids of sulfur with the structure RSO(OH). Sulfonic acid is a hypothetical acid with formula H-S(=O)2-OH. • This compound is a tautomer of sulfurous acid HO-S(=O)-OH, but less stable, and would likely convert to that very quickly if it were formed. • Although this compound is unimportant, there are many derived compounds, with formula R-S(=O)2-OH for various R. These may then form salts or esters, called sulfonates. • Sulfonic esters are considered good leaving groups in nucleophilic aliphatic substitution. Dimethyl Sulfoxide Dimethyl sulfoxide (DMSO) is the chemical compound with the formula (CH3)2SO. This colorless liquid is an important polar aprotic solvent that dissolves both polar and nonpolar compounds and is miscible in a wide range of organic solvents as well as water. • Polar aprotic solvents are solvents that lack an acidic hydrogen. It has a distinctive property of penetrating the skin very readily, allowing the handler to taste it. Some describe it as an "oyster-like" taste, others claim it tastes like garlic. Dimethyl Sulfoxide DMSO is an important polar aprotic solvent. It is less toxic than other members of this class such as dimethylformamide. Because of its excellent solvating power, DMSO is frequently used as solvent for chemical reactions involving the reactions of salts. DMSO is used in the PCR reaction to inhibit secondary structures in the DNA template or the DNA primers. However, use of DMSO in PCR increases the mutation rate. Dimethyl Sulfoxide In cryobiology DMSO has been used as a cryoprotectant and is still an important constituent of cryoprotectant vitrification mixtures used to preserve organs, tissues, and cell suspensions. It is particularly important in the freezing and long-term storage of embryonic stem cells and hematopoietic stem cells, which are often frozen in a mixture of 10% DMSO and 90% fetal calf serum. Dimethyl Sulfoxide In the medical field DMSO is predominantly used as a topical analgesic, a vehicle for topical application of pharmaceuticals, as an anti-inflammatory and an antioxidant. Because DMSO easily penetrates the skin, substances dissolved in DMSO may be quickly absorbed. For instance, a solution of sodium cyanide in DMSO can cause cyanide poisoning through skin contact. DMSO by itself has low toxicity. • Dimethyl sulfoxide can produce an explosive reaction when exposed to acid chlorides. Recently, it was found that DMSO waste disposal into sewers can cause environmental odor problems in cities. • Waste water bacteria transform DMSO under hypoxic (anoxic) conditions into dimethyl sulfide (DMS) that is slightly toxic and has a strong disagreeable odor, similar to rotten cabbage. Reference Books Eren, M. 2015. Organic Chemistry Lecture Notes (in TR) • Thanks very much, Prof. Dr. Meryem EREN. Fromm JR. "http://www.3rd1000.com/chem301/chem301x.htm" Date of access: 14.12.2016 Smith JG (2010). Organic Chemistry, 3rd Edition, McGraw-Hill. Smith JG (2012). General, Organic, & Biological Chemistry 2nd Edition, McGraw-Hill.