* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Capillary Electrophoresis of Proteins
Circular dichroism wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Protein purification wikipedia , lookup
Sample preparation in mass spectrometry wikipedia , lookup
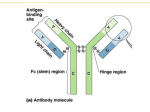
Capillary Electrophoresis of Proteins SDS Capillary Gel Electrophoresis SDS-CGE Outline • CE-SDS Gel Analysis – Description of Technique – Method Development Tips • PA800 plus kits – SDS-MW – IgG Purity & Heterogeneity CE-SDS Gel Analysis • Capillary Gel Electrophoresis – Molecular sieving • CE separations are based on analytes’ differential migration through a gel matrix. – Charge/mass ratio • Requires same charge on all proteins and peptides – SDS or sodium dodecyl sulfate • Anionic detergent • Complexes with the proteins and peptides at a constant ratio of 1:1.4 CE-SDS-Gel Analysis Molecular Sieving Detector capillary filled with entangled polymer solution replaceable sieving matrix improved reproducibility Separation Mechanism protein forms complex with SDS complexes have same charge to size ratio separation solely based on the hydrodynamic size proteins migrate in order of increasing size Detector ProteomeLabTM SDS-Gel Analysis Chemistry for the PA800 Plus SDS-MW Analysis IgG Purity & Heterogeneity Assay ProteomeLabTM SDS-MW Analysis Resolving Power ProteomeLabTM IgG Purity & Heterogeneity Resolving Power ProteomeLabTM SDS-MW Analysis Kit Components • Bare Fused Silica Capillary – 50 µm ID X 57 cm • SDS-MW Gel buffer • proprietary formulation • Store at Room Temperature • SDS Sample buffer • 100 mM TrisHCl pH 9.0, 1% SDS • Store at Room Temperature • SDS Protein Sizing Standards • 10-225 kDa (16 mg/ml) • • 10 kDa Internal Size Standard (5 mg/ml) Acid Wash • 0.1N HCl • Basic Wash • 0.1N NaOH ProteomeLabTM SDS-MW Kit Instrument Setup • Detection – PDA @ 220 nm – 200 m aperture • Bare Fused Silica Capillary – – – – 50 m ID 30.2 cm total length (20 cm length to detector) Capillary ends should be clean, not jagged. Capillary cut should be perpendicular to capillary length, not angled • Interface area should be cleaned before run and after run. • Interface area must be cleaned after 24 hours of Operation ProteomeLabTM SDS-MW Kit Size Standard Preparation • Remove from refrigerator and allow sit for 15 minutes at room temperature • Mix Well by inverting and then centrifuge briefly • Pipette 10 l of Size Standard into micro vial • Add 85 l Sample Buffer • Add 2 l 10 kDa Internal Standard • Add 5 l 2-mercaptoethanol • Cap vial and mix thoroughly • Heat 3 min at 100 C • Cool in Room Temperature water bath for 5 minutes. • Pipette 100 l into 200 l pcr vial ProteomeLabTM SDS-MW Kit Protein Sample Preparation • Final protein concentration 0.2 to 2 mg/ml • Final salt concentration < 50 mM • If higher reduced loading efficiency • Desalt sample or use pressure injection • Reduced Sample • Dilute sample with at least 50 µl Sample Buffer to a total volume of 95 µl • Add 2 µl 10kD Internal Standard • Add 5 µl 2- mercaptoethanol • Cap tightly and mix thoroughly • Heat 3 minutes at 100°C • Cool in Room Temperature water bath for 5 minutes. • Pipette 100 l into pcr vial ProteomeLabTM SDS-MW Kit Protein Sample Preparation • NonReduced Sample • Alkylate minimizes heterogeneity generated by heating • Dilute sample with at least 50 µl Sample Buffer to a total volume of 95 µl (0.2 to 2 mg/ml) • Add 2 µl 10kD Internal Standard • Add 5 µl 250 mM Iodoacetimide • Cap tightly and mix thoroughly • Heat 3 minutes at 70°C • Cool in Room Temperature water bath for 5 minutes. • Pipette 100 l into pcr vial ProteomeLabTM SDS-MW Kit • Good Resolving Power • Assay Precision better than 1% for Mobility and Area% • Linearity Range 0.2 – 2mg/mL of Total Protein ProteomeLabTM SDS-MW Kit Resolving Power ProteomeLab SDS-MW Kit Linearity For Sizing Determination ProteomeLab SDS-MW and IgG Purity Impurity Determination ProteomeLabTM IgG Kit • Separation Mechanism – Capillary Gel Electrophoresis – Identical to SDS-MW Separation • Gel Matrix – Identical to SDS-MW • Method optimized for separation of reduced and non reduced IgG. • Includes system suitability standard with a specified amount of non glycosylated heavy chain. ProteomeLabTM IgG Kit Kit Components • Bare Fused Silica Capillary – 50 µm ID X 57 cm • SDS-MW Gel buffer • proprietary formulation • Store at Room Temperature • SDS Sample buffer • 100 mM TrisHCl pH 9.0, 1% SDS • Store at Room Temperature • • • IgG Control Standard 10 kDa Internal Size Standard (5 mg/ml) Acid Wash • 0.1N HCl • Basic Wash • 0.1N NaOH ProteomeLabTM IgG Kit Instrument Setup • Detection – PDA @ 220 nm – 200 m aperture • Bare Fused Silica Capillary – – – – 50 m ID 30.2 cm total length (20 cm length to detector) Capillary ends should be clean, not jagged. Capillary cut should be perpendicular to capillary length, not angled • Interface area should be cleaned before run and after run. • Interface area must be cleaned after 15 hours of operation ProteomeLabTM IgG Kit IgG Control Preparation • Upon receiving aliquot 95 µl into 0.5 ml microtubes – Store at -20 • • • • • • • • Thaw 1 95 µl aliquot Add 2 µl 10 kDa Internal Standard Add 5 µl 2-mercaptoethanol Cap vial and mix thoroughly Centrifuge at 300 g for 1 minutes Heat 10 min at 70 C Cool in Room Temperature water bath for 5 minutes. Pipette 100 µl into pcr vial ProteomeLabTM IgG Kit IgG Sample Preparation • Final salt concentration < 50 mM – If higher reduced loading efficiency – Desalt sample or use pressure injection • Reduced Sample – – – – – – – – 100 µg IgG to 50 – 95 µl Sample Buffer to a total volume of 95 µl Add 2 µl 10kDa Internal Standard Add 5 µl 2-mercaptoethanol Cap tightly and mix thoroughly Centrifuge 300g for 1 minute Heat 10 minutes at 70°C Cool in Room Temperature water bath for 5 minutes. Pipette 100 µl into pcr vial ProteomeLabTM IgG Kit Protein Sample Preparation • NonReduced Sample • Alkylate minimizes fragmentation • 100 µg IgG to 50 – 95 µl Sample Buffer to a total volume of 95 µl • Add 2 µl 10kDa Internal Standard • Add 5 µl 250 mM Iodoacetimide • Cap tightly and mix thoroughly • Centrifuge 300g for 1 minute • Heat 10 minutes at 70°C • Cool in Room Temperature water bath for 5 minutes. • Pipette 100 µl into pcr vial ProteomeLabTM IgG Kit Methods • Run Preconditioning Method • Run Sequence of 24 injections – First run is IgG Control Standard as a system suitability – 24 is maximum number of injections due to polymer build up on interface block – Buffer vials incremented every 8 runs • Necessary due to polymer build up on the vial cap ProteomeLabTM IgG Kit What to expect? Goodness of fit: 0.999332 ProteomeLabTM IgG Kit Resolving Power PDA - 220nm IgG reduce PDA - 220nm IgG reduce 0.06 0.06 0.05 0.05 0.04 0.04 0.03 0.03 0.02 0.02 0.01 0.01 0.00 0.00 8 10 12 14 16 18 20 Minutes 22 24 Human IgG - Reduced: Reproducibility 26 28 AU AU PDA - 220nm IgG reduce What is the difference between SDS-MW kit and IgG Purity kit? • METHODS – SDS-MW • Rinses and Gel fill once every 6 samples – IgG • Rinses and Gel fill prior to every injection • Size Standards for SDS MW Kit • IgG Control Standards for IgG Purity Kit Tips and Tricks for SDS Gel Analysis • Recommended protein concentration is 1 mg/ml – Higher concentrations can result in tailing peaks due to insufficient protein binding • Salt concentration should be <50 mM in final dilution – Insufficient sample loading • Gel must be degassed and at room temperature – Spikes present on the baseline Tips and Tricks for SDS Gel Analysis • Buffer Vial Caps cannot be reused – – – – – Dried polymer is very difficult to remove Caps degrade over time and with washing Result is poor injection reproducibility Broken capillaries Pressure failures Tips and Tricks for SDS Gel Analysis • Interface block must be cleaned before and after every run – Polymer precipitates on interface block – Do not fill buffer vials with more than 1.5 ml of gel which will minimize polymer on interface block – Lack of cleaning causes broken capillaries and pressure failures