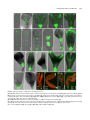
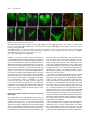
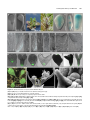
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download POPCORN Functions in the Auxin Pathway to Regulate
Survey
Document related concepts
Transcript
The Plant Cell, Vol. 23: 4348–4367, December 2011, www.plantcell.org ã NRC Canada 2011 POPCORN Functions in the Auxin Pathway to Regulate Embryonic Body Plan and Meristem Organization in Arabidopsis W OA Daoquan Xiang,a,1 Hui Yang,a,1 Prakash Venglat,a,1 Yongguo Cao,a Rui Wen,b Maozhi Ren,a Sandra Stone,a Edwin Wang,c Hong Wang,b Wei Xiao,b Dolf Weijers,d Thomas Berleth,e Thomas Laux,f Gopalan Selvaraj,a and Raju Datlaa,2 a Plant Biotechnology Institute, National Research Council Canada, Saskatoon, Saskatchewan S7N 0W9, Canada of Saskatchewan, Health Sciences Building, Saskatoon, Saskatchewan S7N 5E5, Canada c Biotechnology Research Institute, National Research Council Canada, Montreal, Quebec H4P 2R2, Canada d Wageningen University, Laboratory of Biochemistry, 6703 HA Wageningen, The Netherlands e Department of Botany, University of Toronto, Toronto, Ontario M5S 3B2, Canada f BIOSS, University of Freiburg, 79104 Freiburg, Germany b University The shoot and root apical meristems (SAM and RAM) formed during embryogenesis are crucial for postembryonic plant development. We report the identification of POPCORN (PCN), a gene required for embryo development and meristem organization in Arabidopsis thaliana. Map-based cloning revealed that PCN encodes a WD-40 protein expressed both during embryo development and postembryonically in the SAM and RAM. The two pcn alleles identified in this study are temperature sensitive, showing defective embryo development when grown at 228C that is rescued when grown at 298C. In pcn mutants, meristem-specific expression of WUSCHEL (WUS), CLAVATA3, and WUSCHEL-RELATED HOMEOBOX5 is not maintained; SHOOTMERISTEMLESS, BODENLOS (BDL) and MONOPTEROS (MP) are misexpressed. Several findings link PCN to auxin signaling and meristem function: ectopic expression of DR5rev:green fluorescent protein (GFP), pBDL:BDLGFP, and pMP:MP-b-glucuronidase in the meristem; altered polarity and expression of pPIN1:PIN1-GFP in the apical domain of the developing embryo; and resistance to auxin in the pcn mutants. The bdl mutation rescued embryo lethality of pcn, suggesting that improper auxin response is involved in pcn defects. Furthermore, WUS, PINFORMED1, PINOID, and TOPLESS are dosage sensitive in pcn, suggesting functional interaction. Together, our results suggest that PCN functions in the auxin pathway, integrating auxin signaling in the organization and maintenance of the SAM and RAM. INTRODUCTION Embryogenesis in angiosperms begins with the division of the zygote into the precursor cells of the embryo proper and the suspensor. Subsequently, the establishment of the apical-basal axis, radial organization of tissues, and initiation of cotyledons and apical meristems together constitute the embryonic body plan (De Smet et al., 2010). In recent years, Arabidopsis thaliana has been investigated as a model plant to address the functions of key genetic factors that contribute to embryogenesis. Auxin transport and signaling play critical roles in the establishment of embryonic body plan (Friml et al., 2003; Jenik et al., 2007). During the transition from globular to heart embryo, the apical domain is partitioned to form the shoot apical meristem (SAM) and the 1 These authors contributed equally to this work. correspondence to [email protected]. The author responsible for distribution of materials integral to the findings presented in this article in accordance with the policy described in the Instructions for Authors (www.plantcell.org) is: Raju Datla (raju. [email protected]). W Online version contains Web-only data. OA Open Access articles can be viewed online without a subscription. www.plantcell.org/cgi/doi/10.1105/tpc.111.091777 2 Address cotyledons, whereas the basal domain differentiates into the hypocotyl and the embryonic root apical meristem (RAM). Altered auxin signaling can result in apical patterning defects with cotyledons that are either fused, as seen in the long hypocotyl5 and hy5 homolog double mutant (Sibout et al., 2006), or completely absent, as seen in the auxin response factor (ARF) double mutant monopteros (mp) and nonphototropic hypocotyl4 (Hardtke et al., 2004). The gnom mutant, which is affected in auxin transport, fails to establish apical-basal patterning, resulting in a ball-like embryo with no cotyledons (Mayer et al., 1993). The products of PINFORMED (PIN) gene family members PIN1, PIN3, PIN4, and PIN7 are differentially localized in a polar manner to coordinate the transport of auxin during embryo patterning, and quadruple mutants of all four genes display filamentous embryos with apical-basal polarity defects (Friml et al., 2003). Polar targeting of PIN proteins in cells is regulated via phosphorylation by the kinase PINOID (PID) (Michniewicz et al., 2007). The CUP-SHAPED COTYLEDON (CUC) genes are involved in the partitioning of the apical domain of the embryo. In the cuc1 cuc2 double mutant, the lack of or a defect in the SAM, along with fused cotyledons, results in the termination of seedling development (Aida et al., 1997). CUC overexpression occurs in the pin-1 pid-2 double mutant, leading to the inhibition of cotyledon PCN Regulates Embryo and Meristem primordia initiation (Furutani et al., 2004). Furthermore, CUC genes have been shown to be essential for the expression of SHOOTMERISTEMLESS (STM) (Aida et al., 1999). In the root, auxin has been shown to play major roles in RAM establishment and function (Overvoorde et al., 2010). The auxin response regulators BODENLOS (BDL) and MP mediate auxin signaling and provide root patterning information by activating the PLETHORA (PLT) genes (Hamann et al., 2002; Nawy et al., 2008). The two PLT genes provide positional information to establish the root stem cell niche through the auxin pathway (Aida et al., 2004). The homeobox gene WUSCHEL-RELATED HOMEOBOX5 (WOX5) is expressed specifically in the quiescent center (QC) of the root, and wox5 mutants fail to maintain stem cells in the RAM (Sarkar et al., 2007). Auxin and cytokinin together play antagonistic roles in the SAM and RAM partly by regulating the expression of ARABIDOPSIS RESPONSE REGULATOR7 (ARR7) and ARR15 (Zhao et al., 2010). Proper organization and maintenance of both meristems depends on coordinated cell divisions that balance the number of stem cells and daughter cells that contribute to the initiation of organs. A number of transcription and signaling factors have been implicated in the functions of shoot and root meristems. These include homeobox genes STM and WUSCHEL (WUS) (Mayer et al., 1998; Lenhard et al., 2002), CLAVATA signaling factors (CLV1, CLV2, and CLV3) in the SAM (Fletcher et al., 1999; Schoof et al., 2000), and homeobox gene WOX5 and plantspecific AP2 domain-containing PLT transcriptional factors in the RAM (Aida et al., 2004; Sarkar et al., 2007). Despite the number of meristem regulatory genes that have been identified thus far, mutations in the majority of these genes (i.e., WUS, CLV, and STM) do not result in embryo lethality. This implies that, in addition to the possible existence of unknown genes that function both in the embryo patterning and meristem organization, there is a significant overlap and/or redundancy in the functions of these known genes. In this study, we report the identification of Arabidopsis POPCORN (PCN), which is involved in both embryo and meristem development. PCN encodes a putative WD-40 protein, and mutations in PCN perturb auxin polarity resulting in ectopic auxin maxima in the SAM. We show genetic interactions of PCN with WUS, BDL, MP, TOPLESS (TPL), PIN1, and PID affecting the embryonic and postembryonic development in Arabidopsis. RESULTS Map-Based Cloning of PCN, Which Encodes a WD-40–Related Protein We have determined global gene expression during embryo development in Arabidopsis (Xiang et al., 2011). These data sets provide a resource for further genetic interrogation. Genetic screens and targeted reverse genetic approaches have been successful in identifying key genes in Arabidopsis embryo development. To identify genes that are required for Arabidopsis embryogenesis, we analyzed T-DNA lines with insertions in selected candidate genes that are specifically or differentially expressed during zygote to globular stages of embryo develop- 4349 ment based on our global gene expression data sets (Xiang et al., 2011). One of the lines from this screen showed early embryo lethality among the segregating embryos with a phenotype resembling popped maize (Zea mays), which we named popcorn (pcn). The pcn mutation segregated as a single, recessive allele. However, molecular analysis revealed that this T-DNA insertion was not linked to the mutant embryo phenotype. We therefore used a map-based cloning approach to isolate the PCN gene. A tight linkage was observed with cleaved amplified polymorphic sequences (CAPS) markers at the genomic positions 4,144,848 (SspI enzyme digestion) and 4,369,443 (RsaI enzyme digestion) with no recombination observed in the analysis of 1340 chromosomes (670 plants). The strong globular stage defects in pcn mutant suggest that the expression of gene(s) in this locus is likely critical for embryo patterning and development. We reasoned that the expressed genes in this genomic region would likely include PCN. The 224-kb region between 4,144,848 and 4,369,443 on chromosome 4 contains 53 genes of which 44 encode transposable elements. Of the remaining nine loci, five are of unknown function and the other four are annotated as F-box (At4g07400), WD-40 (At4g07410), PQ loop repeat (At4g07390), and GTP binding (At4g07524). Because TPL is also a WD-40 repeat–containing protein that is critical for embryo patterning that involves specification of the apical-basal polarity and cotyledon formation (Long et al., 2006), we predicted that At4g07410 may be associated with the defective phenotype. At4g07410 also had the highest expression level among the 53 genes in the deduced 224-kb chromosome 4 region in our microarray data for wild-type globular embryos (Xiang et al., 2011). Sequencing of candidate genes in the mapped region of pcn identified a 35-bp deletion overlapping the 5th exon and 5th intron of At4g07410 genic region that would result in a premature stop codon of the predicted open reading frame (Figure 1A). At4g07410 encodes an 815–amino acid protein containing eight WD-40 repeats as determined by InterProScan (Zdobnov and Apweiler, 2001), six at the N terminus and two at the C terminus (Figure 1B). The premature stop codon in pcn-1 was deduced to produce a truncated protein of 240 amino acids. In Arabidopsis, PCN, a single-copy gene, shares 69% identity with At1g27470 at the amino acid level. Putative homologs of PCN are also present in other plant genera (Oryza, Sorghum, Populus, Ricinus, and Vitis), yeast, zebra fish, and humans (see Supplemental Figure 1 and Supplemental Data Set 1 online). In none of these cases is any function known. Using the At4g07410 sequence information, we screened for additional T-DNA insertional lines at this locus and identified a second allele of PCN, pcn-2 (Salk_022607), with an insertion in the 8th exon (Figure 1A). The pcn-2/+ plants segregated defective embryos similar in mutant phenotype to those observed in pcn-1. Heterozygous pcn-1/+ or pcn-2/+ plants did not exhibit any developmental defects other than producing 25% arrested and 75% normal embryos (n = 1028 for pcn-1, n = 1120 for pcn-2), indicating that pcn-1 and pcn-2 mutations are recessive. To confirm that the mutant embryo phenotypes observed in pcn-1 and pcn-2 are caused by the lesions in the At4g07410 locus, we introduced two constructs into heterozygous mutant pcn-1 and pcn-2 plants. The first construct contained the At4g07410 genomic fragment, and the second was the PCN cDNA (representing 4350 The Plant Cell Figure 1. Molecular Characterization of the PCN Locus. (A) A 35-bp deletion (open square) was detected in the pcn-1 mutant, spanning 27 bp of exon 5 and 8 bp of intron 5 (genomic position 4203956 to 4203991). In the pcn-2 mutant, the T-DNA insertion (triangle) was in exon 8 (position 4202286). The boxes indicate the exons and the lines indicate introns. (B) PCN encodes an 815–amino acid protein containing eight WD-40 repeats (gray boxes). the reading frame of 815 amino acids) under the control of the putative PCN promoter. Transformation of both constructs fully complemented the homozygous pcn mutant embryo phenotype. These observations confirmed that PCN, a WD-40 repeat–containing protein, is required for embryo development in Arabidopsis. Because the other PCN homolog, At1g27470, is expressed during embryo development in Arabidopsis (Xiang et al., 2011), we sought to assess any functional similarity of At1g27470 with PCN. No knockout lines for this gene are currently available in SALK or other collections. To test if this gene can complement pcn, we generated a gene construct using the At1g27470 protein coding sequence and expressed it under the control of PCN promoter (to achieve expression in the PCN domain) and introduced this construct into the pcn background. The rationale for this approach is that if At1g27470 shares functional domains in its protein with PCN, complementation (partial or full) of embryo phenotype is expected. However, our transgenic experiments did not show any complementation, suggesting that At1g27470 does not share similar functions with PCN in Arabidopsis. Together, these results confirm that PCN is a single-copy putative WD-40 domain–encoding gene essential for embryo development in Arabidopsis. Embryo Patterning Defects in pcn Mutants We investigated the developmental aspects of pcn using the pcn-1 allele (unless otherwise specified). Homozygous pcn seeds obtained from a pcn/+ mother plant that was grown at 228C did not germinate. However, when cultured in vitro with auxin and cytokinin (Wu et al., 1992), these mutant embryos were able to form calli from which fertile plants could be regenerated. These homozygous pcn plants upon flowering produced embryos that were all defective. As this provided an advantage over the segregating heterozygote, we have used these nonsegregating homozygous lines for more detailed developmental studies (see Supplemental Table 1A online; Figures 2 and 3). During wild-type embryogenesis, the zygote elongates and divides asymmetrically, generating a smaller apical and a larger basal cell (Figures 3A and 3K). The first two divisions of the apical cell occur longitudinally, giving rise to the four-cell embryo proper (Figures 3C and 3M), followed by a round of horizontal divisions to establish the eight-cell embryo. Tangential divisions separate the protoderm from the inner cells at the dermatogen stage. Later, the globular embryo initiates two cotyledonary primordia and a SAM. The descendants of the basal cell divide horizontally to generate a column of cells, the extraembryonic suspensor. Only the uppermost cell of the basal lineage, the hypophysis, contributes to the embryo. After a transverse division, the apical daughter cell of the hypophysis, the lens-shaped cell, gives rise to the QC of the root meristem, whereas the basal daughter cell forms the columella stem cells (Jurgens and Mayer, 1994). Development of pcn embryos was similar to the wild type until the two-cell embryo stage (Figures 3A and 3C versus Figures 3K and 3M). Subsequently, with delayed apical cell divisions, more suspensor cells were observed when compared with the wild type (Figures 3L to 3V). Occasionally, abnormal vertical divisions in suspensor cells (;2%) (Figure 3P), and octant embryo proper with twice the number of cells than in the wild type were also observed in pcn (< 5%) (Figures 3Q and 3R). From the dermatogen stage and onwards, the pcn embryo proper displayed several defects (Figures 3Q to 3Z). Abnormal and delayed divisions were observed in the apical domain (Figures 3P to 3R), resulting in four kinds of phenotypes (Figures 2E to 2M): No cotyledon (36.4% in pcn-1; 33.2% in pcn-2) (Figures 2E to 2G), one cotyledon (29.8% in pcn-1; 31.3% in pcn-2) (Figures 2H and 2I), unequal splayed two cotyledons (31% in pcn-1; 32.4 in% pcn-2), (Figures 2J to 2L) and three cotyledons (2.9% in pcn-1; 3.2% in pcn-2) (Figure 2M; see Supplemental Table 1A online). Since pcn-1 embryos displayed stronger defects than pcn-2, further analysis was done with pcn-1. The SAM region in the majority of arrested embryos was enlarged, and the cotyledons lacked bilateral symmetry and appeared more radialized (Figures 2I to 2M). In the basal domain of pcn embryos, the lens-shaped cell derivatives were smaller than in the wild type (Figures 3I, 3J, 3Y, and 3Z), and the lower hypophyseal cell derivatives divided less frequently, resulting in aberrant columella organization (Figures 3J and 3Y). Together, the results show that loss of PCN activity affects the development of both the embryo proper and the suspensor. Elevated Temperature Rescues pcn Embryo Lethality While testing the growth of pcn plants at different temperatures, we noticed that the defects in pcn embryo development were strongly alleviated after shifting the regenerated homozygous plants from 22 to 298C at the onset of bolting. The majority of the pcn embryos produced at 298C had two wild-type-like cotyledons (83%; see Supplemental Table 1A online) and developed PCN Regulates Embryo and Meristem 4351 Figure 2. Scanning Electron Microscopy Analysis of pcn Mutant Embryos. (A) to (D) Wild-type embryos at early heart (A), mid-heart (B), torpedo (C), and bent (D) stages. (E) to (M) pcn embryos arrested at various stages of development: globular embryo with defects in the apical and basal domains ([E] and [F]) and partially differentiated basal domain (G); one-cotyledon embryo ([H] and [I]); two-cotyledon embryos showing various degrees of abnormal cotyledon growth ([J] to [L]); three-cotyledon embryo (M). pcn embryos have an enlarged SAM ([I] to [M]). Bars = 10 mm in (A) and (B) and 100 mm in (C) to (M). into normal mature embryos. The remaining embryos had no cotyledon (<5%), one cotyledon (10%), and three cotyledons (2%) (see Supplemental Table 1A online). Unlike seeds from pcn homozygous plants grown at 228C, 95% of pcn seeds produced at 298C were able to germinate on 0.53 Murashige and Skoog (MS) medium without the requirement of exogenous hormones and produced fertile plants (see Supplemental Table 1B online; Figure 4B). We took advantage of the temperature sensitivity characteristic of this mutant and propagated pcn and genetic combinations involving pcn by first growing the plants at 228C and then shifting them to 298C at bolting unless specified otherwise. To determine at which embryo stage PCN function is essential, pcn plants were grown at 228C, and flower buds were first emasculated and then hand-pollinated with pcn pollen. A few of the developing siliques were dissected to determine the devel- opmental stage of the embryos. The plants were then shifted to 298C to complete seed development. Mature seeds were either dissected to assess the embryo phenotypes or geminated on 0.53 MS medium. Our results from this study showed that the shift to 298C must occur at or prior to the globular stage in order to rescue the pcn embryo development (see Supplemental Table 1B online). A shift in temperature postglobular stage of embryo development could not suppress embryo lethality. These observations further suggest that PCN function is required already for early phases of embryo development in Arabidopsis. pcn Mutations Impact Postembryonic Development To address the postembryonic functions of PCN, 298C temperature-rescued pcn mutant seeds were germinated and 4352 The Plant Cell Figure 3. pcn Mutants Undergo Aberrant Division Patterns in Early and Mid-Stage Embryogenesis. Nomarski images of whole-mount cleared seeds containing the wild type ([A] to [J]) and pcn ([K] to [Z]) at the one-cell stage ([A] and [K]), two-cell stage ([B] and [L]), quadrant stage ([C] and [M]), octant stage ([D] and [N]), dermatogen stage ([E], [O], [P], and [Q]), globular stage ([F], [G], and [R] to [W]), triangular stage ([H] and [X]), and heart stage ([I], [J], [Y], and [Z]). Delayed apical cell division in the pcn mutant was observed earliest in quadrant stage (M) and in later embryo developmental stages ([N], [P], [T], and [V]). In the apical cell, abnormal planes of cell division were first observed at the octant stage (N). During later developmental stages ([O] to [Z]), delayed and altered cell divisions resulted in an abnormal lens-shaped cell and hypophyseal cell ([W] to [Z]; bottom panel) and defective cotyledon primordia specification ([Y] and [Z]). Bars = 10 mm. plant development was analyzed at 228C growth conditions. pcn mutant rosettes were smaller with narrower leaf shape (Figure 4B) and abnormal vein patterns (Figure 4D) compared with the wild type (Figures 4A and 4C). Flowering pcn plants were shorter and bushier than the wild type. Examination of pcn roots revealed abnormal division patterns in the QC and columella cells of the root cap, and root growth was retarded compared with the wild type (Figures 4F to 4H). Lugol staining for starch granules as a marker for columella cell differentiation revealed defects in the root cap cells in addition to the RAM defects observed in pcn (Figure 4H). Furthermore, the expression of the QC marker WOX5 was expanded in pcn roots (Figures 6N and 6O). These observations suggest that PCN function is also required after germination for several processes associated with meristems both in root and shoot development. PCN Encodes a Nuclear Protein That Is Expressed Broadly in the Embryo Proper and in Postembryonic Meristems To study PCN expression and subcellular localization in detail, two different translational fusions of PCN and green fluorescent protein (GFP) coding regions were expressed under the control of 1.9-kb PCN putative promoter region, one with PCN cDNA (pPCN: cPCN-GFP) and the other with PCN genomic fragment (containing introns; gPCN-GFP) (Figure 5). Independent transgenic lines of both constructs displayed identical expression patterns. For brevity, we will focus on results from the gPCN-GFP reporter. Our earlier study on global gene expression patterns during Arabidopsis embryo development (Xiang et al., 2011) showed that gPCN-GFP is expressed in the ovule but not in the pollen and, after fertilization, is expressed in the zygote, suggesting maternal specificity. During early phases of embryogenesis, the gPCN-GFP signal was detected in all the cells of the embryo proper and the suspensor until the 32-cell embryo stage (Figures 5A and 5B). Subsequently, gPCN-GFP expression was not detectable in the lower suspensor cells, and by the heart stage, the signal was completely absent in the suspensor (Figures 5D and 5E) but remained detectable in the embryo proper (Figures 5E and 5F). In seedlings, gPCN-GFP was detected in the SAM and leaf primordia (Figure 5G) but not in the hypocotyl (Figure 5H). In the root, gPCN-GFP is expressed in the root meristem (Figures 5I and 5J). gPCN-GFP is also expressed in pericycle cells, both quiescent (Figure 5K) and actively dividing (Figures 5K and 5L), that gave rise to the lateral roots and the expression continued throughout the development of lateral root primordia (Figures 5M and 5N). These results indicate that PCN is broadly expressed in the embryo proper but after germination is predominantly expressed in the SAM, RAM, and in cells capable of proliferation. PCN Regulates Embryo and Meristem 4353 Figure 4. pcn Mutant Development Is Affected Postembryonically. (A) and (B) Three-week-old rosette of the wild type (A) and pcn (B). The pcn rosette is smaller than that of the wild type. (C) and (D) Cleared fourth leaf of the wild type (C) and pcn (D). The pcn leaf shows reduced venation compared with that of the wild type. (E) to (H) Cleared and lugol-stained root apices of the wild type (WT) ([E] and [H]) and pcn ([F] to [H]). pcn root meristems were smaller ([F] and [G]) with defective columella cell differentiation of the root cap compared with the wild type ([E] and [H]). Bars = 1 cm in (A) to (D) and 100 mm in (E) to (H). Mutation of PCN Perturbs the Expression Domains of SAM-Organizing Genes To better understand the SAM defects in pcn, we investigated the expression patterns of well-characterized SAM organizing genes using whole-mount embryo in situ hybridization and reporter genes. In the wild-type shoot meristem, WUS and CLV3 expression domains mark the three apical layers of stem cells and the underlying organizing center, respectively (Figures 6A and 6G) (Fletcher et al., 1999; Schoof et al., 2000). In pcn embryos, CLV3 was expressed in an enlarged domain encompassing almost the whole apex and was also ectopically expressed in the cotyledons (Figures 6H and 6I). Compared with the wild type, WUS mRNA expression had expanded into the L1 and L2 layers of the SAM and toward the inner layers of the cotyledons (Figure 6B). Expression of STM marks SAM cells that do not enter into lateral organ formation. During wild-type embryogenesis, STM is first expressed in a band of cells around the central apical region of late globular stage embryos. During the transition to heart stage, the expression becomes restricted to the central core of the SAM (Figure 6C) (Long and Barton, 1998). In pcn, STM was variably expressed below the L3 layer of the SAM. In the majority of the embryos examined (30 out of 44), STM expression was detected in a large apical region of the embryo (Figure 6D). CLV1 is expressed in the L2 and L3 layers of the wild-type SAM (Long and Barton, 1998). In pcn, CLV1 expression was still limited to the L2 and L3 layers, but the expression domain had expanded to a slightly broader region compared with the wild type (Figures 6E and 6F). Furthermore, quantitative RT-PCR (qRT-PCR) results revealed that WUS, CLV3, CUC, and STM in the SAM are significantly upregulated in pcn embryos (see Supplemental Table 2 online). These results suggest that PCN function is required to restrict the expression of STM, WUS, and CLV genes within their respective specified boundaries in the meristem. PCN and WUS Interact Genetically To determine whether PCN genetically interacts with WUS, we analyzed pcn wus-1 double mutants. wus-1 embryos develop normally except for the absence of a shoot meristem in the seedlings after germination (Laux et al., 1996). Double heterozygous pcn/+ wus/+ plants produced homozygous recessive pcn wus double mutant embryos at a ratio of 1:15 that did not initiate cotyledon primordia and had seeds that did not germinate, even when the mother plant was grown at 298C. However, pcn wus-1/+ seeds germinated and produced plants with rosette leaves, albeit at a slower rate than the wild type. After bolting, however, the SAM terminated as pin-like structures with arrested lateral organ primordia and differentiation of the epidermal cells into trichomes near the SAM region, indicating haploinsufficiency for WUS in pcn background (Figures 7J to 7N). These observations indicate that wus embryos cannot complete their development unless PCN function is present. Whereas the lack of PCN can be remedied by high temperature insofar as embryo development is concerned, such a rescue requires WUS. These synergistic defects suggest partial compensatory functions for WUS and PCN during embryo and postembryonic development. Auxin Distribution and Response Are Altered in pcn Defective cotyledon patterning in pcn embryos raised the question whether auxin response and/or auxin transport were affected in pcn embryos. To address this question, we introduced 4354 The Plant Cell Figure 5. GFP Reporter–Based Expression Patterns and Nuclear Localization of PCN in Arabidopsis. (A) to (F) gPCN-GFP expression in embryos at octant stage (A), dermatogen stage (B), globular stage (C), triangular stage (D), heart stage (E), and torpedo stage (F). (A) to (E) show progressive loss of expression of gPCN-GFP in suspensor cells during embryo development. (G) and (H) gPCN-GFP is expressed in the SAM ([G], outlined in yellow) of the 5-d-old seedling and absent in hypocotyl (H). (I) and (J) gPCN-GFP is expressed in the RAM (I) and distal region of the root (J). The yellow boxed region in (K) shows gPCN-GFP expression in the dividing pericycle cells (L) that continues during early stages of lateral root formation ([M] and [N]). The 49,6-diamidino-2-phenylindole (red) and gPCN-GFP (green) colocalized in the nuclei of seedling RAM (O). Bars = 20 mm in (A) to (H), (J), and (L) to (O) and 100 mm in (I) and (K). the auxin response marker DR5rev:GFP, and as a readout of polarized auxin transport machinery, pPIN1:PIN1-GFP and pPIN7: PIN7-b-glucuronidase (GUS), into the pcn mutant. At 228C, DR5rev:GFP expression was observed throughout the embryo proper in pcn similar to the wild type up to the octant stage (Figures 8A, 8F, and 8G) (Friml et al., 2003). However, from 32cell globular stage onwards, the basal auxin maximum, which is restricted to the hypophyseal region in the wild type, expanded into the lower suspensor cells in arrested globular pcn embryos. The two auxin maxima corresponding to the two emerging cotyledon primordia in the wild type (Figures 8B to 8E) were absent in the pcn embryos that did not produce cotyledons (Figures 8I, 8J, and 8L). In pcn embryos that did form cotyledons, the auxin maxima correlated with the number of cotyledons (Figures 8M to 8P). In a few mutant embryos, several auxin maxima were also observed in the apical region of late torpedo stage embryos (Figure 8K). In contrast with DR5rev:GFP expression in the vascular procambial cells of later-stage wild-type embryos (Figure 8E), the GFP signal was discontinuous or absent in the differentiating procambial cells of pcn embryos (Figures 8K to 8P). Unlike the wild type, DR5rev:GFP was ectopically expressed in the SAM of pcn embryos (Figures 8P and 8U). Consistent with the phenotypic recovery, this ectopic DR5 expression was suppressed when pcn embryos were subjected to 298C treatment (Figures 8U and 8V). In the RAM, the expression became restricted to the columella region similar to the wild type (Figures 8Q and 8R). These results indicate that mislocalization of auxin maxima coincides with the defective phenotypes observed in pcn mutant embryos. In wild-type dermatogen stage embryos, PIN1-GFP is present in all the cell boundaries, whereas at the 32-cell stage, PIN1-GFP becomes restricted to the basal boundaries of subepidermal cells (Figure 9A). From late globular stage onwards, PIN1-GFP is localized in cotyledon primordia and in vascular initials as shown PCN Regulates Embryo and Meristem 4355 Figure 6. SAM and RAM Marker Gene Expression Is Altered in pcn Mutants. (A) to (F) Whole-mount embryo in situ hybridization of wild-type (WT) (A) and pcn (B) embryos with WUS probe, wild-type (C) and pcn (D) embryos with STM probe, and wild-type (E) and pcn (F) embryos with CLV1 probe. (G) to (I) pCLV3:CLV3-GUS expression in wild-type embryo (G), pcn embryos (H), and seedlings (I). (J) to (M) Whole-mount embryo in situ hybridization of wild-type (J) and pcn (K) embryos with MP probe and the wild type (L) and pcn (M) with PLT1 probe. (N) and (O) pWOX5-sv40:3GFP expression in wild-type root (N) and pcn mutants (O). All pcn embryos used were grown at 228C. Bars = 50 mm. previously (Friml et al., 2003) (Figures 9B to 9E). In pcn embryos arrested at globular stage, PIN1-GFP appeared in a broad central region of the embryo (Figures 9H and 9I), and the expression was seen in the vascular initials of the cotyledons and the hypocotyl of one cotyledon (Figure 9J) and two cotyledon embryos (Figures 9K and 9L). In contrast with the wild type, PIN1GFP also accumulated in the SAM region of the pcn embryos with two cotyledons (Figures 9K and 9L). While the PIN1 expression profile was relatively normal (see Supplemental Table 2 online), PIN1-GFP was not distributed in a polar manner in the majority of the cells of pcn mutant embryos (Figures 9H to 9L). Since polar localized PIN1 in the wild type can be rapidly internalized to the cytosol by brefeldin A (BFA) treatment (Geldner et al., 2001), we used BFA treatment along with the membrane stain FM4-64 (Vida and Emr, 1995) to examine the localization of PIN1-GFP in the cells of wild-type and pcn embryos. In the wild type, PIN1-GFP signal overlapped with the FM4-64 stain in the basal membrane of cells (Figure 9F), whereas with BFA treatment, PIN1-GFP became internalized in the cytosol (Figure 9G). However, in the pcn mutant embryos, the PIN1-GFP was inter- nalized to the cytosol both without and with BFA treatment (Figures 9M and 9N), suggesting that pcn mutant embryos are defective in polar localization of PIN1. PIN7 expression and localization is coordinated with PIN1 in the embryo proper to create auxin maxima in the hypophyseal cell at the 32-cell stage (Friml et al., 2003). We used pPIN7:PIN7GUS to compare its expression pattern in the pcn embryo and the wild type. In the wild type, PIN7-GUS was detected in the hypophyseal cell derivatives in the embryonic root meristem (Figures 10A and 10B) and the provascular cells in the developing hypocotyl (Figures 10A to 10C). In those pcn embryos that were arrested at early stages, the expression was present in the suspensor but absent from the hypophyseal cell derivatives of the root meristem (Figure 10F). Although the PIN7-GUS expression was present in the provascular cells of the developing pcn embryo, the signal appeared weaker and was restricted to fewer cells (Figures 10G to 10I) and was absent in the root meristem region (Figures 10H and 10I). In wild-type seedlings, PIN7-GUS was expressed in the root meristem and vascular cells of the root and cotyledons. By contrast, in pcn, the PIN7-GUS was weakly 4356 The Plant Cell Figure 7. Dosage-Dependent Phenotypes of pin1, pid-2, wus-1, and tpl-1 in the pcn Mutant. Heterozygous pin1, pid-2, wus-1, and tpl-1 in pcn homozygous recessive background have terminated meristems with a pin-like structure. (A) to (I) Pin-like structures in pcn pin1/+ ([A] and [E]), pin1 ([B] to [D]), pcn pid-2/+ ([F], [H], and [I]), and pid-2 (G). pin1 and pid-2 formed pin-like inflorescences ([B] and [G]) that bear no flowers, and the cells surrounding the tip region show no signs of differentiation in the epidermal layer ([C] and [D]). pcn pin1/+ and pcn pid-2/+ plants form very short pin-like structures that produced lateral organ primordia near the tip ([E], [H], and [I]) and showed arrested meristems with the epidermal layer differentiating guard cells and pavement cells close to the SAM (I). (J) to (N) In pcn wus-1/+ plants (J), the inflorescence meristem terminated in a pin-like structure with arrested lateral organ primordia ([K], [M], and [N]) and trichomes close to the SAM (L). (O) to (R) In pcn tpl-1/+ plants, the inflorescence meristem was aborted ([O] and [P]) or terminated in pin-like structures ([Q] and [P]). Bars = 100 mm in (B), (G), (H), and (K) to (N), 10 mm in (C) to (E) and (I), and 250 mm in (O) to (R). expressed in the vascular cells of the root (Figure 10J) but was absent in the root meristem and vascular cells of the cotyledons (Figures 10J and 10K). These results were consistent with the qRT-PCR analysis that showed that PIN7 was expressed 0.6fold lower than the wild type (see Supplemental Table 2 online). Taken together, pcn mutants displayed altered PIN1 localization and PIN7 expression. To test whether auxin response is altered in pcn mutants, we performed a lateral root induction assay by application of the auxin analog naphthalene acetic acid (NAA) (Evans et al., 1994) to PCN Regulates Embryo and Meristem 4357 Figure 8. Expression Patterns of DR5rev:GFP in the Wild Type and pcn. (A) to (P) DR5rev:GFP expression in wild-type and pcn embryos. DR5rev:GFP in pcn at early embryo stages ([F] to [I]) is similar to the wild type ([A] and [B]) except for ectopic expression in the suspensor cells ([H] to [J]). At later stages, the DR5rev:GFP expression correlates with the specification of cotyledon primordia as observed in wild type ([C] to [E]) and pcn ([M] to [P]) with few exceptions ([K] and [L]). Ectopic DR5rev:GFP expression was observed in the SAM region in the pcn mutant embryo (P). (Q) and (R) DR5rev:GFP expression was reduced in the RAM of pcn (R) in comparison to the wild type (Q). (S) to (V) Ectopic DR5rev:GFP expression was observed in the SAM region in pcn (U) at 228C when compared with the wild type (WT) grown at 228C (S) and 298C (T). This ectopic expression in the SAM was restored back to wild-type expression in pcn (V) when grown at 298C. Bars = 10 mm in (A), (F), and (G), 20 mm in (B) to (E) and (H) to (P), and 100 mm in (Q) to (V). 4358 The Plant Cell Figure 9. Auxin Transport in the Wild Type and pcn. (A) to (E) and (H) to (L) PIN1-GFP localization in wild-type ([A] to [E]) and pcn mutant ([H] to [L]) embryos. The localization of PIN1:GFP in the prospective cotyledon primordia region (B) and later in the developing cotyledon primordia ([C] to [E]) seen in the wild type is absent in pcn embryos that lack cotyledons ([H] and [I]). (F), (G), (M), and (N) To assess the polar localization of PIN1-GFP in the membrane, embryos were stained with FM4-64 and treated with BFA and control. With BFA treatment, the membrane-localized PIN1-GFP in control (F) moved to the cytosol (G) in the wild type, whereas in pcn, with (M) and without (N) BFA treatment, PIN1-GFP was localized to the cytosol. Bars = 10 mm in (A) to (D) and 25 mm in (E) to (N). seedlings. In the wild type, lateral root primordia induced by 2 mg/L NAA displayed normal DR5rev:GFP expression (Figures 11A and 11B). However, in pcn, NAA induced abnormal bulges of pericycle cells with diffused expression of DR5:GFP that did not differentiate into lateral root primordia (Figures 11C to 11E). Thus, while auxin maxima displayed by DR5rev:GFP appeared in the proper position, subsequent root development required PCN activity. In an alternate assay to measure the strength of the auxin response in pcn mutants, we used the pHS:AXR3NT-GUS reporter (Gray et al., 2001). In the pHS:AXR3NT-GUS construct, the GUS gene is fused to the N-terminal domains I and II of the AXR3/ IAA17 and its expression is controlled by a heat shock promoter. Due to the inclusion of the degron-containing domain II of AXR3, the associated GUS is subject to degradation in an auxinresponsive manner (Gray et al., 2001). As shown in Figures 11I to 11K, 11M, and 11N, the fusion protein was more stable in pcn seedlings than in the wild type as visualized by GUS, suggesting that indole-3-acetic acid (IAA) protein turnover is affected in pcn mutant. These results collectively show that PCN plays a role in auxin response pathway. PCN Interacts with the Auxin Response Module Comprising BDL and MP The IAA/AUX-ARF auxin module BDL/MP mediates auxin transcriptional response and together with PLT1 and PLT2 positively regulates PIN expression in root and vascular development (Szemenyei et al., 2008). In pcn, the expression domains of MP and PLT1 in the embryonic RAM were more restricted compared with the wild type (Figures 6J to 6M); also in the vascular initials of the pcn embryo, MP expression was discontinuous and ectopic in the SAM region of the torpedo embryo (Figure 6K). Thus, aber- rations in the expression of MP and PLT1 were more pronounced in the pcn embryos that showed stronger developmental defects. Consistent with this observation, MP-GUS in the pcn mutant showed ectopic expression in the apical region of the embryo that included the SAM (Figures 12A4 to 12A6) and the cotyledon primordia (Figure 12A5). Furthermore, qRT-PCR showed that MP, WOX5, PLT1, PLT2, and PLT3 were significantly upregulated in pcn embryos (see Supplemental Table 2 online). These findings prompted us to investigate potential interactions between pcn and auxin mutants. mp single mutants develop a basal stump instead of a root and produce one or two cotyledons (Berleth and Jurgens, 1993). In the progeny of pcn/+ mp/+ plants, we identified putative double mutants at a ratio 1:15 with a novel embryo phenotype. These double mutant embryos showed cup-like cotyledons with a narrow embryo axis compared with the wild type (Figures 12A1 and 12A2). These embryos were arrested not only at 228C but also at 298C, similar to the observations with double mutants of pcn and wus-1, suggesting that MP functions are required for the rescue of the pcn mutant at 298C. The pcn mp/+ plants were small and had delayed rosette leaf and root growth, and the SAM terminated into pin-like structures (Figure 12A3) compared with pcn single mutant and mp/+ heterozygotes that flower normally. These observations suggest that PCN cooperates with MP to mediate auxin signals during embryonic and postembryonic developmental programs in Arabidopsis. BDL is considered a repressor of MP in auxin signaling during embryo development, and the bdl gain-of-function mutant resembles the mp loss of function mutant (Hamann et al., 2002). We investigated BDL interaction with PCN using a pcn bdl double mutant. The bdl mutant line we used was in the Columbia (Col) background and displayed abnormal horizontal divisions in PCN Regulates Embryo and Meristem 4359 Figure 10. Expression Patterns of pPIN7:PIN7-GUS in the Wild Type and pcn. PIN7-GUS localization in the wild type ([A] to [E]) and pcn ([F] to [K]). PIN7-GUS is present in the provascular initials and suspensor cells of developing wild-type embryos ([A] to [C]), whereas in pcn, PIN7-GUS is absent in the embryo proper of early arrested embryos (F) and in the provascular cells of the radicle of later arrested embryos ([G] to [I]). In the wild-type seedling, PIN7-GUS is present in the RAM (D) and differentiating vascular cells of the root and cotyledon ([D] and [E]), whereas in pcn, PIN7-GUS is absent in the RAM and vascular cells of the cotyledon ([J] and [K]) and weakly expressed in the differentiating vascular cells of the root (J). Bars = 100 mm in (A) to (D) and (F) to (J) and 1 mm in (E) and (K). the apical cell of the embryo as observed in the Landsberg erecta (Ler) background (Hamann et al., 1999) but at a lower frequency (5%, n = 480). In the pcn bdl double mutant in the Col background, 55% of embryos (n = 275) showed abnormal divisions at the octant stage that also include double octant phenotype (Figures 12C1 and 12C2) and tapered embryo phenotypes in later stages of development (Figures 12C3 and 12C4). Thus, during early embryo development, pcn enhanced the bdl embryo defects (Figures 12B1 to 12B3 and 12C1 to 12C4). Furthermore, the pcn bdl double mutant had fewer suspensor cells than both pcn or bdl single mutants (Figures 12B1 to 12B3 and 12C1 to 12C4). At the heart stage, pcn bdl embryos displayed a broader central zone (Figures 12C5 and 12C6) than the wild type or pcn single mutant (Figures 3I, 3J, 3X, and 3Y). However, during embryo development, the globular stage arrest, cotyledon defects, and seed abortion observed in the pcn single mutant at 228C were suppressed by the presence of bdl in the double mutant. The pcn bdl set viable seeds at 228C unlike the pcn single mutant that requires 298C treatment. To examine if BDL expression is altered in pcn mutant, we introduced pBDL:BDL-GFP reporter into pcn. BDL-GFP expression was first detected in the vascular initials and the RAM of the wild-type embryos (Figures 12D1 and 12D2). However, in pcn, BDL expression was detected in the SAM region but was absent in the vascular initials and the embryonic RAM from torpedo stage onwards when grown at 228C (Figures 12D3 to 12D6). This altered expression of BDL in pcn was restored to a normal wild-type -like pattern at 298C, suggesting that the pcn mutant phenotype at 228C is dependent on BDL wild-type function (Figure 12D7). Taken together, the quantitative enhancement of auxin-related defects of both mp and bdl mutants in pcn background suggests that PCN is required for gene functions overlapping with those of MP and BDL in embryogenesis. These embryonic observations are consistent with a contribution of PCN to the auxin-induced degradation of multiple auxin/IAA proteins. We performed a yeast two-hybrid (Y2H) assay to determine whether PCN physically interacts with BDL, MP, or TPL. When PCN fragments other than the C-terminal region or a full-length PCN was used as the bait, PCN showed very strong selfactivation. The C-terminal fragment did not show self-activation, and it did not show any physical interaction between PCN and BDL, MP, or TPL. When we used BDL, MP, or TPL as the bait and PCN as the prey, we did not observe any physical interaction of PCN with BDL, MP, or TPL. However, qRT-PCR analyses of BDL, TPL, and MP in pcn mutant embryo revealed that BDL and TPL expression are similar to the wild type, but MP and its target genes DORNRÖSCHEN (DRN) and DORNRÖSCHEN-LIKE are significantly upregulated, 1.6-, 9.7-, and 4.7-fold, respectively, compared with the wild type (see Supplemental Table 2 online). Previous work has shown that MP binds to the DRN promoter and regulates its transcription positively (Cole et al., 2009). Consistently, upregulated MP expression in pcn also increased the DRN expression in this study (see Supplemental Table 2 online). The loss-of-function mutant drn-1 shows defective embryo patterning and functional postembryonic SAM (Chandler et al., 2007), whereas the gain-of-function drn-D mutant shows defective SAM and upregulated expression of both WUS and CLV3 as seen in pcn (Kirch et al., 2003). The pcn drn-1 double mutant showed the pcn embryo phenotype. However, after 4360 The Plant Cell Figure 11. Auxin-Induced Lateral Root Initiation and IAA Protein Stability Is Altered in the pcn Mutant. (A) to (E) Lateral root induction and DR5-GFP localization in the wild type (WT) ([A] and [B]) and pcn ([C] to [E]). Lateral root primordia induction by NAA (2 mg/mL) in the wild type (A) and pcn (C). DR5rev:GFP expression in NAA-induced (2 mg/mL) lateral roots in the wild type at 2 d (B), pcn at 2 d (D), and pcn at 5 d (E). The polarly localized DR5rev:GFP expression pattern observed in wild-type lateral roots (B) is altered and mislocalized in pcn ([D] and [E]). (F) to (N) pHS:AXR3NT-GUS expression in the wild-type root at 0 h (F), 2 h (H), and 3 h (L) and in pcn root at 0 h (G), 2 h ([I] to [K]), and 3 h ([M] and [N]). The heat shock–induced AXR3NT-GUS expression is degraded within 2 h in the wild type ([F], [H], and [L]), whereas in pcn, the GUS expression is still retained at 3 h ([M] and [N]). Bars = 100 mm. rescue at 298C and during postembryonic development, the young rosettes terminated growth with arrested SAM and radialized leaf primordia, which were not observed in the respective single mutants (Figures 12E1 to 12E3), further supporting the involvement of PCN mediated auxin signaling in meristem regulation. The absence of PCN interaction with BDL in the Y2H assay does not rule out interaction via other partner(s). Taken together, PCN may function in a multiprotein complex that includes BDL and TPL to regulate the downstream genes in the auxin pathway that includes DRN. differentiation of meristem cells. As one of its functions, TPL acts as a corepressor with BDL in a complex with MP (Szemenyei et al., 2008). tpl-1 and tpl-1/+ plants developed similar to the wild type after bolting, whereas the pcn tpl-1/+ inflorescence meristems also terminated in a pin-like inflorescence (Figures 7Q and 7R). Taken together, in the absence of PCN function, inflorescence meristem development becomes more sensitive to the gene dosage of PIN1, PID, and TPL. DISCUSSION PIN1, PID1, and TPL Are Dosage Sensitive in pcn Double mutants of pcn and pin1-1, pid-2, or tpl-1 arrested embryo development at 298C, albeit the respective single mutants completed embryogenesis and produced viable seeds. These observations suggest that the rescue of pcn embryo lethality by elevated temperatures requires the functions of the auxin components PIN and PID and the global corepressor TPL. PIN1 promotes directional auxin efflux, and PID mediates the switch of PIN polarity. Both pin1-1 and pid-2 single mutants upon bolting form pin-like structures with an undifferentiated inflorescence meristem at the tip (Figures 7B to 7D and 7G) (Friml et al., 2004; Kaplinsky and Barton, 2004). Unlike in pcn or pin1-1/+ and pid-2/+ heterozygous plants, in pcn pin1-1/+ (Figure 7A) and pcn pid-2/+ plants (Figure 7F), inflorescence shoot meristem terminated in a pin-like structure, and lateral organ primordia were arrested (Figures 7E, 7H, and 7I). In all cases, trichomes and guard cells, which are normally restricted to leaves or leaf primordia, were found at the tip of the pin-like structure in the meristem region (Figures 7H and 7I), indicative of precocious We identified PCN as an essential gene for Arabidopsis embryo development. Mutations in the PCN locus resulted in developmental defects that link maintenance of the embryonic meristem and cotyledon formation to auxin-mediated processes that regulate early to late patterning events during embryogenesis. Embryo defects in pcn were detectable as early as the four-cell stage and progressively displayed more defects both in the apical and basal programs, resulting in striking cotyledon phenotypes, enlarged SAM, and abnormal RAM, suggesting the importance of PCN for embryo development, including stem cell organization and maintenance. Our studies indicate a significant contribution of PCN in the early steps of auxin signaling by its ability to genetically interact with BDL and, therefore, likely functions as a repressor to regulate several key pathways that operate during embryo patterning (see Supplemental Table 2 online). TPL, another well-studied corepressor functions with BDL (Szemenyei et al., 2008), also genetically interacts with PCN. Auxin maxima and distribution were also altered in the pcn mutant, suggesting a link between auxin-mediated embryo PCN Regulates Embryo and Meristem 4361 Figure 12. Genetic Interactions of mp, bdl, and drn Mutants with pcn. (A1) and (A2) A mp pcn double mutant shows an abnormal embryo phenotype. (A3) A pcn mp/+ rosette SAM terminates in a pin-like structure. (A4) to (A6) MP-GUS in pcn embryo is ectopically expressed in the SAM. (B1) to (B3) and (C1) to (C6) A bdl pcn double mutant shows increased abnormal cell division during early and mid-embryo development ([C1] to [C6]) compared with the bdl single mutant ([B1] to [B3]). (D1) to (D7) pBDL:BDL-GFP expression pattern in wild-type ([D1] and [D2]) and pcn ([D3] to [D7]) embryos. In pcn, BDL-GFP is ectopically expressed in the SAM ([D5] and [D6]) and occasionally absent in the RAM (D6) compared with the wild type ([D1] and [D2]). This altered BDL-GFP expression in pcn could be rescued through growth at 298C (D7). (E1) to (E3) The pcn drn-1 double mutant shows arrested SAM. pcn drn-1 plant with the SAM and young leaf primordia boxed in yellow (E1) and scanning electron micrographs of the arrested SAM and defective leaf primordia ([E2] and [E3]). Bars = 10 mm in (B1) to (B3) and (C1) to (C6), 25 mm in (D1) to (D7), 50 mm in (A4) to (A6), 100 mm in (A1) to (A3) and (E3), and 1 mm in (E2). 4362 The Plant Cell patterning and PCN function. Another interesting feature of the pcn mutant embryo is its ability to recover at 298C, which enabled investigation into the postembryonic roles of PCN. The defects in NAA-induced lateral root formation in pcn seedlings along with the prolonged IAA protein stability in this mutant suggest that PCN is required for auxin-induced response and downstream signaling. PCN genetically interacts with WUS and the transcriptional corepressor TPL. WUS, which functions in a negative feedback loop to specify stem cells of the SAM, has been shown to physically interact with a TPL ortholog in Antirrhinum majus to repress differentiation pathways in the SAM (Schoof et al., 2000; Kieffer et al., 2006). In addition, pcn also genetically interacts with PIN and PID as both pcn pin1-1/+ and pcn pid-2/+ developed pin-like inflorescence with terminal differentiation. This suggests that PCN is required for sustained meristem activity during postembryonic development. Interestingly, the respective homozygous double mutants displayed strong developmental arrest at the globular stage. These observations suggest that PCN functions are coordinated with PIN and PID both during embryo development and postembryonically in developmental programs associated with meristems. The postembryonic phenotypes also suggest that PCN has a significant role in the maintenance of a functional SAM through the auxin signaling pathway; previously, the auxin signaling pathway has been shown to be essential for RAM functions (Benjamins and Scheres, 2008). Based on these observations, it is tempting to speculate that PCN likely functions in the BDL/TPL repressive pathway to regulate auxin-mediated embryo patterning and meristem maintenance. PCN Plays an Important Role in Restricting the Meristem Size An enlarged SAM (Figures 2J and 2M) and ectopic WUS expression in the L1 and L2 layers (Figure 6B) were both observed in pcn mutant. WUS is normally restricted to a few cells in the L3 layer of the wild type (Figure 6A); these WUS-expressing cells act as an organizing center and signal the overlying cells to function as stem cells (Mayer et al., 1998). Ectopic expression of WUS in the L1 layer using the CLV3 promoter results in an abnormally swollen SAM that is arrested in the seedling (Brand et al., 2002), and a recent study showed that WUS directly activates CLV3 expression by binding to its promoter region (Yadav et al., 2011). Therefore, WUS misexpression in the L1 and L2 layers followed by its self-amplification is partly responsible for the meristem defects in pcn. pcn wus-1 double mutants could not develop past the globular stage either at 22 or 298C, indicating that WUS and PCN together are essential for early embryogenesis. The double mutant embryos of pcn tpl-1 and pcn wus-1 are arrested at the globular stage even when grown at 298C. TPL and WUS mutations are haploinsufficient in the pcn background, as the pcn tpl-1/+ and pcn wus-1/+ seeds were viable if grown at 298C, but developed similar pin-like inflorescence phenotypes postgermination. These observations are consistent with the proposed function of TPL as a corepressor and its physical interactions with WUS (Kieffer et al., 2006). Furthermore, these results also suggest that PCN and TPL likely mediate alternative roles in regulating WUS repression and activation to maintain the SAM functions. The replacement of inflorescences with pin-like structures is usually considered as an indicator of the disruption in auxin movement and gradient (Okada et al., 1991; Friml et al., 2004; Kaplinsky and Barton, 2004). Disordered divisions in the SAM and RAM occur at the same time as the loss of organizing center and QC identity in the pcn mutant. Abnormal SAM, RAM, and auxin maxima observed in pcn indicate that PCN plays an important role in maintaining meristem functions through auxin signaling. WUS expression is negatively regulated by the CLV signal transduction pathway (Brand et al., 2000; Schoof et al., 2000). Surprisingly, our study shows a broader expression of CLV3 in pcn (Figures 6G to 6I), although WUS expression was also expanded. One possible explanation is that in pcn mutants, CLV signaling is compromised, resulting in enlarged meristem. Therefore, PCN likely provides a link between genetic regulation of meristem maintenance and auxin signaling pathway. PCN Functions in the Auxin Pathway Regulated degradation of auxin/IAA proteins that repress ARF transcription factors bound to auxin response elements play a crucial role in auxin-mediated signaling (Mockaitis and Estelle, 2008). BDL, an IAA protein, along with the corepressor TPL (Szemenyei et al., 2008), is thought to form a complex with the ARF transcription factor MP (Weijers et al., 2006) to regulate auxin-responsive target genes. BDL and MP are expressed from zygote to early globular stages and are subsequently restricted to the hypophyseal cell derivatives and the developing vascular initials (Hamann et al., 2002). Our study indicates that the pcn mutation interferes with auxin response similar to mutations in the well-characterized bdl (Hamann et al., 1999). Like bdl, mutations in PCN cause abnormal cell divisions in the basal domain during early embryo development. Although pcn enhanced the early embryo phenotypes of the bdl mutant, surprisingly, bdl rescued the pcn mutant embryo even when grown at 228C, suggesting that BDL activity is involved in pcn embryo lethality. Consistent with this, the ectopic expression of BDLGFP and DR5rev:GFP in the SAM of pcn embryos was suppressed in pcn plants grown at 298C for seed/embryo rescue (Figures 8U, 8V, 12D6, and 12D7). Furthermore, the misexpression of STM outside the SAM (Figure 6D) and the auxin maxima observed in the SAM of the pcn embryo (Figure 8P) suggest that PCN is required for tight regulation of auxin response and maxima in the SAM. Therefore, it is likely that ectopic BDL activity in the SAM of pcn embryos contributes to the striking pcn embryo phenotypes, suggesting that BDL function is required for the arrested pcn embryo phenotype. Because the bdl pcn double mutant is not completely reverted to the wild type, it is likely that other genes may be involved in pcn rescue at 228C. Our results suggest that TPL activity may provide some regulatory functions in the pcn mutant background. Consistent with this, the pcn tpl-1 double mutant is embryo lethal with embryos arrested at the globular stage when grown either at either 22 or 298C. Furthermore, the heterozygous tpl-1 in pcn background (pcn tpl-1/+) had a partially terminated SAM, whereas heterozygous pcn in tpl-1 background (pcn/+ tpl-1) showed tpl-1 postembryonic phenotypes (Figures 7O to 7R). PCN Regulates Embryo and Meristem These observations suggest that PCN and TPL have both distinct and overlapping functions. Since PCN appears to function together with BDL and TPL to regulate downstream targets, disruption of the BDL containing complex may cause an unknown alternative pathway to rescue pcn in the bdl mutant background. Although the pcn mp/+ mutant was not arrested at 298C, it showed severe cotyledon phenotypes compared with the mp single mutant and postembryonically the SAM terminated in a pin-like structure (Figures 12A1 to 12A3). As our results indicate that PCN genetically interacts with BDL, it is likely that PCN works with BDL and TPL to negatively regulate MP and its targets. It is interesting to note that both of these genes that genetically interact with BDL encode WD-40 repeat–containing proteins (Szemenyei et al., 2008). In addition to the involvement of PCN in the BDL/TPL repression of MP, the pHS:AXR3NTGUS–based assay (Gray et al., 2001) in the pcn showed that IAA proteins are more stable in the pcn mutant, suggesting that PCN has a broader role in auxin signaling. PCN Integrates Auxin Signaling and Meristem Functions The pcn mutant phenotype, its genetic interactions with factors associated with auxin signaling and meristem fate, and the qRTPCR results of genes that are differentially regulated in the pcn 4363 embryo indicate that PCN functions along with BDL and TPL to mediate MP repression (Figure 13). MP regulates both its own expression and that of BDL, thus functioning as a genetic switch triggered in response to auxin in a threshold-specific manner to degrade BDL and relieve the repression of its target genes (Lau et al., 2011). MP, which is upregulated in the pcn embryo, through negative regulation of ARR7 and ARR15 (downregulated in pcn embryo; see Supplemental Table 2 online), likely regulates the WUS/CLV3 feedback loop in the SAM as previously suggested (Zhao et al., 2010), thus revealing a link between auxin signaling and meristem function. Several studies have shown that PIN polarity and the resulting auxin localization are essential for embryo patterning (Friml et al., 2003; Geldner et al., 2003; Michniewicz et al., 2007; Kitakura et al., 2011). In the absence of functional PCN, PIN polarity is not maintained in the embryo, which leads to ectopic auxin maxima in the SAM region. This likely results in the disruption of BDL-mediated repression of MP and ectopic expression of MP and BDL in the pcn embryonic SAM. Ectopic auxin maxima along with MP/BDL expression in the SAM likely also contribute to the disruption of the WUS/CLV3 feedback loop, leading to the improper partitioning of the SAM and cotyledon primordia in the apical domain of the pcn globular embryo. Furthermore, in pcn embryos, several genes encoding AP2 domain transcription factors and members of the WOX Figure 13. Working Model of PCN Functions in Embryo Development and Meristem Organization via the Auxin Signaling Pathway. This model summarizes key findings of this study. As shown in the top panel, PCN likely functions first as a repressor along with TPL and BDL to regulate known (i.e., DRN and ARR15) and potential (i.e., ARR7 and PLT) MP target genes during embryo development and later during meristem organization and maintenance. The middle panel shows polar localization of PIN1 in the wild-type (WT) embryo (orange lines) followed by patterning events that establish the embryonic SAM via the CLV-WUS–mediated regulatory loop and the embryonic RAM with auxin maxima and WOX5 in the QC. As shown in the bottom panel, disruption of PIN1 polarity in the pcn mutant embryo and auxin maxima in the enlarged pcn SAM results in ectopic expression of DR5GFP, MP, and BDL in the SAM that likely affects the CLV-WUS loop (indicated by the broken line); in pcn, RAM is defective with expanded WOX5 expression and disrupted auxin maxima. Orange lines indicate PIN1 localization; orange arrows show the direction of auxin flow; green highlighted regions indicate auxin maxima as shown by DR5-GFP expression; and blue highlighted regions show WOX5 expression. Heterozygous mutations in TPL, PIN1, PID, MP, and WUS genes and homozygous recessive drn in the pcn background are able to progress through embryogenesis and produce viable seeds with the 298C treatment; however, postembryonically their inflorescence meristems terminate in pin-like structures. 4364 The Plant Cell family that have been shown to maintain the activity of stem cells and control embryo patterning (Haecker et al., 2004; Würschum et al., 2006; Chandler et al., 2007; Galinha et al., 2007) are significantly upregulated as shown by our qRT-PCR analysis (see Supplemental Table 2 online). In the embryonic and postembryonic RAM, auxin induces the expression of AP2 transcription factors, PLT1 and PLT2, to specify the QC (Aida et al., 2004), and both of these genes and WOX5 are upregulated in the pcn embryo (see Supplemental Table 2 online). Collectively, these lines of evidence indicate that PCN functions as a negative regulator or repressor with key roles in embryo patterning and meristem organization/maintenance through auxin mediated signals (Figure 13). In conclusion, based on our developmental and genetic studies, the earliest signs of embryonic pcn defects are associated with auxin transport and response pathway. These mutant phenotypes are further enhanced by mutations in several key components of auxin transport and signaling involving pin-1, pid, tpl, and mp (Figure 13). These genes also have a dosage-dependent auxin transport and signaling defect–associated pin phenotypes after bolting (Figure 13). Interestingly, genetic studies with meristem factor WUS also revealed similar embryonic and postembryonic phenotypes linking the auxin signaling defects with embryonic and postembryonic meristems (Figure 13). The genetic and functional framework developed for PCN in this study (Figure 13) will help future experiments to dissect other components of PCN-mediated pathways that operate during embryonic and postembryonic development in Arabidopsis. METHODS Plant Growth, Mutant Lines, and Map-Based Cloning Arabidopsis thaliana plants were grown on soil or in sterile culture on 0.53 MS medium containing MS salts. After incubation for at least 4 d at 48C in darkness, plants were grown in growth chambers under long-day conditions (16 h light and 8 h dark; light intensity: 60 to 80 mmol m22 s21) at 22 or 298C. Homozygous pcn plants and double mutants of pcn were grown at 228C until they began to flower and then shifted to 298C, except pcn bdl, which was grown at 228C. Details of mutants and marker lines used in this study are listed in Supplemental Table 3 online. Prior to mapping, the pcn-1 mutant was backcrossed four times to wild-type Col-0. pcn-1/+ (BC4, Col-0) 3 wild-type Ler and 670 F2 plants were used for mapping. Crosses with known genetic markers indicated that PCN mapped close to the centromere of chromosome 4. Because no known genetic markers were available in this region, we designed 20 CAPS markers. Then, 87 F2 plants were assayed for these markers on the long arm close to the centromere of chromosome 4. The initial mapping was done using simple sequence repeat markers from The Arabidopsis Information Resource (http://www.Arabidopsis.org). The derived CAPS markers used for fine mapping of the pcn-1 mutation were generated based on the sequence information of Col and Ler. Construction of pPCN:PCN-GFP and Complementation Vectors and Transformation into Arabidopsis pPCN:PCN-GFP constructs were derived as follows: First, we used forward primer1, 59-AACACTGCAGCGCAATCGCACCTATTTTTAATC-39, and reverse primer1, 59-AAAAGCGGCCGCGACGGAAGCAGAAGGAGAAGTGT-39, to amplify the PCN promoter sequence from genomic DNA, and the PCN cDNA was amplified by PCR from a wild-type Col cDNA library using forward primer 2, 59-AAAAGCGGCCGCATGCTCGAGTACCGTTGCAGCTC-39, and reverse primer 2, 59-AAAAGGATCCAGTTCCAAAAATATGTCTGTC-39. Both promoter and cDNA PCR products were digested with NotI and then the fragments were ligated. Finally, we used the primers 59-AAAGCGGCCGCACACTTCTCCTTCTGCTTCCGTC-39 and 59-AAAAGGATCCAGTTCCAAAAATATGTCTGTC-39 to amplify pPCN:PCN using the ligated PCR product as template, which was fused to GFP. The PCN genomic sequence (At4g07410) for fusion with GFP was amplified by PCR using F28D6 BAC clone as template (forward primer, 59-AACACTGCAGCGCAATCGCACCTATTTTTAATC-39; reverse primer, 59-AAAAGGATCCAGTTCCAAAAATATGTCTGTC-39). The At1g27470 coding sequence (forward primer, 59-ATGTTTGAGTACCGGTGCAGCTC-39; reverse primer, 59-TTATGTCCCGAATATATGTCTGT-39) was cloned by PCR using wild-type Col-0 embryo cDNA library as template. The amplified cDNA and genomic DNA were gel purified and cloned into the pCR 2.1 TA vector (Invitrogen; catalog number K4500-01) and the respective inserts sequenced for confirmation. The 6.5-kb fragment containing the PCN gene (forward primer, 59-GAGCATAATAAACATAAACATTA-39; reverse primer, 59-TCAAGTTCCAAAAATATGTCTGT-39), the ligated PCN promoter with PCN cDNA, as well as the ligated PCN promoter with At1g27470 cDNA were subcloned into pRD400 (Datla et al., 1992) and used to transform pcn-1/+ and pcn-2/+, respectively. The transformations of pPCN:PCN-GFP and complementation constructs into Arabidopsis were performed following the protocol described in Yang et al. (2009). PCR-Based Genotyping and qRT-PCR Genotypes of the pcn-1 and pcn-2 loci in the transgenic plants were determined by PCR. To genotype the pcn-1, PCR primers (forward primer, 59-TCTGAGTAACTGTTTCTTCTGTTGC-39; reverse primer, 59- CCACTCACAAGAACTGAACACCT-39) were used; the PCR fragment sizes are 218 bp (mutant) and 254 bp (Col wild type). To genotype pcn-2 (Salk_ 022607), forward primer 59-TTCCGGATGATATACTGCCAG-39, reverse primer 59-CCCCAGGAAACTCTTGATACC-39, and middle primer 59-GCGTGGACCGCTTGCTGCAACT-39 were used. Other primers and methods used for PCR genotyping of double mutants in this study are listed in Supplemental Table 3 online. The qRT-PCR experiment was performed in triplicate on the Applied Biosystem Step One real-time PCR system using the SYBR Green PCR master mix. Three biological replicates of globular to heart stage wildtype and pcn embryos were isolated, and total RNA samples were isolated as described in RNAqueous-micro kit (Ambion; catalog number 1927). Using the isolated total RNA, linear amplification of antisense RNAs (aRNAs) was performed as described in the MessageAmp II aRNA kit following the protocol for first-round aRNA amplification (Ambion; catalog number 1751). The respective double-stranded cDNAs were synthesized from aRNA as described in the MessageAmpTM II aRNA kit (Ambion; catalog number 1751). The concentration of the cDNA template was measured using Nanodrop 8000 (Thermo Scientific) and further normalized using tubulin (At5g12250) as reference prior to performing qRT-PCR. Gene-specific primers were designed using Primer 3 software. qRT-PCR primers and results are listed in Supplemental Table 2 online. Assay for BFA, FM4-64 Staining, Auxin Response, and IAA Degradation For BFA treatment and FM4-64 staining, the wild-type and pcn embryos were isolated and treated as described earlier (Ganguly et al., 2010). To determine the auxin response of pcn, both mutants and wild-type (Col-0) seeds were surface sterilized. These seeds were incubated at 48C for 3 d and then were germinated on 0.53 MS for 4 d. These seedlings were transferred to 0.53 MS supplemented with 0.2 mg/L NAA. Seedling PCN Regulates Embryo and Meristem phenotypes were examined at 2 and 4 d with NAA in the medium. The response was evaluated by analyzing lateral root development as well as DR5rev:GFP expression. Both pHS:AXR3NT-GUS and pcn/pHS: AXR3NT-GUS seeds were germinated in 0.53 MS for 5 d and then heatshocked for 2 h at 378C and incubated at 208C for 1 to 3 h to check the degradation of GUS (Gray et al., 2001). Y2H Assay The Y2H assay was performed as described in the ProQuest two-hybrid system with the Gateway Technology Kit protocol (Invitrogen; catalog number PQ10001-01). Genes and primers are listed in Supplemental Table 4 online. 4365 Supplemental Data Set 1. Text File of the Sequences and Their Alignment Used for the Phylogenetic Analysis Shown in Supplemental Figure 1. ACKNOWLEDGMENTS We thank Jeff Long for providing the tpl-1 line, Mark Estelle for providing the pHS:AXR3NT-GUS line, and John Chandler for providing the drn-1 line. We thank Jeff Long, Mark Smith, and Don Palmer for critical comments on the manuscript. We thank Daryoush Hajinezhad for assistance with confocal microscopy. This research was supported by the National Research Council Canada Genomics and Health Initiative (GHI-4). This is National Research Council Canada publication number 50186. Microscopy Whole-mount embryos were prepared by clearing the ovules in chloral hydrate solution (8:1:2 chloral hydrate:glycerol:water [w/v/v]) (Christensen et al., 1998) for 4 h. Slides were viewed under a Leica DMR compound microscope using differential interference contrast optics. Confocal laser scanning microscopy was performed using Leica SP2 and Zeiss LSM510 microscopes. Histochemical staining for GUS activity in this study was performed following the protocol of Gray et al. (2001). Lugol staining of the roots to detect starch granules was performed as described by van den Berg et al. (1997). Whole-mount in situ hybridization was performed following the protocol of Hejátko et al. (2006). Scanning electron microscopy was performed as described by Venglat et al. (2002). Phylogenetic Analysis The FASTA sequences for At1g27470, PCN, and its putative homologs in other plant genera (Oryza, Sorghum, Populus, Ricinus, and Vitis), yeast, zebra fish, and humans were aligned using ClustalW (Larkin et al., 2007). The phylogram was generated using QuickTree (Howe et al., 2002) and viewed in Archaeoteryx (http://www.phylosoft.org/archaeopteryx) (see Supplemental Data Set 1 and Supplemental Figure 1 online). Accession Numbers Sequence data from this article can be found in the Arabidopsis Genome Initiative or GenBank/EMBL databases under the following accession numbers: PCN (At4g07410), pcn-1 (At4g07410), pcn-2 (Salk-022607; At4g07410), At1g27470, WUS (At2g17950), CLV1 (At1g75820), CLV3 (At2g27250), DRN (At1g12980), STM (At1g62360), MP (At1g19850), BDL (At1g04550), TPL (At1g15750), PIN1 (At1g73590), PID (At2g23450), PIN7 (At1g23080), WOX5 (At3g11260), PLT1 (At3g20840), and tubulin (At5g12250). Supplemental Data The following materials are available in the online version of this article. Supplemental Figure 1. Phylogenetic Relationship between PCN and Its Homolog in Arabidopsis and Putative Homologs from Different Eukaryotic Species. Supplemental Table 1. Temperature Shift–Dependent Recovery of pcn. Supplemental Table 2. qRT-PCR of Selected Genes That Are Involved in Embryo Patterning and Shoot/Root Meristems. Supplemental Table 3. Origins and Ecotypes of Mutants and Reporter Lines and Genotyping Methods. Supplemental Table 4. Primers Used for Yeast Two-Hybrid Assay. AUTHOR CONTRIBUTIONS D.X., P.V., and R.D. designed the experiments. D.X. and H.Y. performed map-based cloning, made the constructs, performed transformation, created the double mutants, and conducted in situ hybridization. P.V. and D.X. performed microscopy. D.X. and Y.C. performed qRT-PCR experiments. D.X. and R.W. performed the Y2H assay. M.R., S.S.., E.W., H.W., W.X., D.W., T.B., T.L., and G.S. provided valuable advice, mutant/ reporter lines, and reagents. D.X., P.V., and R.D. wrote the article with contributions from G.S., T.B., and T.L. Received September 15, 2011; revised October 27, 2011; accepted November 18, 2011; published December 9, 2011. REFERENCES Aida, M., Beis, D., Heidstra, R., Willemsen, V., Blilou, I., Galinha, C., Nussaume, L., Noh, Y.S., Amasino, R., and Scheres, B. (2004). The PLETHORA genes mediate patterning of the Arabidopsis root stem cell niche. Cell 119: 109–120. Aida, M., Ishida, T., Fukaki, H., Fujisawa, H., and Tasaka, M. (1997). Genes involved in organ separation in Arabidopsis: An analysis of the cup-shaped cotyledon mutant. Plant Cell 9: 841–857. Aida, M., Ishida, T., and Tasaka, M. (1999). Shoot apical meristem and cotyledon formation during Arabidopsis embryogenesis: interaction among the CUP-SHAPED COTYLEDON and SHOOT MERISTEMLESS genes. Development 126: 1563–1570. Benjamins, R., and Scheres, B. (2008). Auxin: The looping star in plant development. Annu. Rev. Plant Biol. 59: 443–465. Berleth, T., and Jurgens, G. (1993). The role of the monopteros gene in organising the basal body region of the Arabidopsis embryo. Development 118: 575–587. Brand, U., Fletcher, J.C., Hobe, M., Meyerowitz, E.M., and Simon, R. (2000). Dependence of stem cell fate in Arabidopsis on a feedback loop regulated by CLV3 activity. Science 289: 617–619. Brand, U., Grünewald, M., Hobe, M., and Simon, R. (2002). Regulation of CLV3 expression by two homeobox genes in Arabidopsis. Plant Physiol. 129: 565–575. Chandler, J.W., Cole, M., Flier, A., Grewe, B., and Werr, W. (2007). The AP2 transcription factors DORNROSCHEN and DORNROSCHENLIKE redundantly control Arabidopsis embryo patterning via interaction with PHAVOLUTA. Development 134: 1653–1662. Christensen, C.A., Subramanian, S., and Drews, G.N. (1998). Identification of gametophytic mutations affecting female gametophyte development in Arabidopsis. Dev. Biol. 202: 136–151. Cole, M., Chandler, J., Weijers, D., Jacobs, B., Comelli, P., and Werr, 4366 The Plant Cell W. (2009). DORNROSCHEN is a direct target of the auxin response factor MONOPTEROS in the Arabidopsis embryo. Development 136: 1643–1651. Datla, R.S.S., Hammerlindl, J.K., Panchuk, B., Pelcher, L.E., and Keller, W. (1992). Modified binary plant transformation vectors with the wild-type gene encoding NPTII. Gene 122: 383–384. De Smet, I., Lau, S., Mayer, U., and Jürgens, G. (2010). Embryogenesis - The humble beginnings of plant life. Plant J. 61: 959–970. Evans, M.L., Ishikawa, H., and Estelle, M.A. (1994). Responses of Arabidopsis roots to auxin studied with high temporal resolution: comparison of wild type and auxin-response mutants. Planta 194: 215–222. Fletcher, J.C., Brand, U., Running, M.P., Simon, R., and Meyerowitz, E.M. (1999). Signaling of cell fate decisions by CLAVATA3 in Arabidopsis shoot meristems. Science 283: 1911–1914. Friml, J., Vieten, A., Sauer, M., Weijers, D., Schwarz, H., Hamann, T., Offringa, R., and Jürgens, G. (2003). Efflux-dependent auxin gradients establish the apical-basal axis of Arabidopsis. Nature 426: 147–153. Friml, J., et al. (2004). A PINOID-dependent binary switch in apicalbasal PIN polar targeting directs auxin efflux. Science 306: 862–865. Furutani, M., Vernoux, T., Traas, J., Kato, T., Tasaka, M., and Aida, M. (2004). PIN-FORMED1 and PINOID regulate boundary formation and cotyledon development in Arabidopsis embryogenesis. Development 131: 5021–5030. Galinha, C., Hofhuis, H., Luijten, M., Willemsen, V., Blilou, I., Heidstra, R., and Scheres, B. (2007). PLETHORA proteins as dosedependent master regulators of Arabidopsis root development. Nature 449: 1053–1057. Ganguly, A., Lee, S.H., Cho, M., Lee, O.R., Yoo, H., and Cho, H.-T. (2010). Differential auxin-transporting activities of PIN-FORMED proteins in Arabidopsis root hair cells. Plant Physiol. 153: 1046–1061. Geldner, N., Anders, N., Wolters, H., Keicher, J., Kornberger, W., Muller, P., Delbarre, A., Ueda, T., Nakano, A., and Jürgens, G. (2003). The Arabidopsis GNOM ARF-GEF mediates endosomal recycling, auxin transport, and auxin-dependent plant growth. Cell 112: 219–230. Geldner, N., Friml, J., Stierhof, Y.-D., Jürgens, G., and Palme, K. (2001). Auxin transport inhibitors block PIN1 cycling and vesicle trafficking. Nature 413: 425–428. Gray, W.M., Kepinski, S., Rouse, D., Leyser, O., and Estelle, M. (2001). Auxin regulates SCF(TIR1)-dependent degradation of AUX/IAA proteins. Nature 414: 271–276. Haecker, A., Gross-Hardt, R., Geiges, B., Sarkar, A., Breuninger, H., Herrmann, M., and Laux, T. (2004). Expression dynamics of WOX genes mark cell fate decisions during early embryonic patterning in Arabidopsis thaliana. Development 131: 657–668. Hamann, T., Benkova, E., Bäurle, I., Kientz, M., and Jürgens, G. (2002). The Arabidopsis BODENLOS gene encodes an auxin response protein inhibiting MONOPTEROS-mediated embryo patterning. Genes Dev. 16: 1610–1615. Hamann, T., Mayer, U., and Jürgens, G. (1999). The auxin-insensitive bodenlos mutation affects primary root formation and apical-basal patterning in the Arabidopsis embryo. Development 126: 1387–1395. Hardtke, C.S., Ckurshumova, W., Vidaurre, D.P., Singh, S.A., Stamatiou, G., Tiwari, S.B., Hagen, G., Guilfoyle, T.J., and Berleth, T. (2004). Overlapping and non-redundant functions of the Arabidopsis auxin response factors MONOPTEROS and NONPHOTOTROPIC HYPOCOTYL 4. Development 131: 1089–1100. Hejátko, J., Blilou, I., Brewer, P.B., Friml, J., Scheres, B., and Benková, E. (2006). In situ hybridization technique for mRNA detection in whole mount Arabidopsis samples. Nat. Protoc. 1: 1939–1946. Howe, K., Bateman, A., and Durbin, R. (2002). QuickTree: Building huge Neighbour-Joining trees of protein sequences. Bioinformatics 18: 1546–1547. Jenik, P.D., Gillmor, C.S., and Lukowitz, W. (2007). Embryonic patterning in Arabidopsis thaliana. Annu. Rev. Cell Dev. Biol. 23: 207–236. Jurgens, G., and Mayer, U. (1994). Arabidopsis. In Embryos: Color Atlas of Development, J. Bard, ed (London: Mosby-Year Book Limited), pp. 7–21. Kaplinsky, N.J., and Barton, M.K. (2004). Plant biology. Plant acupuncture: Sticking PINs in the right places. Science 306: 822–823. Kieffer, M., Stern, Y., Cook, H., Clerici, E., Maulbetsch, C., Laux, T., and Davies, B. (2006). Analysis of the transcription factor WUSCHEL and its functional homologue in Antirrhinum reveals a potential mechanism for their roles in meristem maintenance. Plant Cell 18: 560–573. Kirch, T., Simon, R., Grünewald, M., and Werr, W. (2003). The DORNRÖSCHEN/ENHANCER OF SHOOT REGENERATION1 gene of Arabidopsis acts in the control of meristem cell fate and lateral organ development. Plant Cell 15: 694–705. Kitakura, S., Vanneste, S., Robert, S., Löfke, C., Teichmann, T., Tanaka, H., and Friml, J. (2011). Clathrin mediates endocytosis and polar distribution of PIN auxin transporters in Arabidopsis. Plant Cell 23: 1920–1931. Larkin, M.A., et al. (2007). Clustal W and Clustal X version 2.0. Bioinformatics 23: 2947–2948. Lau, S., De Smet, I., Kolb, M., Meinhardt, H., and Jürgens, G. (2011). Auxin triggers a genetic switch. Nat. Cell Biol. 13: 611–615. Laux, T., Mayer, K.F., Berger, J., and Jürgens, G. (1996). The WUSCHEL gene is required for shoot and floral meristem integrity in Arabidopsis. Development 122: 87–96. Lenhard, M., Jürgens, G., and Laux, T. (2002). The WUSCHEL and SHOOTMERISTEMLESS genes fulfil complementary roles in Arabidopsis shoot meristem regulation. Development 129: 3195–3206. Long, J.A., and Barton, M.K. (1998). The development of apical embryonic pattern in Arabidopsis. Development 125: 3027–3035. Long, J.A., Ohno, C., Smith, Z.R., and Meyerowitz, E.M. (2006). TOPLESS regulates apical embryonic fate in Arabidopsis. Science 312: 1520–1523. Mayer, K.F., Schoof, H., Haecker, A., Lenhard, M., Jürgens, G., and Laux, T. (1998). Role of WUSCHEL in regulating stem cell fate in the Arabidopsis shoot meristem. Cell 95: 805–815. Mayer, U., Buettner, G., and Juergens, G. (1993). Apical-basal pattern formation in the Arabidopsis embryo: studies on the role of the gnom gene. Development 117: 149–162. Michniewicz, M., et al. (2007). Antagonistic regulation of PIN phosphorylation by PP2A and PINOID directs auxin flux. Cell 130: 1044– 1056. Mockaitis, K., and Estelle, M. (2008). Auxin receptors and plant development: A new signaling paradigm. Annu. Rev. Cell Dev. Biol. 24: 55–80. Nawy, T., Lukowitz, W., and Bayer, M. (2008). Talk global, act localpatterning the Arabidopsis embryo. Curr. Opin. Plant Biol. 11: 28–33. Okada, K., Ueda, J., Komaki, M.K., Bell, C.J., and Shimura, Y. (1991). Requirement of the auxin polar transport system in early stages of Arabidopsis floral bud formation. Plant Cell 3: 677–684. Overvoorde, P., Fukaki, H., and Beeckman, T. (2010). Auxin control of root development. Cold Spring Harb. Perspect. Biol. 2: a001537. Sarkar, A.K., Luijten, M., Miyashima, S., Lenhard, M., Hashimoto, T., Nakajima, K., Scheres, B., Heidstra, R., and Laux, T. (2007). Conserved factors regulate signalling in Arabidopsis thaliana shoot and root stem cell organizers. Nature 446: 811–814. Schoof, H., Lenhard, M., Haecker, A., Mayer, K.F., Jürgens, G., and Laux, T. (2000). The stem cell population of Arabidopsis shoot meristems in maintained by a regulatory loop between the CLAVATA and WUSCHEL genes. Cell 100: 635–644. PCN Regulates Embryo and Meristem Sibout, R., Sukumar, P., Hettiarachchi, C., Holm, M., Muday, G.K., and Hardtke, C.S. (2006). Opposite root growth phenotypes of hy5 versus hy5 hyh mutants correlate with increased constitutive auxin signaling. PLoS Genet. 2: e202. Szemenyei, H., Hannon, M., and Long, J.A. (2008). TOPLESS mediates auxin-dependent transcriptional repression during Arabidopsis embryogenesis. Science 319: 1384–1386. van den Berg, C., Willemsen, V., Hendriks, G., Weisbeek, P., and Scheres, B. (1997). Short-range control of cell differentiation in the Arabidopsis root meristem. Nature 390: 287–289. Venglat, S.P., Dumonceaux, T., Rozwadowski, K., Parnell, L., Babic, V., Keller, W., Martienssen, R., Selvaraj, G., and Datla, R. (2002). The homeobox gene BREVIPEDICELLUS is a key regulator of inflorescence architecture in Arabidopsis. Proc. Natl. Acad. Sci. USA 99: 4730–4735. Vida, T.A., and Emr, S.D. (1995). A new vital stain for visualizing vacuolar membrane dynamics and endocytosis in yeast. J. Cell Biol. 128: 779–792. Weijers, D., Schlereth, A., Ehrismann, J.S., Schwank, G., Kientz, M., and Jürgens, G. (2006). Auxin triggers transient local signaling for cell specification in Arabidopsis embryogenesis. Dev. Cell 10: 265–270. Wu, Y., Haberland, G., Zhou, C., and Koop, H.-U. (1992). Somatic embryogenesis, formation of morphogenetic callus and normal de- 4367 velopment in zygotic embryos of Arabidopsis thaliana in vitro. Protoplasma 169: 89–96. Würschum, T., Gross-Hardt, R., and Laux, T. (2006). APETALA2 regulates the stem cell niche in the Arabidopsis shoot meristem. Plant Cell 18: 295–307. Xiang, D., Venglat, P., Tibiche, C., Yang, H., Risseeuw, E., Cao, Y., Babic, V., Cloutier, M., Keller, W., Wang, E., Selvaraj, G., and Datla, R. (2011). Genome-wide analysis reveals gene expression and metabolic network dynamics during embryo development in Arabidopsis. Plant Physiol. 156: 346–356. Yadav, R.K., Perales, M., Gruel, J., Girke, T., Jönsson, H., and Reddy, G.V. (2011). WUSCHEL protein movement mediates stem cell homeostasis in the Arabidopsis shoot apex. Genes Dev. 25: 2025– 2030. Yang, H., Xiang, D., Venglat, S.P., Cao, Y., Wang, E., Selvaraj, G., and Datla, R. (2009). PolA2 is required for embryo development in Arabidopsis. Botany 87: 626–634. Zdobnov, E.M., and Apweiler, R. (2001). InterProScan—an integration platform for the signature-recognition methods in InterPro. Bioinformatics 17: 847–848. Zhao, Z., Andersen, S.U., Ljung, K., Dolezal, K., Miotk, A., Schultheiss, S.J., and Lohmann, J.U. (2010). Hormonal control of the shoot stemcell niche. Nature 465: 1089–1092. POPCORN Functions in the Auxin Pathway to Regulate Embryonic Body Plan and Meristem Organization in Arabidopsis Daoquan Xiang, Hui Yang, Prakash Venglat, Yongguo Cao, Rui Wen, Maozhi Ren, Sandra Stone, Edwin Wang, Hong Wang, Wei Xiao, Dolf Weijers, Thomas Berleth, Thomas Laux, Gopalan Selvaraj and Raju Datla Plant Cell 2011;23;4348-4367; originally published online December 9, 2011; DOI 10.1105/tpc.111.091777 This information is current as of August 3, 2017 Supplemental Data /content/suppl/2011/12/07/tpc.111.091777.DC1.html References This article cites 61 articles, 32 of which can be accessed free at: /content/23/12/4348.full.html#ref-list-1 Permissions https://www.copyright.com/ccc/openurl.do?sid=pd_hw1532298X&issn=1532298X&WT.mc_id=pd_hw1532298X eTOCs Sign up for eTOCs at: http://www.plantcell.org/cgi/alerts/ctmain CiteTrack Alerts Sign up for CiteTrack Alerts at: http://www.plantcell.org/cgi/alerts/ctmain Subscription Information Subscription Information for The Plant Cell and Plant Physiology is available at: http://www.aspb.org/publications/subscriptions.cfm © American Society of Plant Biologists ADVANCING THE SCIENCE OF PLANT BIOLOGY