* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cellular response in subretinal neovascularization induced
Survey
Document related concepts
Transcript
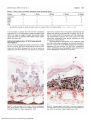
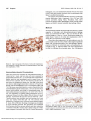
524 Reports 12. Tsujimura A, Shida K, Kitamura M, et al. Molecular cloning of a murine homologue of membrane cofactor protein (CD46): preferential expression in testicular germ cells. Biochem J. 1998;330: 163-168. 13. Bora NS, Kabeer NH, Kim MC, Paryjas S, Atkinson JP, Kaplan HJ. Expression of the complement regulatory proteins in the normal and diseased (EAT) rat eye. In: Nussenblatt RB, Whitcup SM, Caspi RR Gery I, eds. Advances in Ocular Immunology: Proceedings of the 6th International Symposium on the Immunology and Immunopathology of the Eye, Bethesda, Maryland. New York: Elsevier; 1994:83-86. 14. Ballow M, Donshik PC, Rapacz P, Maenza R, Yamase H, Muncy L. Immune responses in monkeys to lenses from patients with con- IOVS, February 1999, Vol. 40, No. 2 tact lens-induced giant papillary conjunctivitis. CLAOJ. 1989; 15: 64-70. 15. Cheung N-K, Walter El, Smith-Mensah WH, Ratnoff WD, Tykocinski ML, Medof ME. Decay-accelerating factor (DAF) protects human tumor cells from complement-mediated cytotoxicity in vitro./C/w Invest. 1988;81:1122-1128. 16. Goslings WRO, Blom DJR, DeWaard-Siebinga I, et al. Membranebound regulators of complement activation in uveal melanomas. Invest Ophthalmol Vis Sci. 1996;37:1884-1891. 17. Bardenstein DS, LaCross G, Medof E. Surface complement regulatory proteins on choroidal melanomas [ARVO Abstracts]. Invest Ophthalmol Vis Sci. 1996;37(3):S246. Abstract nr 1123- Cellular Response in Subretinal Neovascularization Induced by bFGF-Impregnated Microspheres prominent at 8 weeks. CD31-positive endothelial cells were first evident at 14 days and formed neovascular channels that were still present for up to 8 weeks. CD4and CD8-positive lymphocytes appeared in the early stages but were few in number. Hideya Kimura,1'5 Christine Spee,1 Taiji Sakamoto,1 David R. Hinton,2 Yuichiro Ogura,5 Yasuhiko Tabata,4 Yoshito Ikada,4 and Stephen]. Ryan1 CONCLUSIONS. SRN membranes are primarily composed of RPE cells and vascular endothelial cells. The membrane adheres to the retina by a gliotic band. The cellular components involved in the membrane of this model resemble those found in SRN membranes removed from patients with age-related macular degeneration. (Invest Ophthalmol Vis Sci. 1999;40:524-528) determine the sequence of cellular changes associated with a new rabbit model of subretinal neovascularization (SRN) induced by subretinal injection of basic fibroblast growth factor (bFGF)-impregnated microspheres. PURPOSE. TO METHODS. bFGF-impregnated gelatin microspheres, prepared by forming a polyion complex between gelatin and bFGF, were subretinally implanted into rabbit eyes. The eyes were studied by immunochemistry at 3 days to 8 weeks after implantation. Antibodies to CD4, CD8, cytokeratin, CD31, glial fibrillary acidic protein (GFAP), and RAM 11 were used. Cytokeratin-positive retinal pigment epithelial (RPE) cells appeared on day 3 and continued to increase in number in the subretinal space throughout the growth of the SRN membrane, becoming the predominant cell type. Macrophages (RAMH-Positive) appeared early, but most disappeared within 7 days. GFAP-positive Miiller cells were evident early in the retina but migrated into the subretinal space after 7 days; the gliotic adhesion they formed between the retina and the SRN membrane was RESULTS. From the 'Doheny Eye Institute and the departments of Ophthalmology and Pathology, University of Southern California School of Medicine, Los Angeles, California; and the 3 Department of Ophthalmology and Visual Sciences and ''Research Center for Biomedical Engineering, Kyoto University, Kyoto, Japan. Supported in part by Grants EYO1545 and EY03040 from the National Institutes of Health, Bethesda, Maryland; and by a grant from the Hoover Foundation, Pasadena, California. The Department of Ophthalmology is the recipient of an award from Research to Prevent Blindness, New York, New York. Submitted for publication February 13, 1998; revised June 25, 1998; accepted July 31, 1998. Proprietary interest category: N. Reprint requests: Stephen J. Ryan, Doheny Eye Institute, 1450 San Pablo Street, Los Angeles, CA 90033- Downloaded From: http://iovs.arvojournals.org/ on 08/03/2017 ubretinal neovascularization (SRN) is often associated with severe visual impairment, especially in age-related macular Sdegeneration (ARMD). The pathogenesis of SRN is not fully understood; however, the new vessels of SRN are of choroidal origin. Recent studies of surgically excised choroidal neovascular membranes (CNVMs) have provided some pathologic information on the mechanism of CNVM formation.1"3 However, data obtained from these studies have often been limited to late stages in the evolution of the CNVMs. Early stages of CNVMs can only be studied in an animal model. We have developed a new model of SRN in the rabbit based on implanting basic fibroblast growth factor (bFGF)impregnated gelatin microspheres under the retina.4 Approximately 80% of eyes that receive bFGF-impregnated microspheres show fluorescein leakage from the CNVM 2 weeks after implantation. These CNVMs are largely involuted within 8 weeks after microsphere implantation. In this study we examined the time course of cellular response into the subretinal space after initiation of the bFGF SRN model using cell-specific antibodies. The results provide insight into pathogenic mechanisms involved in this lesion and demonstrate relevance to human CNVMs. METHODS Preparation of bFGF-impregnated Microsphere Suspension bFGF-impregnated microspheres were prepared by forming a polyion complex as previously described.4 Briefly, 2.5 mg of cross-linked gelatin microspheres were placed in 100 jixl of distilled water containing 25 /xg of bFGF at 37°C for 1 hour, after which 400 jal of phosphate-buffered solution (PBS; pH IOVS, February 1999, Vol. 40, No. 2 TABLE Reports 525 1. Time Course of Cellular Response in the Subretinal Space Marker Cytokeratin CD31 RAM11 GFAP CD4 CD8 3 Days 1 Week 2 Weeks 4 Weeks 8 Weeks 44- Quantified by counting the number of positive cells in an average of three X40 objective fields: ± , < 1 ; +, 1-10; + + , >10. Subretinal Implantation of bFGF-Impregnated Microspheres One eye each of 18 pigmented rabbits of both sexes, weighing 2.5 to 3.5 kg, was used. All procedures were in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. The animals were anesthe- tized with a mixture (4:1) of ketamine hydrochloride (16 mg/kg) and xylazine hydrochloride (4 mg/kg), and the pupils were dilated with 1% tropicamide and 2.5% phenylephrine hydrochloride eyedrops. The ocular surface was then additionally anesthetized with topical instillation of 0.5% proparacaine hydrochloride. bFGF-impregnated microspheres were implanted under the retina with a glass micropipette as previously described.Jf Each eye received 50 fxl of bFGF-impregnated microsphere suspension (2.5 jLtg of bFGF). The eyes were examined by indirect ophthalmoscopy, photographed, and studied by fluorescein angiography weekly for up to 8 weeks. FIGURE 1. Light micrograph of the lesion 3 days after implantation. Many RAMll-positive cells (arrows) infiltrate under the retina-surrounding microspheres. Original magnification, X500. FIGURE 2. Light micrograph of the lesion 2 weeks after implantation. Note CD31-positive cells (arrows) arising from the choriocapillaris are seen within the membrane. Original magnification, X500. 7.4) was added to prepare the 0.5% (wt/vol) microsphere suspension for in vivo experiments. Gelatin (isoelectric point [p/| of 4,9, molecular weight of 99,000, Nitta Gelatin, Osaka, Japan) and human recombinant bFGF (R&D Systems, Minneapolis, MN) were used. Downloaded From: http://iovs.arvojournals.org/ on 08/03/2017 526 Reports IOVS, February 1999, Vol. 40, No. 2 Burlingame, CA), and aminoethyl-carbazole (Vector) was used to give a red color reaction. Sections were counterstained with Mayer's modified hematoxylin. The primary monoclonal antibodies used were cytokeratin (mouse anti-human; Dako, Carpinteria, CA), CD4 and CD8 (mouse anti-rabbit; Spring Valley, Woodbine, MD), CD31 (mouse anti-human; Dako), glial fibrillary acidic protein (GFAP; Sigma), and RAM11 (mouse anti-rabbit macrophage; Dako). RESULTS s * * * All experimental animals developed SRN and showed a similar sequence of histologic and immunohistochemical findings, which are summarized in Table 1. The eyes showed no fluorescein leakage at 3 days or 1 week. Fluorescein leakage was visible from 2 weeks to 8 weeks after implantation; after 8 weeks the eyes showed no more leakage. Three days after implantation, the microspheres were observed clearly under the retina. Many retinal pigment epithelial (RPE) cells (cytokeratin-positive) and macrophages (RAM 11positive) infiltrated the sub retinal space surrounding the microspheres (Fig. 1). Adjacent Miiller cells were GFAP-positive but did not infiltrate the subretinal space. Few CD4-positive FIGURE 3- Light micrograph of the lesion 2 weeks after implantation. The membrane is mainly composed of cytokeratin-positive cells. Original magnification, X500. Immunohistochemical Examinations Three eyes each were examined by imniunohistochemistry at 3 days and at 1 week, and four eyes each were examined by immunohistochemistry at 2, 4, and 8 weeks after implantation. Briefly, each eye was enucleated, and its cornea, lens, and vitreous were removed. The eye was bisected, and the site of the lesion was delimited by sharp cuts. One half of the lesion was fixed in half-strength Karnovsky's solution (2.5% glutaraldehyde and 2% formaldehyde) in 0.1 M cacodylate buffer (pH 7.4). The tissues were dehydrated in a series of graded alcohols and embedded in glycol methacrylate; 2- to 3-^m sections were stained with periodic acid-Schiff or hematoxylin-eosin. For immunohistochemical examinations, the other half of the tissue was embedded in optimal cutting temperature (O.C.T.) compound (Tissue-Tek; Miles, Elkhart, IN) and frozen in liquid nitrogen. Cryosections (8 (xtxi) were cut and stored at — 70°C. Thawed sections were air-dried, fixed with acetone for 5 minutes, and washed with PBS (pH 7.4). The sections were blocked for 15 minutes with 1% bovine serum albumin (Sigma, St. Louis, MO) in PBS after blocking of endogenous peroxidase by 0.3% hydrogen peroxide. The specimens were incubated for 30 minutes with the primary antibody and then washed for 15 minutes with PBS. Sections were stained using the avidinbiotin complex (Vectastain ABC Elite Kit; Vector Laboratories, Downloaded From: http://iovs.arvojournals.org/ on 08/03/2017 FIGURE 4. Light micrograph of the lesion 4 weeks alter implantation. GFAP-positive Miiller cells are seen traversing the retina. A GFAPpositive plaque is seen between the retina and the neovascular membrane. Original magnification, X500. IOVS, February 1999, Vol. 40, No. 2 mononuclear cells and no CD8-positive cells were seen. No CD31-positive endothelial cells were observed in the subretinal space at this time. One week after implantation, the microspheres were mainly surrounded by cytokeratin-positive RPE cells and RAMll-positive macropliages. GFAP-positive Miiller cells extended occasional processes into the subretinal space. A few CD4- or CD8-positive cells were observed. CD31-positive endothelial cells were not seen in the subretinal space. Two weeks after implantation, the microspheres were still observed but were decreased in number. CD31-positive endothelial cells were now seen in the subretinal space and formed neovascular channels (Fig. 2). The multilayered RPE (cytokeratin-positive) cells (Fig. 3) and endothelial cells formed a membrane-like structure. RAMll-positive macrophages were seen but were decreased in number compared with 1 week. Four weeks after implantation, many RPE (cytokeratinpositive) cells and some endotheiial (CD31-positive) cells were observed in subretinal membranes. Miiller (GFAP-positive) cells were adherent to the retinal surface of the membrane and only rarely extended into the subretinal membrane (Fig. 4). A few RAM11-positive macrophages were seen, but CD4- or CD8positive cells were sparse. A few microspheres were still present in some areas. Eight weeks after implantation, CD31-positive endothelial cells were still seen in a membrane that was largely covered with cytokeratin-positive RPE cells. In some areas, gliosis was observed on the membranes. DISCUSSION In this study, we examined the time course of cellular infiltration into the subretinal space caused by implantation of bFGFimpregnated gelatin microspheres. The time course can be divided into two phases: an early phase (foreign body reaction) and a late phase (neovascularization). During the early phase, within 1 week of implantation, microspheres were surrounded by many RPE cells and macrophages and did not appear to be degraded. No neovascularization was observed. It is likely that the microspheres did not release a large amount of bFGF during this phase because bFGF is physically immobilized in the matrix of the cross-linked gelatin and is released as a biologically active form only after enzymatic degradation of the cross-linked gelatin.4 During the late phase, starting 2 weeks after implantation, the number of microspheres decreased, and endothelial cells migrated from the choroid forming with RPE cells a neovascular membrane. It seems likely that the microspheres were enzymatically degraded by RPE cells or macrophages and released bFGF, resulting in proliferation of RPE cells and choroidal endothelial cells. At 8 weeks after implantation, the neovascular membranes were covered with RPE cells and appeared to be adherent to the retina by a gliotic band. The microspheres were largely degraded. Injection of microspheres alone was also associated with a mild cellular reaction composed of macrophages and RPE cells; however, this was not associated with neovascularization.4 The microspheres used in this study were relatively large, with an average diameter of 40 /xm, and may have caused a foreign body reaction. Subretinal injection itself may also result in damage to outer segments of the photoreceptors or partial dispersion of RPE cells that could incite RPE proliferation or a gliotic response. Downloaded From: http://iovs.arvojournals.org/ on 08/03/2017 Reports 527 During the late phase, neovascularization occurs, and the new vessels show an association with many RPE cells. Gelatin itself enhances phagocytosis of microspheres by macrophages through its opsonizing ability. Some microspheres may be ingested and degraded by intracellular lysosomal enzymes, and others may be degraded by gelatinases in the extracellular space. RPE and endothelial cells both have been shown to be capable of gelatinase (matrix metalloproteinase-2 and -9) secretion in vitro.5 The neovascularization may be a result of several soluble factors. A direct or indirect effect of bFGF itself on choroidal vessels is most likely. bFGF is a prominent promoter of endothelial cell migration and proliferation and stimulates the angiogenic process. Release of bFGF from the microspheres requires microsphere degradation that may occur by phagocytosis by macrophages6 or RPE7 or by proteolytic digestion. A second source of angiogenic factors is from soluble products of infiltrating macrophages6 and RPE. In particular, migrating RPE cells have been shown to be a source of the strongly angiogenic vascular endothelial growth factor (VEGF) in human CNVM.3 The presence of VEGF or other secondary angiogenic factors derived from either RPE or macrophages will be an important focus of future study. bFGF may also promote the development of the SRN lesion by stimulating growth of the nonendothelial cells. bFGF is mitogenic for both RPE8 and glial cells9 and thus may have an important role in promoting the relatively high cellularity found in the lesions. Therefore, although it is clear that the presence of bFGF in the microspheres was the determinant directly associated with the strong subretinal angiogenic response, it is possible that the cellular or physical reactions induced by the microspheres themselves may contribute to the stimulus. Choroidal neovascular membranes in this model show immunohistochemical characteristics similar to those of CNVMs surgically excised from patients with ARMD.1"3 In these membranes, the stromal cells are predominantly composed of RPE and vascular endothelial cells; however, small numbers of macrophages are also seen. Most CNVMs in ARMD include prominent extracellular matrices (ECMs) rich in collagen, laminin, and fibronectin.' CNVMs in this model appear to be composed primarily of cellular components with less prominent ECMs. The strong proliferative response of RPE cells to wound healing in the rabbit may explain why RPE cells are especially prominent in the membranes in this model.10 Identification of ECM components in this model may require the increased sensitivity of immunohistochemical methods. It is possible that the CNVMs of ARMD contain more ECM than in this model because the angiogenic process in ARMD is more protracted. GFAP-positive Miiller cells migrated into the subretinal space after 7 days and formed a gliotic adhesion between the retina and the SRN membrane that was most prominent at 8 weeks. The photoreceptors in the area in which microspheres accumulated were mostly degenerated and were replaced with proliferated Miiller cells. It seems likely that the mechanical damage of microsphere uijection or the interference of the normal metabolism of RPE cells by the accumulated microspheres induced the degeneration of photoreceptors. Miiller cells tend to proliferate into the subretinal space and form multiple layers after the degeneration of photoreceptors." bFGF from the microspheres may also stimulate the proliferation of Miiller cells.9 528 IOVS, February 1999, Vol. 40, No. 2 Reports We have previously demonstrated in this model a clinically relevant characteristic: CNVMs leak fluorescein and can be detected by fluorescein angiography.4 We now show that the immunohistochemical characteristics of cellular components involved in CNVMs of this model bear a striking resemblance to those in ARMD. Further studies of the angiogenic responses in this model may provide some clues leading to new therapeutic approaches to SRN. Acknowledgments The authors thank Thomas E. Ogclen, MD, PhD, for his helpful comments, Susan Clarke for editorial assistance, and Fernando Gallardo for technical assistance. 4. 5. 6. 7. 8. References 1. Grossniklaus HE, Martinez JA, Brown VB, et al. Immunohistochemical and histochemical properties of surgically excised subretinal neovascular membranes in age-related macular degeneration. AmJ Ophthalmol. 1992; 114:464 - 472. 2. Seregard S, Algvere PV, Berglin L. Immunohistochemical characterization of surgically removed subfoveal fibrovascular membranes. Graefes Arch Clin Exp Ophthalmol. 1994;232: 325-329. 3. Lopez PF, Sippy BD, Lambert HM, Thach AB, Hinton DR. Transdifferentiated retinal pigment epithelial cells are immunoreactive for vascular endothelial growth factor in surgically excised age-related A Possible Role for p^ 1 ** 4 in Neuronal Cell Death after Retinal Ischemia-Reperfusion Injury Sachiko Kuroiwa, Naomichi Katai, and Nagahisa Yoshimura study whether cell type-specific death occurs in retinal ischemia-reperfusion injury and the possible roles of pl6INK>l in the determination of cell death. PURPOSE. TO Retinal ischemia-reperfusion injury was induced in rats by a ligation method. After 1 hour of ischemia and a time of reperfusion that varied, rat eyes were enucleated. Cell death in the retina was studied by the TdT-dUTP terminal nick-end labeling method and propidium iodide (PI) staining. Electron microscopic observation of the retina was also performed. Immunohistochemical studies using antibodies against syntaxin and calbindin were performed to detect amacrine cells and horizontal cells, respectively, and immunohistochemical studies using an anMETHODS. 9. 10. 11. macular degeneration-related choroidal neovascularization. Invest Ophthalmol Vis Sci, 1996;37:855-868. Kimura H, Sakamoto T, Hinton DR, et al. A new model of subretinal neovascularization in the rabbit. Invest Ophthalmol Vis Sci. 1995;36:2110-2119. Hunt RC, Fox A, al Pakalnis V, et al. Cytokines cause cultured retinal pigment epithelial cells to secrete metalloproteinases and to contract collagen gels. Invest Ophthalmol Vis Sci. 1993:34:31793186. Sunderkotter C, Steinbrink K, Goebeler M, Bhardwaj R, Sorg C. Macrophages and angiogenesis./Leukoc Biol. 1994;55:4l0-422. Kimura H, Ogura Y, Moritera T, Honda Y, Tabata Y, Ikada Y. In vitro phagocytosis of polylactide microspheres by retinal pigment epithelial cells and intracellular drug release. Curr Eye Res. 1994; 13:353-360. Smith Thomas L, Richardson P, Parsons MA, Rennie IG, Benson M, MacNeil S. Additive effects of extracellular matrix proteins and platelet derived mitogens on human retinal pigment epithelial cell proliferation and contraction. Curr Eye Res. 1996;15:739-748. Lewis GP, Erickson PA, Guerin CJ, Anderson DH, Fisher SK. Basic fibroblast growth factor: a potential regulator of proliferation and intermediate filament expression in the retina. / Neurosci. 1992; 12:3968-3978. Lopez PF, Qiong Y, Kohen L, et al. Retinal pigment epithelial wound healing in vivo. Arch Ophthalmol. 1995; 113:14371446. Erickson PA, Fisher SK, Anderson DH, Stern WH, Borgula GA. Retinal detachment in the cat: the outer nuclear and outer plexiform layers. Invest Ophthalmol Vis Sci. 1983;24:927-942. tibody against pl6 INK4 were performed to study whether this cell cycle-related protein was expressed in dying cells. Most of the calbindin-positive horizontal cells in the outer aspect of the inner nuclear layer (INL) showed morphologic features of necrosis. In contrast, syntaxinpositive amacrine cells in the inner aspect of the INL showed features of apoptosis. Of 320 calbindin-positive horizontal cells, only 11 (3.4%) showed positive PI staining. Those calbindin-positive, horizontal cells were pl6 INK4 positive. In contrast, 746 of 910 (82.0%) syntaxinpositive amacrine cells showed condensed PI staining, and none were pl6 INK4 positive. RESULTS. Expression of pl6 1NK4 may regulate the fate of retinal neurons in ischemia-reperfusion injury, and cell type-specific death thus occurs in the retina after such injury. (Invest Ophthalmol Vis Sci. 1999;40:528-533) CONCLUSIONS. n experimental retinal ischemia-reperfusion injury, neuronal Ilayercells mainly in the ganglion cell layer and the inner nuclear (INL) are damaged. Previous reports have shown the 1 From the Department of Ophthalmology, Shinshu University School of Medicine, Matsumoto, Japan. Supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports and Culture of the Japanese Government (10470567 to NY). Submitted for publication April 23, 1998; revised July 10 and September 11, 1998; accepted October 6, 1998. Proprietary interest category: N. Reprint requests: Nagahisa Yoshimura, Department of Ophthalmology, Shinshu University School of Medicine, n*-*—••-"•— 2 m Q/COI Japan. Downloaded From: http://iovs.arvojournals.org/ on 08/03/2017 occurrence of at least two, and possibly three, morphologically distinct types of cell death after such injury.' Using the same rat model, we have shown that occurrence of DNA fragmentation by TdT-dUTP terminal nick-end labeling (TUNEL) and also by gel electrophoresis of the genomic DNA.2 Therefore, the type of death in most of the affected cells in retinal ischemia-reperfusion injury can be classified as apoptosis. We have also shown that aberrant expression of cell cycle-related genes, especially c-jun and cyclin Dl, plays a role in the process of apoptosis.2