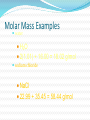* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Types of Changes in Matter
Brownian motion wikipedia , lookup
Electrolysis of water wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Computational chemistry wikipedia , lookup
Chemical plant wikipedia , lookup
Chemical industry wikipedia , lookup
Particle-size distribution wikipedia , lookup
Click chemistry wikipedia , lookup
Rate equation wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Drug discovery wikipedia , lookup
Chemical potential wikipedia , lookup
Electrochemistry wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Marcus theory wikipedia , lookup
Elementary particle wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Isotopic labeling wikipedia , lookup
History of chemistry wikipedia , lookup
Molecular dynamics wikipedia , lookup
George S. Hammond wikipedia , lookup
Chemical reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
VX (nerve agent) wikipedia , lookup
History of molecular theory wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Transition state theory wikipedia , lookup
Stoichiometry wikipedia , lookup
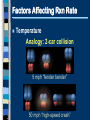
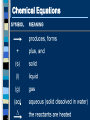
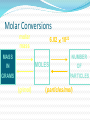
2.1 Science and Technology in Society 2.2 Changes in Matter 2.3 Balancing Chemical Reactions 2.4 Chemical Amount 2.5 Classifying Chemical Reactions 2.6 Chemical Reactions in Solution Chemical Reactions 2.2 Changes in Matter I II III IV V Types of Changes in Matter Physical Changes Change in State No new substance Types of Changes in Matter Nuclear Changes Changes within the nucleus Fusion and Fission (applets) Types of Changes in Matter Chemical Changes A change in chemical bonds Hoffman Demo Signs of a Chemical Change Production of heat and light Formation of a gas Formation of a precipitate Colour change Why Do Reactions Occur? The kinetic molecular theory states that matter is made up of tiny particles in continuous random motion. The average kinetic energy (the energy of motion) of the particles depends on the temperature of the particles. Solid Liquid Gas Types of Particle Motion Translational motion: the motion of a particle in a straight line Rotational movement: a spinning or turning of a molecule. Motion Continued: Vibrational movement: the back and forth motion within a molecule These types of particle movement can be discussed with the three states of matter KMT Continued: The particles this theory refers to may be atoms (Na), ions (Na+) or molecules (CO2). As the particles move, they collide with each other and objects in their path. Collision Theory Reaction rate depends on the collisions between reacting particles. Successful collisions occur if the particles... collide with each other have the correct orientation have enough kinetic energy to break bonds Collision Theory Activation Energy (Ea) minimum energy required for a reaction to occur Activation Energy Collision Theory Activation Energy depends on reactants low Ea = fast rxn rate Ea Factors Affecting Rxn Rate Surface Area high SA = fast rxn rate more opportunities for collisions Increase surface area by… - using smaller particles - dissolving in water Factors Affecting Rxn Rate Concentration high conc = fast rxn rate more opportunities for collisions Factors Affecting Rxn Rate Temperature high temp = fast rxn rate high KE - fast-moving particles - more likely to reach activation energy Factors Affecting Rxn Rate Temperature Analogy: 2-car collision 5 mph “fender bender” 50 mph “high-speed crash” Factors Affecting Rxn Rate Catalyst substance that increases rxn rate without being consumed in the rxn lowers the activation energy Burn a sugar cube with a catalyst Exothermic Reaction reaction that releases energy products have lower energy than reactants energy released 2H2(l) + O2(l) 2H2O(g) + energy Endothermic Reaction reaction that absorbs energy reactants have lower energy than products energy absorbed 2Al2O3 + energy 4Al + 3O2 Law of Conservation of Mass mass is neither created nor destroyed in a chemical reaction total mass stays the same atoms can only rearrange 4H 36 g 2O 4H 2O 4g 32 g Chemical Reactions 2.3 Balancing Chemical Reaction Equations I II III IV V Chemical Equations Chemical reactions are represented by chemical equations A chemical equation lists all of the compounds that participate in the reaction. Chemical Equations A+B C+D REACTANTS PRODUCTS Chemical Equations Writing Equations 2H2(g) + O2(g) 2H2O(g) Identify the substances involved. Use symbols to show: How many? - coefficient Of what? - chemical formula In what state? - physical state Writing Equations Two atoms of aluminum react with three units of aqueous copper(II) chloride to produce three atoms of copper and two units of aqueous aluminum chloride. • How many? • Of what? • In what state? 2Al(s) + 3CuCl2(aq) 3Cu(s) + 2AlCl3(aq) Describing Equations Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) • How many? • Of what? • In what state? One mole of solid zinc reacts with two moles of aqueous hydrochloric acid to produce one mole of aqueous zinc chloride and one mole of hydrogen gas. Balancing Steps 1. Write the unbalanced equation. 2. Count atoms on each side. 3. Add coefficients to make #s equal. Coefficient subscript = # of atoms 4. Reduce coefficients to lowest possible ratio, if necessary. 5. Double check atom balance!!! Helpful Tips Balance one element at a time. Update ALL atom counts after adding a coefficient. If an element appears more than once per side, balance it last. Balance polyatomic ions as single units. “1 SO4” instead of “1 S” and “4 O” Balancing Example Aluminum and copper(II) chloride react to form copper and aluminum chloride. 2 Al + 3 CuCl2 3 Cu + 2 AlCl3 2 1 Al 1 2 3 1 Cu 1 3 6 2 Cl 3 6 The Mole What is the Mole? A counting number (like a dozen) Avogadro’s number (NA) 1 mol = 6.02 1023 items A large amount!!!! A mole of a compound is an observable quantity. It can be weighed and used in experiments. LEFT: Proceeding clockwise from the top, samples containing one mole each of copper, aluminum, iron, sulfur, iodine, and (in the center) mercury. A. What is the Mole? 1 mole of hockey pucks would equal the mass of the moon! 1 mole of basketballs would fill a bag the size of the earth! 1 mole of pennies would cover the Earth 1/4 mile deep! One mole = 6.02 x 1023 atoms, molecules, or ions (Avogadro’s number) When coefficients are used to balance chemical equations, they express a mole-to-mole ratio of the products and reactants. These numbers do not represent the exact number of moles for the reactants or products, but rather give a ratio we can use to compare quantities. Molar Mass Mass of 1 mole of an element or compound. Atomic mass tells the... atomic mass units per atom (amu) grams per mole (g/mol) Round to 2 decimal places Molar Mass Examples carbon 12.01 g/mol aluminum 26.98 g/mol zinc 65.39 g/mol Molar Mass Examples water H2O 2(1.01) + 16.00 = 18.02 g/mol sodium chloride NaCl 22.99 + 35.45 = 58.44 g/mol Molar Mass Examples sodium bicarbonate NaHCO3 22.99 + 1.01 + 12.01 + 3(16.00) sucrose = 84.01 g/mol C12H22O11 12(12.01) + 22(1.01) + 11(16.00) = 342.34 g/mol Molar Conversions molar mass 6.02 1023 MASS NUMBER MOLES IN GRAMS OF PARTICLES (g/mol) (particles/mol) Molar Conversions n = m/M n = number of moles m = mass (g) M = Molar Mass (g/mol) Molar Conversions p = n * NA n = number of moles p = particles NA = Avogadro's number (6.02 x 1023 particles per mole) Molar Conversion Examples How many moles of carbon are in 26 g of carbon? 26 g C 1 mol C 12.01 g C = 2.2 mol C Molar Conversion Examples How many molecules are in 2.50 moles of C12H22O11? 6.02 1023 2.50 mol molecules 1 mol = 1.51 1024 molecules C12H22O11 Molar Conversion Examples Find the mass of 2.1 1024 molecules of NaHCO3. 2.1 1024 molecules 1 mol 84.01 g 6.02 1023 1 mol molecules = 290 g NaHCO3 Chemical Reactions 2.5 Classifying Chemical Reactions I II III IV V Combustion the burning of any substance in O2 to produce heat A + O2 B CH4(g) + 2O2(g) CO2(g) + 2H2O(g) Combustion Products: contain oxygen hydrocarbons form CO2 + H2O 4 Na(s)+ O2(g) 2 Na2O(s) C3H8(g)+ 5 O2(g) 3 CO2(g)+ 4 H2O(g) Synthesis/Formation the combination of 2 or more substances to form a compound only one product A + B AB Synthesis H2(g) + Cl2(g) 2 HCl(g) Synthesis Products: ionic - cancel charges covalent - hard to tell 2 Al(s)+ 3 Cl2(g) 2 AlCl3(s) Decomposition a compound breaks down into 2 or more simpler substances only one reactant AB A + B Decomposition 2 H2O(l) 2 H2(g) + O2(g) Decomposition Products: Binary ionic - break into elements others - hard to tell 2 KBr(l) 2 K(s) + Br2(l) Single Replacement one element replaces another in a compound metal replaces metal (+) nonmetal replaces nonmetal (-) A + BC B + AC Single Replacement Cu(s) + 2AgNO3(aq) Cu(NO3)2(aq) + 2Ag(s) Double Replacement ions in two compounds “change partners” cation of one compound combines with anion of the other AB + CD AD + CB Double Replacement Pb(NO3)2(aq) + K2CrO4(aq) PbCrO4(s) + 2KNO3(aq) Double Replacement Products: switch negative ions one product must be insoluble (check solubility table) Pb(NO3)2(aq)+ 2KI(aq) PbI2(s)+2KNO3(aq) NaNO3(aq)+ KI(aq) N.R. Chemical Reactions in Solution Section 2.6 Why in Solution? Often reactions are carried out in solution in order to speed up the rate of reaction. Dissolving something makes the particles smaller (increase surface area) We can add heat or stir May be a way to dilute volatile substances to make the reaction safer. Solubility Refers to an ionic compound’s ability to dissolve in water If a compound is soluble (completely dissolves in water) it is considered to be aqueous. Aqueous solutions are labeled with the letters (aq) after the chemical formula Example: NaCl(aq) If an ionic compound is not soluble in water, it will remain as a solid (it will form a precipitate). Solids are labeled with a (s) after the compound formula. Example: Ag2SO4(s) How to use the Solubility Table The solubility table will tell us if an ionic compound is soluble in water. Steps to follow: 1) Determine the non-metal of your compound and find it on the top of the chart. 2) Look beneath the non-metal on the chart. If the metal it is matched with is in the section labeled “high solubility”, the compound is soluble in water. - the compound is then labeled with a (aq) 3) If the metal is located in the section labeled “low solubility”, your compound is not soluble in water. -the compound is then labeled with a (s) SEE PAGE 8 IN YOUR DATA BOOKLET Examples: Are these ionic compounds soluble in water? Ag+ and SO42- K+ and NO3 - Ca2+ and S2- Li+ and OH-