* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download What Are Electromagnetic Waves?
Survey
Document related concepts
Transcript
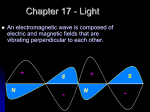
ELECTROMAGNETIC WAVES SONNY P. DE LEON PRESENTOR What Are Electromagnetic Waves? Electromagnetic Waves, like other kind of waves, are caused by vibrations. These waves are produced by the vibrations of electric charges in atoms. The energy in electromagnetic waves is made up of electric and magnetic fields that vibrate at right angles to each other. Wavelength usually stated in units of nanometers (one-thousandth of a micrometer) for the visible light portion of the spectrum. The wavelength is defined as the distance between two successive peaks (or valleys) of the waveform (see Figure 1). The corresponding frequency of the radiated wave, which is the number of sinusoidal cycles (oscillations or complete wavelengths) that pass a given point per second, is proportional to the reciprocal of the wavelength. The longer wavelengths correspond to lower frequency radiation and shorter wavelengths correspond to higher frequency radiation. Frequency is usually expressed in quantities of hertz (Hz) or cycles per second (cps). Amplitude is the height of the wave. Frequency One way of measuring the energy of an Electromagnetic wave is by measuring its frequency. Frequency refers to the number of waves a vibration creates during a period of time – like counting how frequently cars pass through an intersection in a given time. In general, the higher the frequency, or number of waves, the greater the energy of the radiation. Wavelength and frequency are inversely related, meaning that the greater the length of the wave, the lower its frequency will be. Likewise, if the frequency is high, the wave must be shorter. Another way to think of it is: the shorter the wave, the higher the energy. RADIO WAVES Radio waves have the longest wavelengths in the Electromagnetic Spectrum. These waves can be longer than a football field or as short as a football, having a wavelength of 1 mile (1.5 km) or more. Since these are the longest waves, they have lowest energy and are associated with the lowest temperature. MICROWAVE Microwaves have shorter wavelengths than radio waves that they are easily absorbed by water, which heat the food we eat. Ranges from onetenth of a mm to 1 cm. They are also used in telecommunication. Microwaves are good for transmitting information from one place to another because microwave energy can penetrate haze, light rain and snow, clouds, and smoke. • Shorter microwaves are used in remote sensing. These microwaves are used for radar like the Doppler radar used in weather forecasts. Microwaves, are used for radar, are just a few inches long. This microwave tower can transmit information like telephone calls and computer data from one city to another. INFRARED WAVE Infrared Waves have both long wavelengths and short wavelengths, which span from 700 nm-1 mm (from the width of a pinpoint to the size of small plant seeds). Infrared waves with long wavelengths are different from infrared waves with short wavelengths. Infrared long wavelengths can be detected as heat. Your radiator or heater gives off these long infrared waves. We call these thermal infrared or far infrared waves. The sun gives off infrared waves with shorter wavelengths. Infrared light lies between the visible and microwave portions of the Electromagnetic Spectrum. They are the deep red rays you get from a heat lamp. Shorter, near infrared waves are not hot at all-in fact you cannot even feel them. These are the ones used by your TV's remote control. Visible Light Visible Light Waves are the only Electromagnetic waves we can see. We see these waves as the colors of the rainbow. Each color has a different wavelength. Red has the longest wavelength and violet has the shortest wavelength. These waves combine to make white light. Their wavelengths are in the range from 400-700 nanometers (from size of a molecule to a protozoan). Ultraviolet Waves Ultraviolet waves have wavelengths of 10-100nm (about the size of a virus). Ultraviolet light has shorter wavelengths than visible light. Though these waves are invisible to the human eye, some insects, like bumblebees, can see them. Scientists have divided the ultraviolet part of the spectrum into three regions: the near ultraviolet, far ultraviolet, and the extreme ultraviolet. The three regions distinguished by hoe energetic the ultraviolet radiation is, and by the “wavelength” of the ultraviolet light which is related to energy. The near ultraviolet, abbreviated NUV, is the light closest to optical or visible light. The extreme ultraviolet, abbreviated EUV, is the ultraviolet light closest to X-rays, and is the most energetic of the three types. The far ultraviolet, abbreviated FUV, lies between the near and extreme ultraviolet regions. It is the least explored of the three regions X-rays X-rays range in wavelength from 0.001-10 nm (about the size of an atom). As the wavelengths of light decrease, they increase in energy. Xrays have smaller wavelengths and therefore higher energy than ultraviolet waves. We usually talk about X-rays in terms of their energy rather than wavelength. This is partially because X-rays have very small wavelengths. It is also because X-ray light tends to act more like a particle than a wave. X-ray detectors collect actual photons of X-ray light which is very different from the radio telescopes that have large dishes designed to focus radio waves! X-rays are so powerful that they pass easily through the skin allowing doctors to look at our bones. • X-rays were first observed and documented in 1895 by Wilhelm Conrad Roentgen, a German scientist who found them quite by accident when experimenting with vacuum tubes. • Your doctor uses them to look at your bones and your dentist to look at your teeth. • X-rays go through the body and are used for medical purposes. GAMMA RAYS Gamma rays have the shortest wavelengths – 0.001 nm (about the size of an atomic nucleus). This is the highest frequency and most energetic region of the Electromagnetic Spectrum. Gamma rays are the result from nuclear radiations taking place in objects such as pulsars, quasars and black holes. Gamma-rays can kill living cells, a fact which medicine uses to its advantage, using gammarays to kill cancerous cells.