* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download EDS Weathering - HCC Learning Web
Anoxic event wikipedia , lookup
Water pollution wikipedia , lookup
Global Energy and Water Cycle Experiment wikipedia , lookup
Post-glacial rebound wikipedia , lookup
Overdeepening wikipedia , lookup
History of geology wikipedia , lookup
Geomorphology wikipedia , lookup
Algoman orogeny wikipedia , lookup
Age of the Earth wikipedia , lookup
Marine geology of the Cape Peninsula and False Bay wikipedia , lookup
Tectonic–climatic interaction wikipedia , lookup
Provenance (geology) wikipedia , lookup
Composition of Mars wikipedia , lookup
Geochemistry wikipedia , lookup
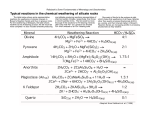
10 Weathering Earth’s Dynamic Systems Eric H Christiansen Brigham Young University The Nature of Weathering Earth's atmosphere and hydrosphere tend to weather rocks at surface: (1) by mechanical disintegration, in which rocks are broken by physical forces, (2) by chemical decomposition Why do rocks weather (break down)? Most rocks and minerals are formed at high temperatures and pressures They are in a state of equilibrium at the T and P of formation At the Earth’s surface, rocks and minerals are mechanically and chemically unstable Secondary minerals and structures form that are in equilibrium at the T, P, and fluid composition common at the Earth’s surface Weathering Chemical weathering The decomposition of rocks and minerals as chemical reactions alter them into new minerals stable at the Earth’s surface Physical weathering The disintegration or disaggregation of rocks by physically breaking them apart Physical Weathering Physical breakage of rocks into smaller pieces Ice Wedging Sheeting Biologic Thermal fracturing No change in chemical composition Ice Wedging Freeze - Thaw cycles are effective at breaking apart rocks Water expands when it freezes Volume increases by 9% The stress of expansion breaks the rock Ice melts and the water percolates deeper into the newly expanded cracks Angular blocks accumulate at base of cliff Ice Wedging This occurs where daily freeze thaw takes place, at least seasonally—not where permanently frozen. Ice Wedging Talus Cones Mount Timpanogos Ice Wedging Talus Cones Sheeting Release of confining pressure on rocks formed deep within the Earth Development of fractures and joints caused by expansion Rocks break along fractures and joints Fig. 10.4. Sheeting in granite Sheeting Half Dome, Yosemite Park Sheeting (Geologic Exfoliation) Other Forms of Physical Weathering Heat Heat causes rocks (most solids) to expand Rocks are poor conductors of heat Outer layer of rock that expands breaks off (spall) Crystal Growth Minerals precipitate along fractures Similar to ice wedging Other Forms of Physical Weathering Root Growth Roots may exert enormous forces in growing Root tips pressures may exceed 10,000 kg per square meter Seeds gather in cracks in rock and germinate Growing plant and roots slowly wedge rock apart Tree Roots, etc. Chemical Weathering Minerals are destroyed or altered by chemical reactions Dissolution Acid Hydrolysis Oxidation Dissolution Some minerals are soluble in water e.g., Halite - NaCl Minerals dissolve into constituent ions Ions removed with water by leaching Solubility of compound controls leachability Hydrolysis Completely dissolves Dissolution H+ attacks minerals by replacing other ions in the mineral structure Promotes dissolution Calcite hydrolysis by carbonic acid solution CaCO3 + H2CO3 = Ca+2 + 2HCO3- Dissolution and Secondary Minerals CO2 mixes with water to produce carbonic acid, H2CO3 Decaying organic matter produces acid Anthropogenic sources of acid Acid rain Dissolves and precipitates new minerals Acid Hydrolysis Hydrogen ions replace other ions in a mineral. Feldspar is the most abundant mineral in the crust. What is the most abundant sedimentary rock type? Acid Hydrolysis Plagioclase feldspar Acid Hydrolysis New “secondary” minerals may be created by this process H+ ion replaces the K+ ion in the feldspar structure K+ ion goes into the water solution Kaolinite, a clay mineral, formed 2KAlSi3O8 + 2H2CO3 + 9H2O = 2K+ + 2HCO3- + 4H4SiO4 + Al2Si2O5(OH)4 Solution and Secondary Minerals Oxidation Valence state increases Often associated with free O2 in the environment Iron is usually found as the Fe+2 ion in silicate minerals Exposed to the atmosphere it will oxidize to the Fe+3 ion Change in valence state disrupts crystal structure Oxidation works in combination with hydrolysis and dissolution Oxidation of olivine 2FeSiO4 + 4H2O + O2 = 2 Fe2O3 + 2H4SiO4 Trends in Chemical Weathering Alkali and alkaline earth elements removed into solution (Where?) Al, Si, and Fe are enriched in secondary minerals For example, Fe is stored in stable (insoluble) ferric oxides (rust) Secondary minerals have water and oxidized iron Increases in T and moisture increase weathering rates Resistance to Weathering: Relative stability of minerals Mechanical and Chemical Weathering Linked Principle: Mechanical weathering enhances chemical weathering by producing more surface area. Chemical weathering proceeds at the surface of a mineral. Differential Weathering Caused by variations in weathering rate Occurs over a broad range of scales Unusual weathering features Arches, spindles Differential Weathering Differential Weathering Differential Weathering Differential Weathering Products of Weathering Dissolved ions in solution Broken up rock material Secondary minerals Spheroidal rock forms Mechanical weathering--Fractures and joints Regolith: A blanket of broken up rock, clay, and soil that covers the bedrock. Soil—all soil is regolith, but not all regolith is soil! The Result of Weathering? Geometry of Weathered Rocks Fractures in rock form Generally form in groups Parallel joints Intersecting joints Cut large blocks into smaller blocks Then weathering eats away at margins making spheroids Products of Weathering Bedrock Regolith Soil: A special type of regolith Principle: Soils are more weathered at the top. Dissolved matter from the top of a soil column travels downward, and is deposited in the lower portions. Fig. 10.14 Soil Horizons Common soil profile A Horizon A dark gray to black horizon, rich in organic matter. Leaching carries dissolved ions and fine particles B Horizon Accumulates leached material from above Enriched in clay minerals and oxides C Horizon Parent rock in various stages of weathering Climate & Weathering Climatic conditions strongly influence weathering reactions Amount of rainfall Most reactions need water Average temperature Increase of 10oC doubles reaction rate 10 Weathering Physical weathering is the mechanical fragmentation of rocks from stress acting on them. Ice wedging may be the most important type. Chemical weathering involves chemical reactions with minerals that progressively decompose the solid rock. The major types of chemical weathering are dissolution, hydration, and oxidation. Joints and fractures facilitate weathering because they permit water and gases in the atmosphere to attack a rock body at considerable depth. They also greatly increase the surface area on which chemical reactions can occur. The major products of weathering are spheroidal rock forms, a blanket of regolith, and dissolved ions. Soil is the upper part of the regolith--a mixture of clay minerals, weathered rock particles, and organic matter. Climate greatly influences the type and rate of weathering. The major controlling climatic factors are precipitation and temperature. 10 Weathering: Expected Learning Outcomes Recognize bedrock and regolith from observations in the field or from photographs of the landscape. Explain the mechanics of ice wedging and the factors that control its functioning. Show by simple diagrams how joints influence weathering and why there is a universal tendency toward spheroidal weathering. Explain how climate controls types of weathering and resulting weathered products. Explain hydrolysis, dissolution, and oxidation of rock materials, and list the products of these reactions. List the major products of weathering