* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Block 1: Pathology Dr. Rosenzweig Test 1: Free Radicals Oxidative
Survey
Document related concepts
Biochemical cascade wikipedia , lookup
Paracrine signalling wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Biochemistry wikipedia , lookup
Gene regulatory network wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Signal transduction wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Proteolysis wikipedia , lookup
Metalloprotein wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Transcript
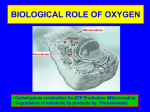
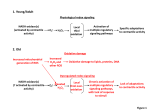
Block 1: Pathology Dr. Rosenzweig Test 1: Free Radicals Oxidative Stress injury by free radicals, particularly ROS, is important mechanism of cell damage in many pathological conditions o eg: chemical/radiation injury, ischemia-reperfusion injury, cellular aging, and microbial killing by phagocytes Free radicals: chemical spp have single Unpaired electron in outer orbit o highly reactive o attack organic/inorganic chemicals—proteins, lipids, carbs, nucleic acids—many of which are components of cell membranes and nuclei o some reactions= autocatalytic- propogating chain of damage ROS: type of oxygen-derived free radical whose role in cell injury is well-est. o produced normally during respiration, but typically removed by cellular defense systems o FRs may be present at small concentrations, but don’t cause damage o oxidative stress—excess of FRs o produced in large amts by leukocytes (neutros and macros) during inflammation; aimed at killing microbes o implicated in cell injury, cancer, aging, and some degenerative diseases such as Alzheimer’s 1 Block 1: Pathology Dr. Rosenzweig Test 1: Free Radicals Generation of FRs red-ox reactions that occur during normal metabolic processes molecular O2 reduced to water with 4 electrons small amounts of partially reduced intermediates are produced in which diff numbers of electrons have been transferred from O2 o superoxide anion (one electron) o hydrogen peroxide (two electrons) o hydroxyl ions (three electrons absorption of radient energy o ionizing radiation (UV, X-rays) can hydrolyze water to hydroxyl rad. and H FRs rapid bursts of ROS produced in activated leukocytes during inflammation o controlled reaction carried out by plasma membrane multiprotein complex o uses NADPH oxidase for redox rxn o some intracellular oxidases generate superoxide anion transition metals o Fenton reaction: (H2O2 + Fe2+ Fe-3++ .OH + OH-) o Fe/Cu donate/accept free electrons during intracellular reactions o most intracellular free iron = ferric (Fe3+); must be reduced to ferrous state to participate in Fenton Removal of FRs generally decay spontaneously superoxide anion O2 and H2O2 in presence of water conversion of H2O2 by SOD decomposition to H2O by glutathione peroxidase, catalase antioxidants: block FR formation/inactivate FRs o eg: lipid-soluble vitamins E and A; ascorbic acid and glutathione in cytosol Free Fe and Cu catalyze formation of ROS o normal circumstances: binds to storage and transport proteins (eg: transferring, ferritin, lactoferrin, and ceruloplasmin) prevents from participating in ROS generation Enzymes act as FR-scavengin systems breaks down H2O2 and superoxide anion o catalase: peroxisomes, decomposes H2O2 to O2 and H2O o Superoxidase dismutases (SODs): found in many cell types and convert superoxide to H2O2 o Glutathione peroxidase: protects against injury by catalyzing free radical breakdown H2O2 + 2GSH → GSSG [glutathione homodimer] + 2H2O, or 2˙OH + 2GSH → GSSG + 2H2O Pathologic Effects of FRs Lipid peroxidation in membranes o in presence of O2, FRs may cause peroxidation of lipids within plasma and organellar membranes 2 Block 1: Pathology Dr. Rosenzweig Test 1: Free Radicals Oxidative modification of proteins o FRs promote oxidation of AA side chains, formation of covalent protein-protein cross-links (disulfide bonds) and oxidation of protein backbone o oxidative modification of proteins may damage the active sites of enzymes, disrupt the conformation of structural proteins, and enhance proteasomal degradation of unfolded or misfolded proteins Lesions in DNA o can cause single- and double- strand breaks in DNA, cross-linking of DNA strands, and formation of adducts o implicated in cell aging and malignant transformation of cell Traditional thinking about FRs: cause necrosis (and they do) BUT can also trigger apoptosis 3