* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Atomic Theory
Survey
Document related concepts
Transcript
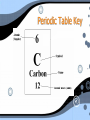
Atomic Theory Atomic Theory • Democritus- 440 B.C. • He proposed that if you kept cutting something in half you would eventually end up with an “uncutable” particle. Which he called an atom • Greek: Atomos - indivisible • Aristotle- 338-322 B.C • He disagreed with Democritus. He believed you would never end up with an indivisible particle. Atomic Theory • Dalton- 1808 • In search of understanding why elements combine in specific ratios he discovered through his experiments that this happens because they are made of individual atoms. • His theory states 3 things 1- Atoms are small and cannot be created, divided or destroyed. 2- Atoms of the same element are all alike 3-Atoms join with other atoms to make new substances Atoms Atomic Structure The Atom • Atoms are extremely small. • Atoms contain a nucleus. • Atoms are made of subatomic particles • Protons • Neutrons • Electrons How small is small? • If you could enlarge a penny until it was as wide as the continental United States(about 3000miles), each of its atoms would be only about 3 cm in diameter- about the size of a ping pong ball. Protons • • • • • Positively charged particles (+). Found in the nucleus. All protons are identical Each proton= 1 amu (atomic mass unit) Every atom of an element has the same number of protons. Neutrons • • • • Particles that have no charge Found in nucleus All neutrons are identical Protons & Neutrons are the most massive particles in the atom but located in a very small area (nucleus) Electrons • Negatively charged particles (e-) • Located outside of the nucleus in rings or levels called atomic clouds • Their mass is so small that it is usually considered zero. • It takes more than 1,800 electrons to equal the mass of one proton. • However electrons occupy most of an atoms volume. Some Things to Remember! • In an atom, the number of protons equals the number of electrons. As a result, the positive charge from protons equals the number of electrons which makes the atom neutral. • All atoms of an element have the same atomic number. Periodic Table Key Atomic Number • Atomic # = the number of protons in an atom. Atomic Mass • Atoms are too small to be measures in everyday units of mass (grams). • Scientists use units knows as atomic mass units (amu). • A proton or a neutron has a mass equal to about 1 amu. • Atomic Mass is equal to the sum of protons and neutrons in an atom. Isotopes • The number of neutrons in an atom can vary. • Atoms with the same number of protons and a different number of neutrons are called isotopes. • We identify an isotopes by the atoms mass. Isotopes Arrangement of Electrons • The first ring can only hold 2 electrons Arrangement of Electrons • The 2nd and 3rd rings can both hold 8 electrons