* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Crystal Field Theory part A
Survey
Document related concepts
Transcript
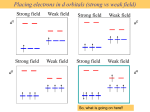
5.0 The Crystal-Field Theory By Prof Yeap Guan Yeow 5.1 Introduction This theory emphasizes on the electrostatic bonding of the chemical bond in a complex between the positive nucleus on the metallic ion with the negative charge on the electrons coming from the ligands. The diagram shows that electrons from the ligands causes electrostatic field around the metallic ion Complex M There are three interactions between the ligand field with the metallic atom (i) Ligand field with s orbital (ii) Ligand field with p orbital (iii) Ligand field and d orbital (i) Ligand field with s orbital For 4s orbital, because of its spheric shape, all the ligands that enter the octahedral field or in whatever arrangement will experience the same electrons repulsion in every direction in Cartes system . (ii) Ligand field with p orbital p orbitals directed to x, y and z axis causing the charge density to face along the Cartes axises. p orbitals in the three directions on the Cartes axis that interact with the ligands will experience the same electron repulsion. As a result, the levels of p orbitals will be elevated and are in the degeneration state. (iii) Ligand field and d orbital For d orbital, the scattering of electron density for the five orbitals can be shown in the diagrams below: _ dz2 and dx2-y2 orbitals have the charge density which is directed to (i) z axis , (ii) x and y axis, respectively. _ The lobes for the three dxy, dxz, and dyz orbitals orbitals are placed 45° in between the axes. 5.2 Types of Complex 5.2.1 Octahedral complex, ML6 (a) As the set of dz2, dx2-y2 and the set of dxy, dxz and dyz orbitals are oriented in different manner in the space, therefore, these two sets of orbitals will experience different electronic repulsion. (b) Greater electron repulsion in the dz2 and dx2-y2 orbitals set compared to dxy, dxz and dyz orbitals. (c) As a result, the levels of dz2 and dx2-y2 orbitals are elevated (destabilized). (d) the level of dxy, dyz, dxz orbital are decreased (stabilized). d orbitals will not degenerate within the ML6 complex but will remain at the different energy levels as follows : eg orbital set t2g orbital set dz2, dx2-y2 dxy, dxz, and dyz The splitting of crystal field of ML6 complex can be represented by the following diagram. • The directory of d orbitals 5.2.2 Tetrahedral complex contradicts with those found in the octahedral complex. z • dz2 and dx2-y2 are not directed parallel with the charge of ligands and this causes the energy level to decrease. y x However, the difference in energy between the two d orbital sets in the tetrahedral complex is found to be lower as compared to the energy difference found in an octahedral complex . This phenomenon can be illustrated as follows: Note: The subscript g and u are not written for the tetrahedral complex as this complex does not have a symmetrical centre. 5.2.3 Other complexes Based on the tetrahedral and octahedral complexes, the explanations for other complexes (square planar, trigonal bipyramidal, and square pyramidal) can be obtained. The conclusion for every complex can be shown in the following diagram. 5.3 High spin-Low-spin Complexes The Crystal Field or Ligand Field Theory can be used to elaborate on the existence of high spin and low spin complexes. For octahedral complex: d1 – d10, the occupancy of the d orbitals by electrons is categorized according to its ability to do so. (a) For d1 , d2 , d3 , d8 , d9 and d10, electrons are filled in between t2g and eg orbital by one possible method only. d1 d2 d3 u= 1.7 2 .8 3.9 d8 d9 2.8 1.7 d10 0 (b) For d4, d5, d6 and d7, electrons are filled in between t2g and eg orbitals by two possible methods. In this case, it is clear that two complexes exist; the high spin and low spin complexes. A main factor determining the formation of high spin or low spin complex is the splitting of ligand field parameter Δ. The value of Δ can be characterized based on the comparison with the energy value for pairing up the electrons (P). There are two situations in which either a high spin or low spin complex will be obtained. There are two situations in which either a high spin or low spin complex will be obtained. If Δ > P ; high field (strong) → low spin complex If Δ < P ; low field (weak ) → high spin complex where P = pairing energy For example, For low spin complexes: [Co(NH3)6]3+ (t2g6) There are three pairs of electrons residing in t2g. 5.4 Spectrochemical series Spectrochemical series is a series for different types of ligands which give different effect on the field of ligand and central metallic ion. The differences are caused by the different ∆ magnitudes (10Dq) and it depends on the type of ligands. The value of Δ can be determined experimentally through the electron spectrum analysis obtained for the transition metal complexes. The investigation on the effect of different series of ligands on different metallic ions has been summarized in the following table. Δ (cm2) for octahedral complex Based on the above table, we can see an obvious trend as follows: (i) For a certain ligand, Δ among transition metal ions will not change much in the same oxidation state. For example, Δ for hydrated M2+ is found in the range of 7500-12000 cm-1. (iii) For a certain ligand and stereochemical, the metallic ions can be arranged following the increasing ∆. This order is independent on the natural characteristics of the ligands. (ii) For a certain ligand, Δ increase if the oxidation state of the metal increases. For example, Δ for hydrated M3+ is found in the range of 14000-25000 cm-1. (iv)For a certain metallic ion, the ligands can be arranged according to the natural characteristics of the metal. Both characteristics spelt in (iii) and (iv) are known as spectrochemical series. In general, spectrochemical series for metallic ions can be shown as follows: Mn2+ < Ni2+ < Co2+ < Fe2+ < V2+ < Fe3+ < Cr3+ < V3+ < Co3+ As for ligands, the series is as follows: I- < Br- < CI- < S2- < F- < OH- < CH3COO- < C2O42- = O2< H2O < py = NH3 < en < bipy < phen< PR3 < CO = CN Question: Why do ligands F- and CN- that have negative charges are found in the position of the weak and strong series? This can be explained using the π-bond effect and can be elaborated through the following diagrams. (a) For complex that contains M-F bond e g* e g* ( t2g ) t 2g (i) Ligands donate electrons to the central metal M through π orbital on ligand F. (ii)Electrons from the set orbital t2g that are originated from the metallic ions are filled in the anti-bonding π* molecular orbital which is at a higher energy level as compared to t2g (iii)The effect is the reduction of ∆. (b) For a complex which contains M-CN bond e g* e g* t2g t2g (i) The ligands have high-energy π* orbital which is empty (CO, CNand H2C=CH2 are ligands that have the empty π* orbitals ) (ii) As a result, the density of electrons from central metal M can be donated to the ligands through the back-bonding. (iii) Electrons that are filled in π(t2g) orbital are from metal. (iv) The effect is the stabilization of t2g orbitals. • For octahedral complex, for example d1 complex: electrons place themselves at t2g orbital which is at the energy level Δo less than the energy level of d orbital which does not undergo splitting. • The additional stabilization caused by the splitting of d orbital is known as the crystal field stabilization energy (CFSE). • Every electron in the t2g orbital set contribute -Δo for (CFSE). However, electrons in the eg orbital set resides at the higher energy level from d orbital which does not undergo splitting and every electron contributes Δo to CFSE.