* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Which scientist developed the quantum mechanical model of the
Survey
Document related concepts
Particle in a box wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Matter wave wikipedia , lookup
Molecular orbital wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Wave–particle duality wikipedia , lookup
Hydrogen atom wikipedia , lookup
Electron scattering wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Tight binding wikipedia , lookup
Atomic orbital wikipedia , lookup
Transcript
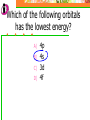
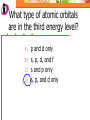
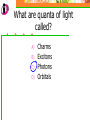
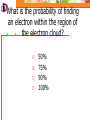
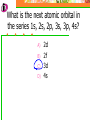
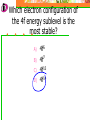
Which scientist developed the quantum mechanical model of the atom? A.) Bohr B.) Rutherford C.) Schrodinger D.) Heisenberg Draw a wave that shows low energy and gradually changes to high energy. Label the following on your wave: Crest Trough Amplitude Wavelength Which of the following orbitals has the lowest energy? A) B) C) D) 4p 4s 3d 4f 2 2n The formula represents _________. A) B) C) D) The number of sublevels in any energy level The maximum number of electrons that can occupy an energy level The number of orbitals None of the above What type of atomic orbitals are in the third energy level? A) B) C) D) p and d only s, p, d, and f s and p only s, p, and d only The quantum mechanical model of the atom_______. A) B) C) D) Was proposed by Hund Defines the exact path of an electron Is concerned with the probability of finding an electron in a certain position Was proposed by Bohr What is the maximum number of d orbitals in a principal energy level? A) B) C) D) 5 10 7 2 Write the complete electron configuration for titanium. The principal quantum number indicates what property of an electron? A) B) C) D) Speed Orbital shape Spin Energy level Which one of the following electron configurations is most stable? A) B) C) D) 5s1 5s3 5s2 5s4 4d5 4d3 4d4 4d2 What color of visible light has the shortest wavelength? A) B) C) D) Blue Green Yellow Orange Draw the orbital diagram for silicon. What are quanta of light called? A) B) C) D) Charms Excitons Photons Orbitals The atomic emission spectra of a sodium atom on earth and of a sodium atom in the sun would be _______. A) B) C) D) The same The same as each other only in UV range The same as those of several other elements Different from each other A wave has wavelength= 2 5.80 x 10 nm Convert this wavelength to centimeters. The lowest energy state of an atom is called the ______. A) B) C) D) Excited state Dependent state Independent state Ground state What is the approximate energy of a photon having a frequency of 4.0 x 107 Hz ? What is the approximate frequency of a photon having an energy of 5.25 x 10-24 J ? What is the difference between a 2s and a 3s orbital? A) B) C) There is no difference More electrons can occupy a 3s The 3s is larger in volume and a greater distance from the nucleus than a 2s Write Noble gas configurations for the following elements: A) B) C) D) Nb (41) At (85) Ag (47) Cm (96) ANSWERS A) B) C) D) Nb (41) At (85) Ag (47) Cm (96) [Kr] 5s2 4d3 [Xe] 6s2 4f14 5d10 6p5 [Kr] 5s1 4d10 [Rn] 7s2 5f8 What is the probability of finding an electron within the region of the electron cloud? A) B) C) D) 50% 75% 90% 100% What is the next atomic orbital in the series 1s, 2s, 2p, 3s, 3p, 4s? A) B) C) D) 2d 2f 3d 4s What is the correct number of electrons in the outermost energy level of an oxygen atom? A) B) C) D) 2 4 6 8 Which of the following states that no more than two electrons can occupy an atomic orbital and that two electrons in the same orbital must have opposite spins? A) B) C) D) Hund’s rule Pauli exclusion principle Dalton’s theory Aufbau principle Which electron configuration of the 4f energy sublevel is the most stable? A) B) C) D) 4f6 4f7 4f13 4f14 A wave has wavelength= -7 5.80 x 10 m Convert this wavelength to nanometers. Which of the following electromagnetic waves have the highest frequencies? A) B) C) D) ultraviolet light waves X-rays Microwaves Gamma rays How do the energy differences between the higher energy levels of an atom compare with the energy differences between the lower energy levels of the atom? A) B) C) They are greater in magnitude. They are smaller in magnitude. There is no significant difference in the magnitudes of these differences. Things to know…….. 1. 2. 3. 4. Principles of electron configurations Explanation for discrete lines in atomic emission spectra You will be given c and h as well as a periodic table. Hydrogen’s spectrum and series of lines Suggested Book Problems Pg. 149 22,25,27,29,30,31,33,35,37,39,40,41, 44,49,50,55,58,59,61,63,65,68