* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Assessment of a right ventricular metastasis using contrast
Heart failure wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Cardiothoracic surgery wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Coronary artery disease wikipedia , lookup
Electrocardiography wikipedia , lookup
Cardiac surgery wikipedia , lookup
Jatene procedure wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Myocardial infarction wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
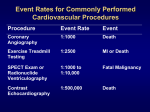
European Journal of Echocardiography (2008) 9, 326–328 doi:10.1093/ejechocard/jen067 Assessment of a right ventricular metastasis using contrast echocardiography perfusion imaging Sherif E. Moustafa, Claude Sauvé, and Robert Amyot* Department of Cardiology, Hôpital du Sacré-Coeur de Montréal, University of Montreal, 5400 Boul. Gouin W, Montreal, Quebec H4J 1C5, Canada Received 30 August 2007; accepted after revision 6 October 2007 KEYWORDS Contrast echocardiography; Perfusion imaging; Papillary thyroid carcinoma; Intracardiac metastasis Cardiac metastases of thyroid carcinomas are rarely diagnosed ante mortem and infrequently reported in the literature. A 68-year-old man with known papillary thyroid carcinoma presented to the hospital with progressive shortness of breath. A transthoracic echocardiogram revealed a right ventricular mass. Contrast echocardiography perfusion imaging was used to evaluate the vascularity of the mass. Cardiac tumours are an unusual pathologic finding and most often represent metastatic involvement of the heart rather than a primary cardiac tumour. Echocardiography is a useful technique for the detection and evaluation of cardiac tumours. Although the echo characteristics of an intracardiac mass and its associated cardiac findings may suggest an etiology, its specific histologic nature cannot be determined. The vascularity of a mass may help discriminate between benign tumours, malignant tumours, and thrombi. We describe the case of an end-stage papillary thyroid carcinoma with multiple metastases, including cardiac involvement discovered at echocardiography and further characterized by contrast echo perfusion imaging. Case presentation A very active 68-year-old man was admitted to the hospital with progressive shortness of breath over the previous 4 weeks. Eleven years earlier, in 1996, he had been diagnosed with end-stage papillary thyroid carcinoma for which he had undergone total right lobectomy of the thyroid and 131I therapy. Recurrence of his tumour in 2002 led to complete thyroidectomy and a second course of 131I therapy. Two years later, multiple metastases in the lungs, bones, and lymph nodes were discovered. Physical examination revealed a regular tachycardia at 107/min and a blood pressure of 117/80 mmHg. Blood oxygen saturation was 98% on 2 L of oxygen. The cardiovascular examination * Corresponding author. Tel: þ1 514 338 2222, ext 3809; fax: þ1 514 338 2381. E-mail address: [email protected] revealed normal heart sounds and no gallop or murmur. Jugular venous pressure was normal. Grade 2/4 peripheral oedema was present bilaterally. Rales were present bilaterally in the bases of the lungs. The liver was palpable 6 cm below the costal margin in the right mid-clavicular line and hard in consistency. The electrocardiogram showed left axis deviation and non-specific repolarization changes from V1 to V5. The chest X-ray revealed multiple metastases bilaterally. Laboratory data revealed a normochromic normocytic anaemia with haemoglobin at 112 g/L (normal: 140–174 g/L), a hypoalbuminaemia at 25 g/L (normal: 35–50 g/L), and a mild elevation of troponin T at 0.13 ng/ mL (normal, 0.03 ng/mL). Transthoracic echocardiography was technically difficult and demonstrated a heterogeneous, 40 mm 24 mm mass filling the right ventricular apex (Figure 1). Right ventricular contractility was normal at the basal and mid levels, but, adjacent to the mass, the apex was akinetic. Left ventricular dimensions and systolic function were normal with an estimated left ventricular ejection fraction of 65% and mild diastolic dysfunction. No other intracavitary mass or intramural lesions were detected. The inferior vena cava was normal in diameter and response to respiration. To assess the vascularity of the mass, multiple boluses of 0.1 to 0.2 mL of perflutren (DefinityTM, Bristol-Myers Squibb Medical Imaging, North Billerica, MA, USA) were administered intravenously. Images were acquired with an HDI 5000 system (Advanced Technology Laboratories, Seattle, WA, USA) using the low mechanical-index, pulse-inversion modality. Images were acquired with an HDI 5000 system (Advanced Technology Laboratories, Seattle, WA, USA) using the low Published on behalf of the European Society of Cardiology. All rights reserved. & The Author 2008. For permissions please email: [email protected]. Assessment of a right ventricular metastasis Figure 1 Transthoracic echocardiogram in modified apical fivechamber view showing an intracardiac mass filling the right ventricular apex (arrow). LV, left ventricle; RV, right ventricle. mechanical-index, pulse-inversion modality. Contrast enhancement of the mass was observed, confirming a highly vascularized lesion. Video intensity of the mass (mean ¼ 7.7 dB) was comparable with the video intensity of the adjacent myocardial tissue (mean ¼ 7.8 dB) and less than the signal from the right ventricular blood pool (mean ¼ 10.5 dB), confirming a highly vascularized mass (see Supplementary material online, Video 1). At this point, the patient was considered terminal and was treated palliatively. He died 2 weeks later. The autopsy was declined. Discussion Cardiac metastases have been described in autopsy series in up to 20% of patients with malignancies of other organs, and are up to 40 times more common than primary cardiac tumours.1,2 Metastatic tumours can involve the heart by direct invasion of adjacent malignancies, or by lymphatic or haematogenous dissemination of distant malignancies. Cardiac metastases originate most frequently from lung and breast carcinomas, lymphoma, leukaemia, and malignant melanoma.3,4 The malignant melanoma is the tumour with the highest rate of metastatic cardiac involvement, affecting the heart in more than 50% of cases in autopsy series.3,5 The most common echocardiographic finding in patients with metastatic cardiac disease is pericardial effusion because of the frequency of pericardial seeding.6 Cardiac metastases are discovered principally in patients with widespread systemic tumour dissemination. The heart may be relatively spared from tumour deposition by vigorous cardiac contractility and rapid coronary blood flow.2 The frequency of thyroid metastasis to the heart was very low (0–2%) in two large autopsy series and fewer than 20 cases have been published in the literature over a 40-year period, usually describing poorly differentiated tumours. Giuffrida and co-workers7–9 published the first review article to describe cardiac metastases from thyroid neoplasia and included 10 cases. Thyroid tumours usually have a good prognosis. Nevertheless, as opposed to differentiated thyroid tumours (papillary and follicular), anaplastic thyroid tumours have a dismal outcome. 327 Echocardiography represents the first-line imaging modality and allows dynamic evaluation of intracardiac masses, but it is often difficult to differentiate thrombi from tumours, and benign from malignant tumours. Such a distinction between these etiologies carries major therapeutic and prognostic implications. Echocardiographic characteristics suggesting a malignant lesion include rightsided tumours, extension to the heart through the venae cavae or pulmonary veins, and intramural mass, commonly with associated intracavitary or pericardial extension. Although a benign mass is unlikely to display these features, there are no pathognomonic findings of benign or malignant tumours in two-dimensional echo.10 Commercially-available echo contrast agents consist of microbubbles filled with high-molecular-weight gases and comparable in size with red blood cells. When injected intravenously, they first appear in the right cardiac chamber, then cross the pulmonary circulation and reach the left heart, opacifying the left cardiac chambers. Moreover, the microbubbles enter into the coronary circulation and opacify the myocardium, enabling the assessment of myocardial perfusion.11,12 Contrast echo perfusion imaging has been utilized to characterize the vascularity of cardiac masses and discriminate malignant/highly vascular tumours from benign tumours and thrombi.10 Kirkpatrick and co-workers3,13 demonstrated that malignant and highly vascular tumours hyper-enhance with contrast echo perfusion imaging, while stromal tumours and thrombi hypo-enhance when compared with the adjacent myocardium. Malignant or highly vascular tumours showed higher video intensity than the myocardium with contrast injection, whereas myxomas (less vascular) demonstrated less perfusion than the myocardium, and thrombi (mainly avascular) revealed no perfusion. To the best of our knowledge, this is the first case report in the literature assessing the vascularity of an intracardiac end-stage papillary thyroid carcinoma metastasis using contrast echo perfusion imaging. Based on the clinical scenario, the patient discussed in this report appeared to have an intracardiac metastasis, since he was known to have metastatic end-stage papillary thyroid carcinoma. Moreover, the echo features were in favour of a malignant process: right ventricular location, and akinesia of adjacent myocardium suggesting right ventricular wall infiltration. An alternate diagnosis, although less likely, was a thrombus developing in an akinetic right ventricular apex. This second hypothesis was flawed by the atypical nature of primary contractile abnormalities limited to the right ventricular apex and the fact that thrombi described in the right chambers are usually in transit. The contrast echo perfusion imaging study demonstrating a highly vascularized mass definitely excluded the possibility of a thrombus and was clearly in favour of a malignant process. Conclusions Two-dimensional echocardiography is a non-invasive, inexpensive, and widely available imaging modality commonly utilized to evaluate cardiac masses in order to narrow the differential diagnosis and establish the most likely etiology. Further characterization can be obtained by assessing the vascularity of intracardiac masses using contrast echo perfusion imaging, providing additional information to differentiate malignant lesions from benign tumours and thrombi. 328 Supplementary material Supplementary material associated with this article can be found in the online version. Conflict of interest: none declared. References 1. Burke A, Virmani R. Atlas of tumor pathology. In: Tumors of the Heart and Great Vessels. Third series, fascicle 16. Washington, DC: Armed Forces Institute of Pathology; 1996. p231. 2. Peters PJ, Reinhardt S. The echocardiographic evaluation of intracardiac masses: a review. J Am Soc Echocardiogr 2006;19:230–40. 3. Ragland MM, Tak T. The role of echocardiography in diagnosing spaceoccupying lesions of the heart. Clin Med Res 2006;4:22–32. 4. Roberts WC. Primary and secondary neoplasms of the heart. Am J Cardiol 1997;80:671–82. S.E. Moustafa et al. 5. Gibbs P, Cebon JS, Calafiore P, Robinson WA. Cardiac metastases from malignant melanoma. Cancer 1999;85:78–84. 6. Goldman JH, Foster E. Transesophageal echocardiographic (TEE) evaluation of intracardiac and pericardial masses. Cardiol Clin 2000;18:849–60. 7. Abraham KP, Reddy V, Gattuso P. Neoplasms metastatic to the heart: review of 3314 consecutive autopsies. Am J Cardiovasc Pathol 1990;3:195–8. 8. Klatt EC, Heitz DR. Cardiac metastases. Cancer 1990;65:1456–9. 9. Giuffrida D, Gharib H. Cardiac metastasis from primary anaplastic thyroid carcinoma: report of three cases and a review of the literature. Endocr Relat Cancer 2001;8:71–3. 10. Lepper W, Shivalkar B, Rinkevich D, Belcik T, Wei K. Assessment of the vascularity of a left ventricular mass using myocardial contrast echocardiography. J Am Soc Echocardiogr 2002;15:1419–22. 11. Yelamanchili P, Wanat FE, Knezevic D, Nanda NC, Patel V. Twodimensional transthoracic contrast echocardiographic assessment of metastatic left ventricular tumors. Echocardiography 2006;23: 248–50. 12. Miller AP, Nanda NC. Contrast echocardiography: New agents. Ultrasound Med Biol 2004;30:425–34. 13. Kirkpatrick JN, Wong T, Bednarz JE, Spencer KT, Sugeng L, Ward RP et al. Differential diagnosis of cardiac masses using contrast echocardiographic perfusion imaging. J Am Coll Cardiol 2004;43:1412–9.