* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Ch. 07 Notes ch7notes
Chemical bond wikipedia , lookup
Fluorescence correlation spectroscopy wikipedia , lookup
History of chemistry wikipedia , lookup
Inductively coupled plasma mass spectrometry wikipedia , lookup
Chemical element wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Particle-size distribution wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Elementary particle wikipedia , lookup
Isotopic labeling wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Mass spectrometry wikipedia , lookup
History of molecular theory wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Molecular dynamics wikipedia , lookup
Stoichiometry wikipedia , lookup
Aerosol mass spectrometry wikipedia , lookup
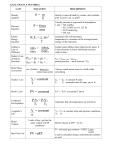
Chapter 7 7.1 The Mole • The SI base unit used to measure the amount of a substance whose number of particles is the same as the number of atoms of carbon in exactly 12 grams of carbon-12. • The mole is a counting unit used to count out a given number of particles. • Avogadro’s number- 6.022 x 1023, the number of atoms or molecules in 1.00 mol. o 6.022 x 1023 particles = 1 mole • Molar Mass – The mass in grams of one mole of a substance. Converting between Moles, Particles and Molar Mass Moles to number of particles: How many ions are in 0.187mol of Na+ ions? 0.187 mol Na+ ions x (6.022 x 1023 ions/ 1 mole) = 1.123 x 1023 ions. Number of Particles to moles: 3.01 x 1023 molecules SO2 x (1 mole / 6.022 x 1023 molecules) = 0.50 mol Particles to mass 2.11 x 1024 atoms Cu x (1mol/6.022 x1023 atoms) x (63.55 grams/1 mole) = 2.23 x 102g Mass to Particles 237 g Cu x (1 mol/63.55 g) x (6.022 x 1023/1 mol) = 2.24 x 1024 atoms 7.2 Average Atomic Masses Most elements are mixtures of isotopes. Average atomic masses represent weighted averages of the atomic masses of all naturally occurring isotopes of an element. Calculating an average atomic mass Isotope Percentage Decimal Fraction Contribution Copper-63 69.17% 0.6917 62.94 amu x 0.6917 Copper-65 30.83% 0.3083 64.93 amu x 0.3083 The average atomic mass is the sum of the contributions. (62.94amu x 0.6917) + (64.93amu x 0.3083) = 63.55amu Chemical Formulas and Moles • Chemical formulas give a ratio of elemental components. • Ionic formulas show the simplest ratio of cations and anions. • Covalent formulas (including polyatomic ions) show both elements and the number of each element. Formulas can be used to calculate Molar Masses • From formulas we can tell what elements (or ions) are present and in what quantities. • Molar masses of individual elements (found on the periodic table) are summed to determine molar masses of molecular compounds. Calculating the molar mass of a compound • 1 mol CaCl2 = 1mol Ca + 2 mol Cl • 1mol Ca = 1 x 40.078g/mol = 40.078g • 2mol Cl = 2 x 35.4527g/mol = 70.9054g • 1mol CaCl2 = 40.078g + 70.9054g = 110.983g 7.3 Percentage composition – The percentage by mass of each element in a compound. • Percent composition can verify a substance’s identity. • Percent Composition can be calculated from any chemical formula. o Find the total mass of each elemental component. o Divide the element’s total mass by the compound's molar mass. o Multiply by 100. Example: CO2 Molar mass = 44.01 g/mol Carbon 1 x 12.01 = 12.01 x 100 = 27.3% 44.01 Oxygen 2 x 16.00 = 32.00 x 100 = 72.7% 44.01 Total = 100.0% Empirical Formula - These chemical formulas show the composition of a compound in its simplest ratio. Formaldehyde = CH2O Acetic Acid = C 2 H4 O2 Glucose = C6H12O6 Each has the same empirical formula = CH2O Elemental analysis can provide this ratio. How? It’s all about the number of moles! Example: Expirimental analysis yields the following… Pb - 74.51% Cl - 24.49% 1. Convert to grams assuming 100g sample Pb = 74.51% x 100g = 74.51g, Cl = 24.49% x 100g = 24.49g 2. Convert to moles Pb 74.51g x (1mol/207.2) = 0.36 mol Cl 24.49g x (1mol/35.453) = 0.69 mol 3. Convert to whole numbers by dividing each subscript by the smallest subscript. 0.36/0.36 = 1, 0.69/0.36= 1.9 (round up) Emperical Formula = PbCl2 Molecular Formula While formulas of ionic compounds already show the simplest whole # ratios. Molecular formulas are multiples of empirical formulas. • Experimental analysis can provide Molar Mass. • By dividing the experimental molar mass by the empirical molar mass you can determine the multiple for the molecular formula. Example: Empirical Formula CH2O Empirical Molar Mass 30.03g/mole Experimental Molar Mass 180.18g/mole 30.03g/180.18g = 6 Therefore Molecular Formula C6H12O6