* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Ethers
Survey
Document related concepts
Transcript
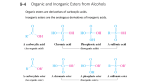
Hydroxy derivatives of hydrocarbons (alcohols,
phenols, ethers) and sulfur analogues:
Bonding system characterization. Physical properties. Acidbase properties, the structural determinants of acidity. Theirs
reactions connected with their nucleophilic properties
(alkylation, acylation, sulphonic acid, producing inorganic
esters), alcohols, acid-catalyzed conversion. Oxidation of
alcohols and phenols. Ethers properties, cleaved of ethers.
Special bonding systems of ethers (epoxides and hemiacetals,
acetals and enol ethers) and their chemical reactions. Their
synthesis.
Grouping of compounds with C-OH, C-O-C, C-SH and C-S-C bonds
Starting point: classical valence theory:
Formally - a water/a hydrogen sulphide is
substituted with hydrocarbon radicals
BUT! for sulphur empty d orbitals, S = O bond is
established ("four and six valence" sulphur!)
1-C-OH bonded compounds (alcohols, phenols)
according to the hybrid status of the pillar carbon:
alcohols (R-OH): sp3 carbon
phenols, enols (Ar-OH, C = C-OH): sp2 carbon
- not arbitrary, different bonding!
ethanol
phenol
vinyl alcohol
according to the order of sp3 carbon atoms
(alcohols): primary (1°), secondary (2°), tertiary (3°)
similarity to the halogen derivatives
according to the number of hydroxyl groups (diol, triol,
polyol) in case of diols:
according to their position to each other: geminal, vicinal,
disjunct
according to the nature of the hydrocarbon group:
saturated / unsaturated / acyclic / cyclic
n1
Nomenclature
Functional class nomenclature
Functional class names of alcohols are derived by naming the alkyl group that bears the hydroxyl
substituent (±OH) and then adding alcohol as a separate word. The chain is always numbered beginning
at the carbon to which the hydroxyl group is attached.
Substitutive nomenclature
Substitutive names of alcohols are developed by identifying the longest continuous chain that bears the
hydroxyl group and replacing the -e ending of the 5-Chloro-2-methylheptane corresponding alkane by
the suffix -ol. The position of the hydroxyl group is indicated by number, choosing the sequence that
assigns the lower locant to the carbon that bears the hydroxyl group.
Hydroxyl groups take precedence over (“outrank”) alkyl groups and halogen substituents in
determining the direction in which a carbon chain is numbered.
Trivial names (common names)
Several alcohols are commonplace substances, well known by common names that reflect their origin
(wood alcohol, grain alcohol) or use (rubbing alcohol).
Wood alcohol is methanol (methyl alcohol, CH3OH), grain alcohol is ethanol (ethyl alcohol, CH3CH2OH),
and rubbing alcohol is 2-propanol [isopropyl alcohol, (CH3)2CHOH].
Glycerol (glycerin, propane-1,2,3-triol), glycol (ethylene glycol, ethane-1,2-diol).
Classification of C-OH, C-O-C, C-SH and C-S-C compounds 2.
2. C-O-C compounds (ethers)
According to the linked groups: symmetrical and non-symmetrical (mixed) ethers
According to the hydrocarbon group
- Aliphatic ethers (R-O-R, R-O-R1)
- Aliphatic-aromatic ethers (R-O-Ar)
- Aromatic ethers (Ar-O-Ar1 + heteroaromatic analogues)
- Specific, other ethers different (higher) reactivity enol ethers [-C = C-OR (Ar)]
Special types of ether
Ether derivatives of geminal diols
Cyclic ethers
epoxides (oxiranes)
special compounds:
different reactivity than
cyclic ether
hemiacetal
acetal
Similarity to aldehydes and
ketones
orthoester
Relation to carboxylic acids
3. C-O-O-H, C-O-O-R compounds
Formally, the alkylated / arylated derivatives of hydrogen peroxide
Nomenclature of ethers
Substitutive IUPAC nomenclature
Ethers are named, in substitutive IUPAC nomenclature, as alkoxy derivatives of alkanes.
ONLY alkoxy / aryloxy prefix + base carbon chain (+ local number multiplier members)
Functional class IUPAC nomenclature
Functional class IUPAC names of ethers are derived by listing the two alkyl groups in the general
structure ROR in alphabetical order as separate words, and then adding the word “ether” at the end.
When both alkyl groups are the same, the prefix di- precedes the name of the alkyl group.
Ethers are described as symmetrical or unsymmetrical depending on whether the two groups
bonded to oxygen are the same or different. Unsymmetrical ethers are also called mixed ethers.
Diethyl ether is a symmetrical ether; ethyl methyl ether is an unsymmetrical ether.
Nomenclature of ethers 2.
Cyclic ethers have their oxygen as part of a ring—they are heterocyclic compounds
Several have specific IUPAC names.
trivial names, additive nomenclature, Hantzsch-Widman nomenclature
Hantzsch-Widman nomenclature:
(additive nomenclature)
(trivial names)
(trivial names)
In each case the ring is numbered starting at the oxygen. The IUPAC rules also permit oxirane (without
substituents) to be called ethylene oxide. Tetrahydrofuran and tetrahydropyran are acceptable
synonyms for oxolane and oxane, respectively.
Many substances have more than one ether linkage. Two such compounds, often used as solvents,
are the diethers 1,2-dimethoxyethane and 1,4-dioxane. Diglyme, also a commonly used solvent, is a
triether.
4. Compounds with C-S-H, C-S-R bonds (sulfur analogs of alcohols , phenols and ethers)
In close analogy to the oxygen-containing analogues - thioalcohols, thiophenols, thioethers
thioalcohol
thiophenol
sulfide (thioether)
R1=R2 or R1≠R2
disulfide
if n>2 polysulfide
different from oxygen
Derivatives with more than two ligands
sulfinyl group sulfonyl group
Relationship with
sulfones according to the
binding system,
chemically similar to
carboxylic acids
Nomenclature of thiols
Substitutive IUPAC names
Thiols are given substitutive IUPAC names by appending the suffix -thiol to the name of the
corresponding alkane, numbering the chain in the direction that gives the lower locant to
the carbon that bears the ―SH group. The final -e of the alkane name is retained. When the
―SH group is named as a substituent, it is called a mercapto group. It is also often referred
to as a sulfhydryl group, but this is a generic term, not used in systematic nomenclature.
At one time thiols were named mercaptans. Thus, CH3CH2SH was called “ethyl mercaptan”
according to this system. This nomenclature was abandoned beginning with the 1965
revision of the IUPAC rules but is still sometimes encountered, especially in the older
literature.
aromatics: thiophenol-based nomenclature
Nomenclature of sulfides
Substitutive nomenclature of sulfides
The sulfur analogs (RS―) of alkoxy groups are called alkylthio groups. The first two of the
following examples illustrate the use of alkylthio prefixes in substitutive nomenclature of
sulfides.
Prefixes: alkylthio/alkylsulfanyl, arylthio/arylsulfanyl, alkylpolythio
Functional class IUPAC names of sulfides
Functional class IUPAC names of sulfides are derived in exactly the same way as those of
ethers but end in the word “sulfide.”
hydrocarbon group names + sulfide, disulfide, polysulfide suffix
Sulfur heterocycles have names analogous to their oxygen relatives, except that ox- is
replaced by thi-. Thus the sulfur heterocycles containing three-, four-, five-, and sixmembered rings are
named thiirane, thietane, thiolane, and thiane, respectively.
Substitutive nomenclature:
Functional class IUPAC names:
Nomenclature of sulfoxides and sulfones
Substitutive nomenclature
prefix: alkylsulfinyl, arylsulfinyl, alkylsulfonyl, arylsulfonyl
Functional class nomenclature
hydrocarbon group names + sulfoxide / sulfone suffix
Additionally: Compound Name + S-oxide / S, S-dioxide suffix
Substitutive nomenclature:
Functional class nomenclature:
Dimethyl sulfoxide
Methylsulfinylmethane
Dimethyl sulfon
Methylsulfonylmethane
Dimethyl sulfide S-oxide Dimethyl sulfide S,S-dioxide
Bonding system of alcohols and ethers
Starting point: structure of water - sp3 hybrid state for oxygen (h12h22h31h41)
Alcohols – C(sp3)-O(sp3) hetero nuclear -bond
Ethers – two C(sp3)-O(sp3) hetero nuclear -bond
tetrahedral compound but the bond angle is deformed
(R,R1 groups have more space demand)
Bond E – both C-O and O-H are strong
C-O: 355-380 kJ/mol (compare to: C-C: 345-355 kJ/mol)
O-H: 460-465 kJ/mol (compare to: C-H: 400-415 kJ/mol)
Bond distance
Phenols, phenol ethers, enol ethers: shortening bond distance more stronger bond!!
(greater bond order)
Reason: interaction between nonbonding e-pair and -e-system (+M effect)
Resonance structures:
(sp2 hybrid state
for oxygen)
Seven-center bond with eight
electrons - electron delocalization!
(parallel PZ orbitals)
C-O bond: the increasing double bond character
Aromatic ring increased electron density ( OH, OR
first order directing groups, activating substituents!)
Electron negativities
ENC = 2.5, ENO = 3.5, ENH = 2.1
polar hetero nuclear bonds, charge separation
permanent dipole moment
Tioalcohols and tioethers
Formal similarity between O and S
BUT in case of sulphur:
3s23p43do
(electon cofiguration of oxygen: 1s22s22p4)
e. g.
or
Thiophene (aromatic compound)
Further differences: S has larger atom radius (rS = 0.102 nm, rC = 0.077 nm, ro = 0.073 nm)
longer and weaker bonds compare to oxygen
Non-bonding e-pairs have greater space demand
larger deformation compare to oxygen analogues
Physical properties of alcohols, ethers and their thio analogous
Boiling point, melting point – Typically, higher than alkanes, and alkyl halides, it has
homologous series
Increasing length of carbon chain makes it closer to R-Cl, RH characteristics (dispersion forces
between the alkyl chains become increasingly dominant)
Boiling
points (oC)
Forces
R-OH
R-SH ~ R-OR1 ~ R-Cl
R-H
H bond
dipole-dipole
Induced dipole - Induced dipole
H-OH > Me-OH, Et-OH, Pr-OH
H-OH > H-SH, R-OH > R-SH
worse fit, weakening association
weakening H-bond (S nonbonding pair is diffuse)
Melting points of n-alcohols (ROH)
Melting point: minimum curve – longer alkyl group
incorporation into the diamond-like H-bond structure of the ice
is not occurs completely so
If the alkyl chain is long than „alkane-like” mp can be expected
Hydrogen bonding
A dipole–dipole attraction between the positively polarized proton of the OH group of one ethanol
molecule and the negatively polarized oxygen of another. The term hydrogen bonding is used to
describe dipole–dipole attractive forces of this type. The proton involved must be bonded to an
electronegative element, usually oxygen or nitrogen.
Protons in C―H bonds do not participate in hydrogen bonding.
Hydrogen bonding in ethanol
involves the oxygen of one molecule
and the proton of an ―OH group of
another. Hydrogen bonding is much
stronger than most other types of
dipole–dipole attractive forces.
Di- and polyols: highly elevated mp, bp
Reason: intermolecular H-bonds, long chains
Density: 1 (H2O) – alkyl groups makes the molecule „lighter”
Solubility:
In Water: H bonds, in low concentrations the solubility is good
(n = 1-3: unlimited!)
Ethers: worse solubility in water, BUT S(Et2O) = 8 g/100 ml!!
„one-sided” H bonds, ether only H acceptor!
Thiols: weak H bonds weak solubility in water
Preparation of alcohols
1. From alkyl halides by SN reaction
Problem: competing elimination
(alkene formation) →
contaminated product
probability R = 1° <2° <3°
direction is increasing
2. Hydrolysis of esters
The acidic variant an equilibrium reaction,
reversal of the esterification.
Better: alkaline hydrolysis (≥ 1 equiv base.)
Typical: NaOH (KOH)/H2O or NaOH/alcohol, dioxane etc – H2O (solubility!), then H3O
A two-step pathway for avoiding elimination
SN reaction with a less nucleophile partner,
easy ester cleavage
Preparation of alcohols
3. Hydration of alkenes (formal or actual water addition)
3.1. Acid-catalyzed addition of water
The structure of the major product (regioselectivity)
is defined by the Markovnikov’s rule.
dilute
3.2. Oxymercuration – demercuration
The structure of the major product (regioselectivity)
is defined by the Markovnikov’s rule.
3.3. Hydroboration
The structure of the major product
(regioselectivity) is defined by the Markovnikov’s
rule BUT anti-Markovnikov product is formed.
4. Reduction of oxo compounds
Opportunities:
1. catalytic reduction (H2/cat., cat. = Pd-C, Pt, PtO2, Raney-Ni, etc.)
2. Metal hydrides (NaBH4/R-OH, LiAlH4/Et2O or THF --- H-)
3. Dissolving metal reduction (Zn/HCl or NaOH, Na/EtOH, etc. --- formation of H2)
Preparation of alcohols
5. Reduction of esters
In laboratory: LiAlH4 (LAH)/Et2O;
Industry: catalytic reduction (harsh
conditions, eg. copper-chromite
(Cu2Cr2O5)/150-400 oC, 100-300 bar)
6. Reactions of oxo comp. / carboxylic acid derivatives and Grignard reagenst
Good news: you already know these reactions
Preparation of phenols
In laboratory: „cooking” of diazonium salts
By-products
Diazonium salt
Phenol
Industry: nowadays starting from cumene
The world phenol production:
8.9 million tonnes in 2012.
The global phenol foreign trade
exceeded USD 3.6 billion in 2012.
The world phenol supply is expected
to go beyond the 10.7 million tonnes
mark in 2016.
Preparation of thiols
Nucleophilic substitution.
Disadvantage: symmetrical ether formation, cause the resultant product is also
reactive in nucleophilic reactions so a possible secondary reaction can take place
Preparation of thioalcohols 2.
A better substituion reaction:
Synthesis of thiols through isothiouronium salt
The most often
used method
Preparation of thioethers
In a nucleophile substitution reaction
Analogy with Williamson’s ether synthesis
Thiols and thiophenols reacts readily,
Reason: the great nucleophilicity of S (+ easy formation of thiolate, thiophenolate)