* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download phys chapter 9 [12-11
Management of acute coronary syndrome wikipedia , lookup
Heart failure wikipedia , lookup
Coronary artery disease wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Electrocardiography wikipedia , lookup
Antihypertensive drug wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Cardiac surgery wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Myocardial infarction wikipedia , lookup
Jatene procedure wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Heart arrhythmia wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
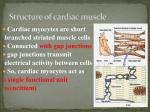
Phys Ch 9 Cardiac Muscle Heart is composed of 3 major types of cardiac muscle o Atrial muscle o Ventricular muscle o Excitatory (conductive) muscle fibers Atrial and ventricular muscles contract in the same way as skeletal muscle, but the duration of contraction is much longer Specialized excitatory and conductive fibers contract only feebly because they contain few contractile fibrils and exhibit either automatic rhythmical electrical discharge in the form of action potentials or conduction of action potentials through the heart, providing an excitatory system that controls rhythmical beating of heart Cardiac muscle has typical myofibrils that contain actin and myosin filaments almost identical to those found in skeletal muscle o These filaments slide along one another during contraction in the same manner as occurs in skeletal muscle Intercalated discs – cell membranes that separate individual cardiac muscle cells from one another – at each disc, cell membranes fuse with one another in gap junctions that allow rapid diffusion of ions o Ions move with ease in intracellular fluid along longitudinal axis of cardiac muscle fibers o This makes cardiac muscle a syncytium of many heart muscle cells in which the cardiac cells are so interconnected that when one of them becomes excited, the action potential spreads to all of them, from cell to cell throughout the latticework interconnections Heart is composed of atrial syncytium (walls of the 2 atria) and the ventricular syncytium (walls of 2 ventricles) o Atria separated from ventricles by fibrous tissue that surrounds AV valvular openings between atria and ventricles o Normally potentials are not conducted from atrial syncytium into ventricular syncytium o AP’s conducted by AV bundle o This allows atria to contract a short time before ventricular contraction Action Potentials in Cardiac Muscle Average AP is 105 mV (intracellular potential rises from -85 mV to +20 mV during each beat) After initial spike, membrane remains depolarized for about 0.2 seconds, exhibiting a plateau followed at the end of the plateau by abrupt repolarization o Presence of plateau causes ventricular contraction to last as much as 15 times as long as a skeletal muscle contraction Action potential of skeletal muscle is caused almost entirely by sudden opening of large numbers of fast sodium channels that allow tremendous amounts of sodium ions to enter skeletal muscle fiber from extracellular fluid, however cardiac muscle is caused by these AND slow calcium channels (calcium-sodium channels) o Calcium channels are slower to open and remain open for several tenths of a second (as opposed to the few thousandths of a second the Na channels are open for) o During the time the channel is open, large quantities of both Ca and Na ions flow through the channels to the interior of the cardiac muscle fiber, and this maintains the prolonged period of depolarization, causing the plateau o Calcium ions that enter in this phase also help activate muscle contractile process (in opposition to skeletal muscle cells that rely solely on Ca from sarcoplasmic reticulum) Immediately after onset of action potential, permeability of cardiac muscle membrane for potassium ions decreases about fivefold (does not occur in skeletal muscle) – results from excess Ca influx through Ca channels – greatly decreases outflux of positively charged K ions during AP plateau and prevents early return of AP voltage to resting potential When channels finally close, membrane permeability to K ions also increases rapidly, immediately returning the membrane potential to resting level Velocity of conduction of excitatory action potential signal along both atrial and ventricular muscle fibers is 0.30.5 m/s (1/250 the velocity in very large nerve fibers and 1/10 the velocity in skeletal muscle fibers) Velocity of conduction in Purkinje fibers is about 4 m/s in most parts of the system Does undergo a short refractory period (0.25-0.30 sec in the ventricles) – additional relative refractory period of 0.05 sec during which the muscle is more difficult than normal to excite but can be excited by a very strong excitatory signal o Refractory period of atrial muscle much shorter than that for ventricles (0.15 sec) Excitation-contraction coupling – mechanism by which AP causes myofibrils of muscle to contract o When AP passes over cardiac muscle membrane, AP spreads to interior of cardiac muscle fiber along membranes of T tubules o T tubule AP’s act on membranes of longitudinal sarcoplasmic tubules to cause release of Ca ions into muscle sarcoplasm from sarcoplasmic reticulum o These Ca ions diffuse into the myofibrils and catalyze the chemical reactions that promote sliding of actin and myosin filaments along one another, producing muscle contraction o In addition to Ca ions that are released into sarcoplasm from cisternae of sarcoplasmic reticulum, Ca ions also diffuse into sarcoplasm from T tubules themselves at time of AP, which opens voltagedependent Ca channels in the membrane of the T tubule o Ca entering the cell then activates calcium release channels (ryanodine receptor channels) in sarcoplasmic reticulum membrane, triggering release of Ca into sarcoplasm o Ca ions in sarcoplasm then interact with troponin to initiate cross-bridge formation and contraction by same basic mechanism as skeletal muscles o Without calcium from T tubules, strength of cardiac muscle contraction would be reduced considerably because sarcoplasmic reticulum of cardiac muscle is less well developed than that of skeletal muscle and does not store enough Ca to provide full contraction o T tubules of cardiac muscle have a diameter 5 times that of skeletal muscle tubules (25x the volume) o Inside T tubules is large quantity of mucopolysaccharides that are electronegatively charged and bind an abundant store of Ca ions, keeping these always available for diffusion to interior of cardiac muscle fiber when T tubule AP appears Strength of contraction depends on concentration of Ca ions in extracellular fluids – openings of T tubules pass directly through cardiac muscle cell membrane into extracellular spaces surrounding cells, allowing same extracellular fluid in cardiac muscle interstitium to percolate through T tubules as well At the end of the AP, Ca transport back into sarcoplasmic reticulum achieved with help of calcium-ATPase pump o Ca ions also removed from cell by Na-Ca exchanger o Na that enters cell during exchange then transported out of cell by Na-K ATPase pump Cardiac muscle begins to contract a few milliseconds after AP begins and continues until a few milliseconds after AP ends, so duration of contraction of cardiac muscle is mainly a function of duration of AP, including the plateau Cardiac Cycle Everything that happens from beginning of one heartbeat to the beginning of the next Initiated by spontaneous generation of AP in sinus node o AP travels from here rapidly through both atria and then through AV bundle into ventricles o There is a delay of more than 0.1 sec during passage of cardiac impulse from atria to ventricles, which allows atria to contract ahead of ventricular contraction Duration of cardiac cycle is the reciprocal of the heart rate When heart rate increases, the duration of each cardiac cycle decreases, including contraction and relaxation phases o Duration of AP and systole decrease, but not by as great a percentage as does diastole o Thus at very fast heart rate, heart does not remain relaxed long enough to allow complete filling of the cardiac chambers before next contraction Electrocardiogram o P wave caused by spread of depolarization through atria and is followed by atrial contraction, which causes a slight rise in atrial pressure o QRS wave (0.16 sec after P wave) is result of electrical depolarization of ventricles, which initiates contraction of ventricles and causes ventricular pressure to begin rising just before ventricular systole o Ventricular T wave – represents stage of repolarization of ventricles when ventricular muscle fibers begin to relax, so T wave occurs slightly before end of ventricular contraction Blood normally flows from great veins into atria, and 80% of blood flows directly through atria into ventricles before atria contract – atrial contraction usually causes an additional 20% filling of ventricles, so atria are only primer pumps that increase ventricular pumping effectiveness by 20% Heart usually has the capability to pump 300-400% more blood than is required by resting body, so person with atrial pumping trouble wouldn’t even notice unless they try to exercise Atrial pressure curve has a, c, and v pressure waves o A wave caused by atrial contraction – left increases more than right (7-8 mm Hg instead of 4-6 mm Hg) o C wave occurs when ventricles begin to contract, caused partly by slight backflow of blood into atria at onset of ventricular contraction but mainly by bulging of AV valves backward toward atria because of increasing pressure in ventricles o V wave occurs toward end of ventricular contraction and results from slow flow of blood into atria from veins while AV valves closed during ventricular contraction When ventricular contraction is over, AV valves open, allowing stored atrial blood to flow rapidly into ventricles, causing v wave to disappear Period of rapid filling of ventricles – after ventricles relax and sudden release of pressure on AV valves makes them open and allow blood to rapidly flow into ventricles o Lasts for about the first 1/3 of diastole o During middle 1/3 of diastole, only small amount of blood normally flows into ventricles – this is blood that comes in from veins and goes straight through atria into ventricles o During last 1/3 of diastole, atria contract and give additional thrust to inflow of blood into ventricles, accounting for about 20% of the filling of the ventricles during each heart cycle Period of isometric (isovolumic) contraction – immediately after ventricular contraction begins, ventricular pressure rises abruptly, causing AV valves to close o Additional 0.02-0.03 sec required for ventricle to build up sufficient pressure to push semilunar valves open against pressures in aorta and pulmonary artery o During this period, contraction is occurring in the ventricles, but there is no emptying Period of ejection – when left ventricular pressure rises slightly above 80 mm Hg and right ventricular pressure is slightly above 8 mm Hg, ventricular pressures push semilunar valves open – 70% of blood empties in first 1/3 of this period, with remaining 30% emptying during next 2/3 o First 1/3 of this period called period of rapid ejection and rest of this period called period of slow ejection Period of isometric (isovolumic) relaxation – at end of systole, ventricular relaxation begins suddenly, allowing both right and left intraventricular pressures to decrease rapidly o Elevated pressures in arteries that have just been filled with blood push blood back toward ventricles, which snaps aortic and pulmonary valves closed o For another 0.03-0.06 sec, ventricular muscle continues to relax, even though ventricular volume does not change o Intraventricular pressures decrease rapidly back to low diastolic levels During diastole, normal filling of ventricles increases volume of each ventricle to 110-120 mL (end-diastolic volume) o As ventricles empty during systole, volume decreases about 70 mL (stroke volume output) o o o Remaining volume in each ventricle (40-50 mL) is called end-systolic volume Fraction of end-diastolic volume that is ejected called ejection fraction (about 60%) When heart contracts strongly, end-systolic volume can be decreased to as little as 10-20 mL – when large amounts of blood flow into ventricles during diastole, ventricular end-diastolic volumes can become as great as 150-180 mL in a healthy heart By increasing end-diastolic volume and decreasing end-systolic volume, stroke volume output can be increased to more than double normal Aortic Pressure Curve When left ventricle contracts, ventricular pressure increases rapidly until aortic valve opens – after this, ventricle pressure rises much less rapidly because blood immediately flows out of ventricle into aorta and then into systemic distribution arteries Entry of blood into arteries causes walls of arteries to stretch At end of systole, after left ventricle stops ejecting blood and aortic valve closes, the elastic walls of the arteries maintain high pressure in the arteries, even during diastole Incisura occurs in aortic pressure curve when aortic valve closes – caused by short period of backward flow of blood immediately before closure of the valve, followed by sudden cessation of backflow After aortic valve has closed, the pressure in the aorta decreases slowly throughout diastole because blood stored in distended elastic arteries flows continually through peripheral vessels back to the veins Pressure curves in right ventricle and pulmonary artery similar to those in aorta, except that the pressures there are about 1/6 of that in aorta Work Output of Heart Stroke work output of heart – amount of energy that heart converts to work during each heartbeat Minute work output – total amount of energy converted to work in 1 minute Work output of heart is in 2 forms o Major proportion used to move blood from low-pressure veins to high-pressure arteries (volumepressure work or external work) o Minor proportion used to accelerate blood to its velocity of ejection through aortic and pulmonary valves (kinetic energy of blood flow) Right ventricular external work output is normally 1/6 of the left ventricle because of sixfold difference in systolic pressures that they pump against Additional work output of each ventricle required to create kinetic energy of blood flow is proportional to mass of blood ejected times square of velocity of ejection Ordinarily work output of left ventricle required to create kinetic energy of blood flow is about 1% of total work output of the ventricle and is ignored in calculation of total stroke work output Reason for diastolic pressure’s sudden rise around 150 mL is that above this, the fibrous tissue in the heart will stretch no more and because the pericardium that surrounds the heart becomes filled nearly to its limit Reason for decline in systolic pressure in above is because about 150 mL, actin and myosin filaments of cardiac muscle fibers are pulled apart far enough that strength of each cardiac fiber contraction becomes less than optimal Concepts of Preload and Afterload Preload – degree of tension on muscle when it begins to contract (end-diastolic pressure) Afterload – load against which muscle exerts its contractile force (pressure in the aorta leading from the ventricle) - shaded area represents net external work output of ventricle during contraction cycle – a graph like this is formed during studies to calculate cardiac work output Preload and Afterload Preload – degree of tension on muscle when it begins to contract Afterload – load against which the muscle exerts its contractile force Preload usually considered to be end-diastolic pressure when ventricle has become filled Afterload of ventricle is pressure in aorta leading from ventricle In many abnormal functional states of the heart or circulation, the pressure during filling of the ventricle (preload), the arterial pressure against which the ventricle must contract (afterload), or both are severely altered from normal Chemical Energy Required for Cardiac Contraction Heart muscle uses chemical energy to provide work of contraction About 70-90% of this energy normally derived from oxidative metabolism of fatty acids with 10-30% coming from other nutrients, especially lactate and glucose Rate of oxygen consumption by heart is excellent measure of chemical energy liberated while heart performs its work Experimental studies have shown that oxygen consumption of heart and chemical energy expended during contraction directly related to external work plus potential energy Oxygen consumption nearly proportional to tension that occurs in heart muscle during contraction multiplied by duration of time that contraction persists (tension-time index) More chemical energy is expended even at normal systolic pressures when ventricle is abnormally dilated because heart muscle tension during contraction is proportional to pressure times diameter of ventricle Much of expended chemical energy converted to heat, and much smaller portion to work output – ratio of these is efficiency of cardiac contraction (also called efficiency of the heart) o Maximum efficiency of heart is between 20-25% and in heart failure drops as low as 5-10% Regulation of Heart Pumping When a person is at rest, the heart pumps 4-6 L of blood each minute During severe exercise, heart may pump 4-7x normal amount Under most conditions, amount of blood pumped by heart each minute is normally determined almost entirely by venous return Frank-Starling mechanism of the heart – intrinsic ability of heart to adapt to increasing volumes of inflowing blood by this concept – the greater the heart muscle is stretched during filling, the greater the force of contraction and the greater the quantity of blood pumped into the aorta o Within physiologic limits, the heart pumps all the blood that returns to it by the way of the veins When an extra amount of blood flows into the ventricles, the cardiac muscle itself is stretched to greater length causes muscle to contract with increased force because actin and myosin filaments are brought to a more nearly optimal degree of overlap for force generation ventricle, because of increased pumping, automatically pumps extra blood into the arteries Stretch of right atrial wall directly increases heart rate by 10-20%, which helps increase amount of blood pumped each minute, although its contribution is much less than that of Frank-Starling mechanism External Forces That Can Affect Heartrate Pumping effectiveness of heart controlled by vagus (sympathetic and parasympathetic) nerves Strong sympathetic stimulation can increase heart rate of prime adults from 70 bpm to 180-200 bpm and, under extreme circumstances, 250 bpm o Sympathetic stimulation increases force of heart contraction to as much as double normal, increasing volume of blood pumped and increasing ejection pressure o So sympathetic stimulation can increase max cardiac output as much as 2-3 fold Under normal conditions, sympathetic nerve fibers to heart discharge continuously at a slow rate that maintains pumping at about 30% above that with no sympathetic stimulation o When activity of sympathetic nervous system is depressed below normal, this decreases both heart rate and strength of ventricular muscle contraction, decreasing level of cardiac pumping as much as 30% below normal Strong stimulation of parasympathetic nerve fibers in vagus nerves to heart can stop heartbeat for a few seconds, but then heart “escapes” and beats 20-40 bpm as long as parasympathetic stimulation continues o Strong vagal stimulation can decrease strength of heart muscle contraction by 20-30% o Vagal fibers distributed mainly to atria and not much to ventricles, explaining why vagus stimulation mostly decreases heart rate rather than strength of heart contractions Excess potassium in extracellular fluids causes heart to become dilated and flaccid and slows heart rate – large quantities can block conduction of cardiac impulse from atria to ventricles through AV bundle o 2-3x normal potassium concentration can cause such weakness of the heart and abnormal rhythm that death occurs o High potassium concentration in extracellular fluids decreases resting membrane potential (partially depolarizes cell membrane) – as membrane potential decreases, intensity of AP decreases, making contraction of heart progressively weaker Excess of calcium ions causes heart to go toward spastic contraction because of direct effect of Ca ions to initiate cardiac contractile process o Deficiency of Ca ions causes cardiac flaccidity o Calcium ion levels in blood are normally regulated to very narrow range Increased body temperature causes greatly increased heart rate and decreased body temperatures decrease heart rate o These result from the fact that heat increases permeability of cardiac muscle membrane to ions that control heart rate Contractile strength of heart often enhanced temporarily by moderate increase in temperature, as occurs during body exercise, but prolonged elevation of temperature exhausts metabolic systems of the heart and eventually causes weakness Increasing arterial pressure in aorta does not decrease cardiac output until mean arterial pressure rises above 160 mm Hg