* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Biogeochemical Cycles
Biosequestration wikipedia , lookup
Biochemistry wikipedia , lookup
Metalloprotein wikipedia , lookup
Photosynthesis wikipedia , lookup
Microbial metabolism wikipedia , lookup
Sulfur cycle wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Nitrogen dioxide poisoning wikipedia , lookup
Citric acid cycle wikipedia , lookup
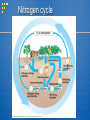
Biogeochemical Cycles Water Nitrogen Carbon Dioxide Phosphorus Sulfur Biogeochemical Cycles: chemical elements are required by life from the living and nonliving parts of the environment. These elements cycle in either a gas cycle or a sedimentary cycle In a gas cycle elements move through the atmosphere. Main reservoirs are the atmosphere and the ocean. Sedimentary cycle elements move from land to water to sediment. Carbon Cycle What are the 2 main processes in the carbon cycle? Carbon Cycle Is an atmospheric cycle Carbon (C) enters the biosphere during photosynthesis: CO2 + H2O C6H12O6 + O2 + H2O Carbon is returned to the biosphere in cellular respiration: O2 +H2O + C6H12O6 CO2 +H2O + energy Carbon Cycle In the ground C in organic molecules in organisms is moved to C in fossil fuels over millions of years by pressure, heat and bacterial action. C in limestone (CaCO3) is released slowly to C in CO2 when exposed to O2 or H2O Largest reservoir of C is sedimentary rock Carbon Cycle Second largest reservoir of C is the ocean Dissolved CO2, living things in the ocean In Water: CO2 + H2O HCO3-1 + CO3 2 Ca2+ + CO32- CaCO3 in shells/skeletons of aquatic organisms CaCO3 buried, long periods of time, pressure Limestone Carbon Facts Every year there is a measurable difference in the concentration of atmospheric CO2 with changes in the seasons. For example, in winter there is almost no photosynthesis ( higher CO2 ) During the growing season there is a measurable difference in the concentration of atmospheric CO2 over parts of each day. Carbon Cycle Human Impact: Removal of vegetation reduces absorption of CO2 for photosynthesis from atmosphere. Increases atmospheric CO2 Burning of fossils fuels increases atmospheric CO2 Increase in atmospheric CO2 leads to increased Greenhouse Effect = Global Warming Nitrogen cycle Nitrogen Cycle Nitrogen (N) is an essential constituent of protein, DNA, RNA, and chlorophyll. Is an atmospheric cycle Nitrogen is the most abundant gas in the atmosphere. Plants and animals cannot use free N2 gas in the atmosphere. Nitrogen must be fixed or converted into a usable form. Nitrogen Cycle Free N2 in atmosphere is “fixed” by nitrogen fixing bacteria to NH3 (ammonia): N2 + 3H2 2NH3 Nitrogen fixing bacteria live in nodules on the roots of leguminous plants (soybeans, peas, clover and alfalfa) Water in the soil reacts with NH3 (ammonia) to form NH41+ ammonium ion Nitrogen Cycle Another species of bacteria can perform nitrification once NH41+ has been formed NH41+ NO21- NO31- Assimilation – absorption of NH3, NH41+, NO31- for use by plants to make nucleic acids and proteins. Animals get fixed nitrogen by eating plants or animals Nitrogen Cycle Plants and animals are broken down by still other bacteria that convert nitrogen containing organic molecules in organisms to an inorganic form of nitrogen (NH3 or NH41+) This is ammonification Once this ammonia is formed, still another group of bacteria can perform denitrification: NH3 or NH41+ NO21 NO31- N2 and N2O and/or Nitrogen Cycle Nitrogen is often the limiting factor in plant growth because NH3, NH41+ and NO31- are all water soluble and can be leached from the soil. Nitrogen Cycle Human Intervention: In the atmosphere: N2 + O2 2NO (nitric oxide) produced when burning fuel or forests NO + O2 NO2 (nitrogen dioxide gas) NO2 + H2O HNO3 Nitric acid dissolved in water causes acid deposition N2O released from decomposition of fertilizer and waste. Nitrogen Cycle Excess nitrogen added to aquatic systems by run off of artificial fertilizer, farm waste and discharge of sewage. This stimulates growth of algae. Breakdown of algae by aerobic decomposers depletes water of oxygen. Oxygen Cycle (Photosynthesis) Sources of Oxygen: Photosynthesis and respiration Photo disassociation of H2O vapor CO2 and O2 circulates freely throughout the biosphere. Some CO2 combines with Ca to form carbonates. O2 combines with nitrogen compounds to form nitrates. O2 combines with iron compounds to form ferric oxides. O2 in the troposphere is reduced to O3 (ozone). Ground level O3 (ozone) is a pollutant which damages lungs. Phosphorus (P) Cycle Phosphorus (P) Cycle Component of DNA, RNA, ATP, proteins and enzymes Cycles in a sedimentary cycle – does not include the atmosphere A good example of how a mineral element becomes part of an organism. The source of Phosphorus (P) is rock. Phosphorus is soluble in H2O as phosphate (PO43-) Phosphate on land and in ocean sediment released by weathering into water and taken up by plants. Can be limiting factor for plant growth – is present in artificial fertilizer. Phosphorus (P) Cycle Animals get phosphorus by eating plants or other animals. Decomposition changes organic molecules with phosphorus back into phosphate which dissolves in water which returns the phosphorus to ocean sediment or deposited as rocks. Phosphorus (P) Cycle Human intervention: Mining of phosphate for fertilizers and soap causes disruption to ecosystems Removal of phosphorus from ecosystems by cutting down vegetation. Most phosphorus is taken up as biomass. Excess phosphorus in runoff from fertilizers, discharge of sewage, farm waste causes algae growth, etc (same as nitrogen) Sulfur (s) Cycle Component of protein Cycles in both a gas and sedimentary cycle but primary is an atmospheric cycle The source of Sulfur is the lithosphere (earth's crust) Sulfur (S) enters the atmosphere as hydrogen sulfide (H2S) during fossil fuel combustion, volcanic eruptions, gas exchange at ocean surfaces, and decomposition. H2S + O2 SO2 + H2 SO2 + O2 SO3 (sulfur trioxide) or SO2 + H2O H2SO4 (sulfuric acid) Acid deposition Sulfur (s) Cycle Sulfur in soluble form is taken up by plant roots and incorporated into amino acids such as cysteine. It then travels through the food chain and is eventually released through decomposition SO2 and water vapor makes H2SO4 ( a weak sulfuric acid), which is then carried to Earth in the form of acid rain. Sulfur (s) Cycle Human Intervention: Sulfur containing coal, when burned , releases SO2 Other industrial processes release SO2 into the atmosphere. Summary The building blocks of life :Water ,Nitrogen, Carbon Dioxide, Phosphorus, Sulfur Continually cycle through Earth's systems, the atmosphere, hydrosphere, biosphere, and lithosphere, on time scales that range from a few days to millions of years. These cycles are called biogeochemical cycles, because they include a variety of biological, geological, and chemical processes.