* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
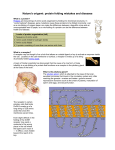
Download Neuroendocrine regulation of GnRH release and expression of
Survey
Document related concepts
Transcript
Vol. 10, No. 2 85 REVIEW Neuroendocrine regulation of GnRH release and expression of GnRH and GnRH receptor genes in the hypothalamus-pituitary unit in different physiological states Magdalena Ciechanowska1, Magdalena Łapot, Krystyna Mateusiak, Franciszek Przekop The Kielanowski Institute of Animal Physiology and Nutrition, Polish Academy of Sciences, Jabłonna, Poland Received: 31 July 2009; accepted: 17 June 2010 SUMMARY This review is focused on the relationship between neuroendocrine regulation of GnRH/LH secretion and the expression of GnRH and GnRH receptor (GnRHR) genes in the hypothalamic-pituitary unit during different physiological states of animals and under stress. Moreover, the involvement of hypothalamic GABA-ergic, β-endorphinergic, CRH-ergic, noradrenergic, dopaminergic and GnRH-ergic systems in the regulation of expression of the GnRH and GnRHR genes as well as secretion of GnRH/LH is analyzed. It appears that the neural mechanisms controlling GnRH gene expression in different physiological states may be distinct from those regulating GnRH/ Corresponding autor: The Kielanowski Institute of Animal Physiology and Nutrition, Polish Academy of Sciences, 05-110 Jabłonna, 3 Instytucka Street, Poland; e-mail: m.ciechanowska@ ifzz.pan.pl 1 Copyright © 2010 by the Society for Biology of Reproduction 86 Regulation of GnRH secretion LH release. The hypothalamic GnRHR gene is probably located in different neural systems and may act in a specific way on GnRH gene expression and GnRH release. Reproductive Biology 2010 10 2: 85-124. Key words: ewe, hypothalamus, GnRH mRNA, GnRHR mRNA, LH INTRODUCTION Gonadotropin cells in the anterior pituitary gland are regulated by the release of GnRH from the hypothalamus into the primarily portal capillaries in the median eminence (ME) and its delivery to the target via the hypophyseal portal vein [176]. The GnRH cells in the central nervous system are not segregated into neural clusters but appear as a loose network spred through many cytoarchitectonic structures. In most species, GnRH cells form a loose continuum from the diagonal band of Broca and more dorsal septal areas, to the bed nucleus of the stria terminalis and diencephalic areas. Also in this continuum there are cells lying dorsal to and within the supraoptic nucleus (SOP). GnRH release is controlled by numerous stimulatory and inhibitory factors as well as factors with biphasic effects on GnRH neurons [64, 201]. After the isolation and sequencing of porcine and ovine GnRHs, it appeared that these peptides isolated from several classes of vertebrates showed multiple substitutions in their sequence when compared with pig or sheep GnRH [66, 117, 125, 144, 172]. Until now, more than a dozen isoforms of GnRH sharing 10-50% amino acid identity has been found in vertebrates. The conservation of the length of these peptides, NH2 terminus and COOH terminus indicates that these features are critically important for receptor binding and activation. It is generally thought that most vertebrates possess at least two, and usually three, forms of GnRH which differ in their amino acid sequence, localizations and embryonic origins [21]. The most ubiquitous is chicken GnRH II which is the evolutionary conserved member of the GnRH peptide family. Its form in mammals differs from GnRH I by three amino acid residues at position 5, 7, 8 [21, 117]. It has been shown that the biological Ciechanowska et al 87 functions of GnRH I and GnRH II are different. Whereas GnRH I plays a pivotal role in the regulation of reproduction by stimulating the pituitary release of LH and FSH, GnRH II participates mainly in the control of puberty, reproductive behavior, feeding and energy balance [21]. Growing evidence shows that both GnRH I and GnRH II are potentially important autocrine/paracrine regulators in some extrapituitary compartments [21]. The actions of GnRHs are mediated by the GnRH receptors (GnRHR) which belong to a G protein-couple receptor (GPCR) subfamily. The GnRHR cDNA encodes a 327-328 amino acid protein with seven putative membranespanning domains characteristic for GPCR [86, 93, 117, 118, 182]. Only one conventional GnRH receptor subtype (GnRH-IR) uniquely lacking a carboxyl-terminal tail has been found in mammals [21]. The GnRH-IR lacks a typical intracellular carboxyl terminus, making it one of the smallest receptors with the seven-transmembrane segment motive. Recent cloning of the GnRH-II receptor subtype in non-human primates revealed that it is structurally and functionally distinct from the mammalian GnRH-IR [22]. The most striking difference between these receptor subtypes is the relation of a presence of 56-residue cytoplasmatic tail domain at the carboxyl-terminus of GnRH-IIR with its absence in GnRH-IR [21, 66, 144]. The presence of a cytoplasmatic tail domain alters receptor trafficking dynamics. Indeed recent studies have demonstrated that the marmoset GnRH-IIR undergoes a more rapid agonist-induced internalization compared with the human GnRH-IR [144]. In vitro GnRH I and GnRH II can bind with different affinities to GnRH receptor isoforms. The GnRHIR binds GnRH I with higher affi nity than GnRH-IIR, and GnRH-IIR has higher affinities for GnRH II than for GnRH I [66, 117, 125]. Other important sequence differences between receptor subtypes occur in the extracellular, transmembrane and cytoplasmic loop domains. In GnRHIIR receptors, the N-terminal domain is two-residues longer and more negatively charged than the GnRH-IR [66]. Excellent reviews have been written on these subjects, to which the reader can refer for a more description of GnRH I and GnRH II interaction with GnRH-R I and GnRH-R II and their physiological significance in the physiology of reproduction [21, 66, 117, 118, 144]. 88 Regulation of GnRH secretion REGULATION OF GNRH/LH RELEASE AND EXPRESSION OF GNRH AND GNRHR GENES DURING THE ESTROUS CYCLE AND ANESTROUS PERIOD IN EWES The regulation of GnRH secretion in mammals is associated with complex interplay among excitatory and inhibitory neurotransmitter- and neurohormone -systems activity within the hypothalamus (fig. 1). The activities of these systems and their functions in the control of GnRH release from the hypothalamus during the estrous cycle depend mainly upon estrogen and progesterone (P4) secretion [51, 71, 94, 156]. Both estradiol (E2) and P4 exert positive and negative action on GnRH secretion. In the interval between luteolysis and preovulatory gonadotropin surges in ewes, E2 exerts a dosedependent suppression of GnRH from the hypothalamus [37]. This effect is mediated by a reduction in the GnRH pulse size and occurs despite a stimulatory action on GnRH pulse frequency [50]. Whereas both E2 and P4 inhibit episodic GnRH release, high circulating levels of E2 during preovulatory gonadotropin surge stimulate GnRH output [18, 51]. The ability of E2 to activate GnRH cells in the hypothalamus in ewes is affected by P4 through the temporal relationship between E2 and P4 exposure. Indeed, in the natural estrous cycle, the preovulatory surge of GnRH is stimulated by elevated E2 secretion during the follicular phase in which P4 concentration is low. In contrast, increases in E2 secretion that accompany follicular growth during the luteal phase are unable to stimulate GnRH release despite being of sufficient magnitude, due to the presence of enhanced P4 concentration. However, exposure to P4 prior to an independent increase of E2, which occurs during the follicular phase, augments GnRH surge in ewes [51]. Progesterone by itself has not been shown to inhibit GnRH secretion consistently in ovariectomized ewes, and it has been suggested that E2 is necessary to sensitize the neuroendocrine axis to negative feedback action of progesterone [156]. It has also been shown that the circulating level of E2 is a regulator of physiological synaptic plasticity in the arcuate nucleus (ARC) in rats [128]. In the late follicular phase of the estrous cycle the level of E2 begins to rise resulting in both the inhibition of GnRH release and sensitization of the pituitary gonadotrops to GnRH. It has been documented Ciechanowska et al 89 5 -HT HT ? ER NA/NPY A1 ER NA A2 GABA DA CRH PVN DA NPY β-END Figure 1. Afferent inputs of some neural systems to GnRH neurons in the preoptic area; stem brain nuclei: A1, A2; nucleus arcuatus: ARC; corticotropin neurons: CRH; dopamine neurons: DA; estradiol receptors: ER; gamma-aminobutyric acid neurons: GABA; serotonine neurons: 5-HT; noradrenaline neurons: NA; noradrenaline and neuropeptide Y neurons: NA/NPY; neuropeptide Y neurons of ventromedial hypothalamus: NPY; β-endorphin: β-END; preoptic area: POA; periventricular nucleus: PVN; ventromedial nucleus: VM; triangle: synapse; rectangle: close apposition; circle: nerve terminals. that the abrupt and exquisitely timed release of GnRH at the peak of circulating estrogen in rats follows the disruption of inhibitory synapses in the ARC that have been supporting the decrease of GnRH/gonadotropins release 90 Regulation of GnRH secretion during increasing pituitary sensitization to GnRH [128]. The estrogen with GnRH regulates GnRH receptor activity in the anterior pituitary gland and LH release in ewes [200]. On the other hand, GnRH/LH may regulate estrogen level which suggests considerable crosstalk between these two processes [128]. In ewes, E2 enhances GnRH-mediated release of LH from the pituitary gland during the breeding season but not during anestrus or the period of transition into or out of the breeding season [126]. However, in anestrous ewes treatment with E2 but not P4 decreased pituitary LHβ mRNA in the hypothalamic-pituitary disconnected animals [42]. In cycling ewes, P4 suppresses GnRH-R gene expression in the anterior pituitary gland [200]. However, the mechanism of P4 inhibitory action on the expression of GnRHR gene in the anterior pituitary gland has not been fully recognized. There is evidence that P4 negatively influences GnRHR gene expression through E2 as well as GnRH receptor mechanism on gonadotropin cells. Since E2 affects its target cells via intracellular receptors and GnRH binds to receptors on the plasma membrane of gonadotropic cells, P4 is assumed to affect GnRHR biosynthesis through different cellular mechanisms [200]. The numerous studies on rats have also documented that E2 [11, 146, 161, 164, 196] and P4 [23, 97], in different physiological states and experimental conditions, may increase or decrease GnRH gene expression in the hypothalamus. In the ovariectomized progesterone-estradiol treated ewes, GnRH/LH release was not closely associated with the GnRH mRNA level suggesting that the role of E2 in the neuroendocrine mechanism controlling GnRH gene expression in the hypothalamus is distinct from that regulating GnRH release [70]. Progesterone displays an exclusive regulatory influence on GnRH release from the hypothalamus in ewes but has no effect on GnRH gene expression in these species [163]. On the other hand, P4 seems to decrease GnRH gene expression in cows [205]. The effects of ovarian steroids on the expression of GnRH gene in the hypothalamus may be species-specific. The experiments on the relationship between the GnRH mRNA level in the hypothalamus and LH secretion in sheep have answered the question to what extent the patterns of LH secretion in various physiological states are related with GnRH gene expression [25, 27, 111]. It has been shown that Ciechanowska et al 91 GnRH mRNA level in the entire hypothalamus of anestrous ewes was significantly lower than in the follicular and/or luteal phase animals (fig. 2). The low GnRH gene expression in the hypothalamus of anestrous ewes may be responsible for the decrease in GnRH biosynthesis and release as well as the non-ovulatory state of ewes during a long photoperiod. It is worth noting, that the level of GnRH mRNA in the hypothalamus is the net result of the transcriptional activities of the GnRH gene, the degradation of transcriptional product and the utilization of cytoplasmic GnRH mRNA in the biosynthesis of GnRH. Thus the GnRH mRNA level may not reflect a clear relationship between the transcriptional activity of GnRH gene with GnRH release. Similarly to GnRH gene expression, the GnRHR mRNA level in the hypothalamic-pituitary axis (fig. 2) of anestrous ewes is lower than in the follicular and/or luteal phase female [25, 27, 111]. Several experiments on rats have documented that the estrogen and P4 affect the GnRHR gene expression in the hypothalamus and the anterior pituitary gland, but their role in rats may be different [84, 174]. In the anterior pituitary glands of rats [12, 208] and ewes [1, 100, 199, 200], E2 increases GnRHR gene expression, but P4 suppresses GnRHR mRNA level [199, 200]. The stimulatory effect of GnRH on GnRHR gene expression in the anterior pituitary gland in female rats [12, 87, 109, 199, 200] and sheep [100, 200] is markedly enhanced by E2. Homologous up – [12, 87, 109, 199, 208] or down – [1, 22, 166, 200, 207] regulation of GnRHR gene expression by GnRH in the anterior pituitary gland depends to a high degree on the mode of treatment (continuous vs. pulsatile) and concentration (high vs. low) of GnRH. The physiological significance of GnRHR mRNA in the hypothalamus for GnRH gene expression and GnRH release still remains not well understood. The results obtained on rats [84, 174] and sheep [26] suggest that GnRHR mRNA in the hypothalamus may be involved in the control of GnRH release from the GnRH nerve terminals as well as GnRH gene expression in the preoptic area (POA). The fact that the GnRH neurons in the hypothalamus have a few or no steroid receptors [75, 76, 103] has lead to the conclusion that E2 and P4 affect GnRH cell activity primarily via the steroid-receptive inter-neural systems in the hypothalamus. Among the number of neural systems in the hypothalamus affecting directly and/or indirectly GnRH activity, the no- Regulation of GnRH secretion 92 2 A Anestrous GnRH/GAPDH (arbitrary units) Luteal phase Follicular phase 1,5 1 ** ** 0,5 2 ** * 0 GnRH/GAPDH (arbitrary units) ** AH POA ** VM B *** 1,5 *** 1 0,5 0 ** ** POA ** AH ** VM ** ** SME AP Figure 2. GnRH (A) and GnRH receptor (GnRHR) mRNA (B) levels (mean±SEM) in the hypothalamic-pituitary unit of anestrous, luteal and follicular phase ewes (n=6). GnRH and GnRHR mRNA levels are presented as GnRH or GnRHR mRNA/ GAPDH mRNA ratio where GAPDH (glyceraldehyde 3-phosphate dehydrogenase) is a reference gene. The significance of differences in the mRNA levels among groups was assessed by one-way ANOVA followed by Tukey’s test, asterisks depict differences between anestrous and luteal or follicular phase ewes; *p≤ 0.05, **p≤ 0.01, ***p≤ 0.001; preoptic area: POA; anterior hypothalamus: AH; ventromedial hypothalamus: VM; stalk/median eminence: SME; anterior pituitary gland: AP. Ciechanowska et al 93 radrenergic cells from the brain steam [43, 75, 171], GABA-ergic neurons from the POA [43, 75] and β-endorphin perikarya from the ARC [29, 43, 75] are best recognized in synchronizing the firing of different groups of GnRH neurons in the hypothalamus. All noradrenergic cells from the A1 and A2 groups (nuclei of the brain stem) in rats project axons to the POA and synapse with GnRH perikarya and may directly stimulate GnRH release through α1 receptors located on the GnRH cells [43, 75]. The location and exact targets of noradrenergic neurons that affect GnRH secretion at the GnRH nerve terminals within the ME are unknown [43, 75]. Axons from noradrenergic cells also activate GABA perikarya in the POA to suppress GnRH secretion [43]. In the luteal phase ewes, P4 increases GABA-ergic activity and decreases GnRH pulse frequency through the GABA-ergic system [43]. During the early follicular phase, the period of declining P4 secretion, estradiol inhibits the autoinhibitory system of GABA cells, so, the increased GABA concentrations are maintained and suppress GnRH/LH release [43]. Prolonged secretion of E2 during the follicular phase leads to an increase in the stimulatory influence of noradrenaline (NA) on GnRH cells in the POA with a concomitant suppression of GABA output in this structure. The inhibitory influence of GABA on GnRH release in the follicular phase ewes occurs through GABAA receptors, whose activation by muscimol (agonist of GABAA receptors) decreases GnRH/LH release [26]. The GnRH perikarya in the POA are also under the inhibitory influence of opioid neurons from the ARC [43, 179, 189]. Indeed, the opioid perikarya project axons to the POA and synapse directly with GnRH cells [43]. Opioid neurons also synapse with NA interneurons in the POA [43, 179]. There is good evidence of a presynaptic inhibition of noradrenaline release by β-endorphin in the POA of the rat [14, 41, 92, 168] and sheep [96]. The estrogen-induced decrease in tonic input of opioids on noradrenergic neurons may further contribute to the elevation of preoptic noradrenaline concentration and stimulate GnRH surge. This data indicate that noradrenergic neurons projecting to the vicinity of the GnRH cells are likely to be stimulated directly by estrogen and in association with reduced presynaptic opioid restraint, are responsible for the increased noradrenaline output within 94 Regulation of GnRH secretion the POA [75]. It is interesting that a decrease in opioid activity just prior to the LH surge occurs synchronously with the uncoupling of NA influence on GABA cells in the POA [43]. In rats, opioids restrain stimulatory catecholaminergic, glutaminergic and nitric oxide inputs and enhance the inhibitory action of GABA on GnRH neurons [52]. The presented results have shown that the activity of GABA and GnRH perikarya in the POA are modulated by brain stem noradrenergic neurons in response to a change in estrogen and P4 secretion. The estradiol-induced increase of GnRH mRNA in the POA in rats is blocked by pretreatment with α-adrenergic receptor antagonist suggesting that the noradrenergic system in the hypothalamus is involved in the regulation of GnRH gene expression and biosynthesis [75, 108]. Similarly, the suppression of noradrenergic neurotransmission significantly reduces GnRH mRNA level in the hypothalamus [98, 99]. Much less is known about the gonadal steroid-induced changes in NA release within the ME, although this region in rats, is particularly sensitive to ovarian hormones. However, it seems that the estrogen-dependent increase of NA release in ME is likely to alter both GnRH release and biosynthesis. It is suggested that NA stimulates GnRH secretion within the ME by inducing the synthesis of prostaglandin E2 and nitric oxide in male rats [137, 138, 155]. In anestrous ewes, the activation of GABAB receptors agonist in the mediobasal hypothalamus-ME by specific GABAB receptor agonist, baclophen, stimulates GnRH release; its action probably occurs by suppressing inhibitory action of NA on GnRH release [193]. The presented results have indicated that physiological regulation of hypothalamic GnRH release and GnRH gene expression is associated with complex interplay of gonadal hormones within different hypothalamic neural systems. EFFECT OF STRESS ON GNRH/LH RELEASE AND EXPRESSION OF GNRH AND GNRHR GENES DURING DIFFERENT PHYSIOLOGICAL STATES It is well documented that stressors alter the activity of many neural systems in the hypothalamus which stimulate or inhibit GnRH secretion and Ciechanowska et al 95 that the GnRH cell response are dependent upon the kind of stress, its duration, physiological state of animals and gender. It has been shown that immediate response of GnRH cells to acute stress depends primarily upon central neural systems and in some circumstances may by stimulatory in rats [159, 185], monkeys [74] and sheep [111, 190]. These animals exposed to acute stress respond with a small and often short-lived increase in LH secretion. Prolonged stressful stimuli additionally modulate GnRH secretion by hormones, neurotransmitters, neuropeptides, excitatory and inhibitory amino acids released during the time-course of stressful stimuli, and all these compounds released from the hypothalamo-pituitary-gonadal and hypothalamo-pituitary-adrenal axes act mainly within the hypothalamus and anterior pituitary gland to mediate the suppressive effect on gonadotropin secretion [159]. Indeed, an inverse relationship between prolonged or chronic stress and normal reproductive efficiency has frequently been observed in domesticated animals [10, 31, 32, 38]. The psycho-emotional state evoked by prolonged intermittent footshock stimulation of cyclic ewes suppresses the preovulatory LH surge and, consequently, causes long-lasting disturbances in the course of the estrous cycle in some animals [152]. Premating stress has also been linked with lowering of the ovulation rate in ewes [45] and inhibition of the preovulatory LH surge in heifers [121, 181]. Confinement stress stimuli affect episodic secretion of LH in ovariectomized sheep [154]. Diestrous rats exposed to footshocks display marked changes in the course of the estrous cycle during the post-stress period [24]. Recent findings revealed that stress affects GnRH and LH secretion by different neuroendocrine mechanisms. Direct monitoring of the pulsatile GnRH secretion in the pituitary portal blood has shown that psychological stress in ewes reduces the amplitude of GnRH pulses; this process is independent of the stress-induced cortisol level [203]. However, prolonged enhanced secretion of cortisol contributes to the suppression of GnRH pulse frequency but only in the presence of ovarian steroids [135]. Studies on ovariectomized sheep have indicated that either psychological stress [16, 180] or an increment of plasma cortisol level compared to that attained during psychological stress [17, 180] acutely reduces LH pulse amplitude 96 Regulation of GnRH secretion by two mechanisms. First mechanism involves cortisol action via type II glucocorticoid receptor to inhibit pituitary responsiveness to GnRH, and second by changes in the profile of hypothalamic GnRH secretion (changes in GnRH pulse amplitude and pulse frequency). A number of studies have indicated a sex differences in the inhibitory effect of cortisol on pulsatile LH secretion in sheep with rams being more sensitive than ewes [180], and that E2 increases sensitivity to the inhibitory actions of cortisol on LH secretion and GnRH/LH pulse frequency [135, 136]. However, in the hypothalamo-pituitary disconnected sheep model, cortisol did not reduce the amplitude of LH pulses driven by exogenous GnRH [180]; the mechanism of this action is unknown. It may suggest that cortisol acts indirectly via the hypothalamus to elicit a mediator that inhibits its response to GnRH and/or that the undisturbed communication with the hypothalamus is required to maintain cells in the anterior pituitary responsive to cortisol. The above findings help to explain why responses to stress differ between female and male as well as in various physiological states of females. It is worth noting that different kinds of stress may exert a specific effect on hypothalamic-pituitary-ovarian activity. For example, endogenous CRH participates in the suppression of the hypothalamic-pituitary-ovarian axis activity during some challenges, such as footshocks [160], fasting [114] and lactation [204] but not in others, such as immune activation [15, 157]. These observations suggest that different stressors may act differently on the hypothalamic stimulatory and/or inhibitory systems involved in the control of GnRH cell activities; the final effect of stress on GnRH release is determined by the net results of these systems. It was reported that different stressors may act in various manners on the activities of particular neural systems in the brain [140]. In this respect, the stressors are divided into four main categories: 1/ physical stressors, that have either a negative or a positive psychological components; 2/ psychological stressors, that reflect a learned response to previously experienced adverse conditions; 3/ social stressors reflecting disturbed interactions among individuals and 4/ stressors that change metabolic homeostasis. Despite a number of studies on GnRH release in animals under stress condition, there is no coherent evidence how stress affects the interneural Ciechanowska et al 97 events related with biosynthesis of GnRH and GnRHR in the hypothalamicpituitary axis. The few results on laboratory animals have indicated that the expression of GnRH and GnRHR genes, similarly as the gonadotropin secretion, is highly dependent upon the kind of stress, its duration, physiological states of animals and gender. For example, cold stress suppresses GnRH gene expression in the POA in rats [185], while prolonged food deprivation stress does not affect GnRH gene transcription in the hypothalamus of cycling female rats [129] but provokes a deep inhibition of GnRHR mRNA in the anterior pituitary gland (AP). In contrast, in male rats a decreased number of neurons expressing GnRH mRNA in the hypothalamus were found after prolonged fasting stress [65]. Acute neurogenic stress caused similar downregulation of GnRHR gene expression in the anterior pituitary gland throughout the entire estrous cycle, but attenuated the GnRHR mRNA level in the hypothalamus in a relatively short period at proestrus [129]. In the castrated male rats, centrally administered IL-1β decreased GnRHR mRNA level in the hypothalamus and LH secretion [90]. Thus, differences in the GnRH and GnRHR gene expression in animals under various stressors during the different reproductive states may activate or inhibit different afferents to GnRH and GnRHR expressing neurons in the hypothalamus. In anestrous ewes, short as well as prolonged footshock stimulation significantly increase GnRH mRNA and GnRHR mRNA levels in the hypothalamic-pituitary axis, but the increase of LH secretion in these animals was noted only during acute stress [111]. These results indicate that the neuroendocrine processes involved in the expression of GnRH and GnRHR genes under short or prolonged stressful stimuli are distinct from those regulating GnRH/LH secretion (tab. 1). In the follicular phase ewes, the GnRH gene expression increased after short footshock stimulation, whereas prolonged stressful stimuli evidently decreased GnRH mRNA level in the hypothalamus [25]. In short-stressed ewes, the significant increase in mRNA encoding GnRHR was detectable in the hypothalamic-pituitary axis. However, the GnRHR mRNA levels were reduced in the hypothalamus/stalk median eminence (SME) and AP but were elevated in the POA of these animals. The changes in GnRH mRNA and GnRHR mRNA in the hypothalamic-AP of the follicular phase ewes Regulation of GnRH secretion 98 Table 1. Effects of short and prolonged footshocks stimulation* on GnRH and GnRHR mRNA levels in the hypothalamic-anterior pituitary unit as well as LH concentration in blood plasma of anestrous (A) and follicular phase (B) ewes Prolonged footshocks Short footshocks stimulation stimulation mRNA GnRH GnRHR GnRH GnRHR Area POA AH VM SME AP LH A B A B A B A B ↑ ↑ ↑ nd nd ↑ ↑ ↑ nd nd ↑ ↑ ↑ ↑ ↑ ↑ ↑ ↑ ↑ ↑ ↑ ↑ ↑ nd nd ↓ ↓ ↓ nd nd ↑ ↑ ↑ ↑ ↑ ↑ ↓ ↓ ↓ ↓ A ↑ B ↑ A -- B ↓ *stress was induced by repetitive trains of 3 mA alternative current of 0.5 s on and 1 s off during 20 min period of every hour; short footshocks stimulation: 3 h daily during one day (08:00 – 11:00 am); prolonged footshocks stimulation: 5 h daily over four days (08:00 am – 01:00 pm); GnRHR: GnRH receptor; POA: preoptic area; AH: anterior hypothalamus; VM: ventromedial hypothalamus; SME: stalk/median eminence; AP: pituitary gland. Non stressed animals served as a control.↑: increased level, ↓: decreased level, –: no changes, nd: not detected. under short and/or prolonged stressful stimuli were related in a parallel way with LH secretion (tab. 1) suggesting the existence of a direct relationship between GnRH mRNA and GnRHR mRNA expression with GnRH and GnRHR protein biosynthesis and GnRH release. The significance of GnRHR mRNA in the biosynthesis of GnRH and its release in the hypothalamus of anestrous and follicular phase ewes needs further studies. Similarly, the differences in the expression of GnRHR gene in the POA compared with those in the hypothalamus/SME cannot be explained adequately in follicular phase ewes subjected to prolonged stress. It is likely that the GnRHR in the discrete hypothalamic structures are located in different neural systems, which react in a specific way to stressful stimuli. The differences in the expression of GnRH and GnRHR genes in the hypothalamicpituitary axis in anestrous and follicular phase ewes subjected to prolonged Ciechanowska et al 99 stress may result, as in the case of GnRH release, from the different action of some neural systems in the hypothalamus on activities of GnRH and GnRHR gene-expressing cells during breeding and non-breeding season. INFLUENCE OF GABA-ERGIC SYSTEM IN THE HYPOTHALAMUS ON THE GNRH/LH RELEASE AS WELL AS GNRH AND GNRHR GENES EXPRESSION IN THE HYPOTHALAMIC-ANTERIOR PITUITARY UNIT IN FOLLICULAR PHASE EWES The noradrenergic system in the hypothalamus of ewes has been shown to activate GnRH/LH release from the mediobasal hypothalamus just prior to the preovulatory LH surge [44], while in the anestrous ewes it has a rather inhibitory influence on gonadotropin secretion [62, 193]. Consistent results from several laboratories have shown that GABA-ergic, opioidergic and dopaminergic systems also exert a different effect on GnRH release in ewes during breeding and non-breeding seasons. A large body of evidence indicates that GABA-ergic neurons in the hypothalamus are involved in controlling GnRH release [162]. GABA, the dominant inhibitory neurotransmitter in the hypothalamus, affects GnRH release by two different classes of membrane receptors: GABAA [153, 175] and GABAB [95, 127]. GABA-ergic synapses has been identified on GnRH neurons [82, 106, 178], and GnRH cells express both types of receptors [85, 124, 141, 145]. It has been documented that GABA acting through a GABAA- and GABAB – receptor mechanism may stimulate [53, 78, 79, 80, 83, 122] or inhibit [4, 72, 120, 169, 170, 194, 195] GnRH release and GnRH cell activities dependent upon the physiological state of animals, site of its action and experimental conditions. GABA-ergic neurons may also serve as a primary integrating center for many different inputs to GnRH. Signals from steroid hormones [43, 184] and neural systems in the hypothalamus [43, 183] have been shown to alter the GABA-ergic drive to GnRH neurons which may contribute to a change in GnRH output. 100 Regulation of GnRH secretion It is generally accepted, that muscimol, a selective agonist of GABAA receptor, decreases LH secretion in sheep [169, 170, 194, 195]. The effects of bicuculline (GABAA receptor blocker) on this hormone secretion, however, have been variable. In ovariectomized ewes, injection of bicuculline into the POA inhibited LH secretion under all conditions except the breeding season when LH secretion was greatly suppressed by estrogens [169, 170]. Perfusion of bicuculline into the POA or ventromedial hypothalamus (VM) of follicular-phase ewes had no evident effect on extracellular concentrations of GnRH in the perfusates [194, 195]. Dialysis of this drug into the POA of castrated rams reduced LH release [55]. Thus, in sheep the effect of a blockade of GABAA receptor on GnRH/LH release is highly dependent upon physiological states and gender. It has been documented in in vitro studies that the activation of GABAA receptors depolarizes as well as hyperpolarizes GnRH cells [39, 67] and that the endogenous GABA can excite and inhibit the firing of GnRH neurons [68, 122]. Studies exploring the effects of GABAA receptor antagonists on GnRH neurons showed that the majority of these cells were depolarized and/or excited by blocking of GABAA receptors [68]. The mechanism underlying the effect(s) of GABA on the release of GnRH and GnRH cell activity is still poorly understood. Perhaps the most reasonable explanation for contradictory data may be the assumption that GABAA receptors are located not only on GnRH neurons but also on numerous stimulatory and inhibitory interneurons that impinge on GnRH cells; the final effect of their action is determined by the net results of inhibition and disinhibition of these systems controlling the GnRH neurons activity. It is also documented that in rodents, GABA acts via GABAB receptors to reduce GnRH secretory activity. Pharmacological activation of these receptors by agonists inhibits the preovulatory LH surge [3, 4, 5] as well as the neurotransmitter-mediated stimulation of GnRH/LH release [6, 72, 116]. In addition, GABAB receptor systems are involved in mediating the suppressive actions of GABA on pulsatile LH release [5]. In contrast, in ovariectomized-estrogen treated ewes [170] and castrated testosteroneprimed rams [54, 80] during non-breeding season, the activation of GABAB receptors in the POA or VM by baclofen (a GABAB receptor agonist) greatly increases GnRH/LH secretion. On the other hand, neither activation nor Ciechanowska et al 101 the blockade of GABAB receptors in ovariectomized estrogen-treated ewes during the breeding season affects GnRH/LH secretion [169, 170]. Available data have shown that changes in GABA-ergic neurotransmission in the hypothalamus of rats also affect the expression of GnRH [56, 89, 105] and GnRHR [165, 173] genes. The modulation of the activities of GABAA and GABAB receptors strongly suggests that each receptor acts on GnRH gene expression in a specific way and that the influence of GABAA receptors on GnRH gene expression mostly depends upon the experimental condition. It has been shown that chronic stimulation of GABAA receptor decreases GnRH transcript [56, 107] whereas acute treatment with low, but not high, doses of muscimol increases GnRH mRNA levels in the POA/anterior hypothalamus (AH) area in rats [89]. Stimulation of GABAB receptors with baclofen augments hypothalamic GnRH mRNA level in a dose-dependent manner [89]. In follicular phase ewes, the prolonged modulation of GABAA receptor activities in the hypothalamus profoundly affects the expression of GnRH and GnRHR genes in the hypothalamic-pituitary axis (tab. 2). The activation of GABAA receptors caused a significant reduction in GnRH and GnRHR transcripts, whereas inhibition of GABAA receptor activity increased GnRH mRNA and GnRHR mRNA levels suggesting that down- or up- regulation of GnRH and GnRHR gene expression is dependent upon central GABA-ergic action [26]. The decrease or increase in hypothalamic GnRH mRNA and GnRHR mRNA levels of ewes were parallel to GnRHR gene expression in the anterior pituitary gland and LH secretion. They indicate that GABA acting through the GABAA receptor mechanism on GnRH and GnRHR genes expression in the hypothalamus is involved in biosynthesis of GnRH and GnRHR protein and release of GnRH. THE CENTRAL EFFECT OF Β-ENDORPHINERGIC SYSTEM ON THE GNRH/LH RELEASE AS WELL AS GNRH AND GNRHR GENES EXPRESSION IN THE HYPOTHALAMIC-ANTERIOR PITUITARY UNIT IN FOLLICULAR PHASE EWES Evidence for the role of opioidergic peptides in the hypothalamus within the GnRH network has shown that β-endorphin constitutes important *the infusions of Ringer’s solution into the third cerebral ventricle of control ewes were performed at a rate of 2 μl/min during 20 min of every hour for five hours daily (08:00 am – 01:00 pm) on 14th – 16th days of the estrous cycle. The drug-treated ewes received infusion of β-endorphin, naloxone (antagonist of μ-opioid receptors), muscimol (agonist of GABAA receptors), bicuculline (antagonist of GABAA receptors) CRH or α-helical CRH9-41 (CRH-antagonist; 20 µg of respective drug/ml Ringer’s solution). GnRHR: GnRH receptor; POA: preoptic area: AH: anterior hypothalamus; VM: ventromedial hypothalamus; SME: stalk/median eminence; AP: pituitary gland. In comparison to control ewes: ↑: increased level, ↓: decreased level, –: no changes, nd: not detected. POA AH VM SME AP LH B C β-endorphin nalokson muscimol biccuculine CRH CRH-antagonist GnRH GnRHR GnRH GnRHR GnRH GnRHR GnRH GnRHR GnRH GnRHR GnRH GnRHR ↓ ↑ --↓ ↓ ↑ ↑ ↓ ↑ ↑ ↓ ↓ ↓ --↓ ↓ ↑ ↑ ↓ ↓ ↑ -↓ ↓ --↓ ↓ ↑ ↑ ↓ ↓ ↑ ↑ ↓ -↓ ↑ ↓ ↑ nd nd nd nd nd nd ↓ -↓ ↑ ↓ ↑ nd nd nd nd nd nd ↓ -↓ ↑ ↓ ↑ A Table 2. Effect of stimulation or inhibition of µ-opioidergic (A), GABAA-ergic (B), and CRH-ergic (C) receptors on GnRH and GnRHR mRNA levels in the hypothalamic-anterior pituitary unit and LH concentration in blood plasma of follicular phase ewes* 102 Regulation of GnRH secretion Ciechanowska et al 103 inhibitory compound of the neural circuitry in the hypothalamus that involved in the central control of GnRH secretion in ewes and other species during the estrous cycle [33, 36, 44, 52, 76, 77, 197]. Cell bodies containing β-endorphin are exclusively located in the periarcuate region of the hypothalamus [177, 179, 206]. β-endorphin nerve terminals synapse on perikarya and dendrites of GnRH neurons [82, 189] and can affect GnRH release at two levels: POA and VM /SME. The GnRH fibers in the hypothalamus also contain opioid receptors [48, 75, 179] and β-endorphinergic neurons are connected indirectly with GnRH cells through GABA-ergic [52], nitricoxidergic [52], noradrenergic [43, 75, 91, 92], dopaminergic [192, 197] and NPY-ergic cells [43] raising the possibility that β-endorphinergic sensitive interneurons may also indirectly modulate GnRH release and GnRHR gene expression. It was found that β-endorphin has a suppressive effect on GnRH/LH release in ewes during the estrous cycle but not in seasonally anestrous animals [29, 197]. Recent studies have shown that infusion of β-endorphin into the third cerebral ventricle of follicular phase ewes decreases GnRH gene expression in the hypothalamus and leads to complex changes in GnRHR mRNA level in discrete hypothalamic areas (tab. 2): an increase in the POA, no changes in the AH and a decrease in VM/SME [29]. In β-endorphintreated ewes, the GnRHR mRNA level in the anterior pituitary gland and LH secretion decreased significantly. This suggests that β-endorphin may participate in the processes of biosynthesis of GnRH and GnRHR. However, the physiological importance of GnRHR mRNA in different parts of the hypothalamus in the control of GnRH secretion waits to be established. The inverse relationship between the preoptic area GnRHR mRNA and GnRH mRNA in the β-endorphin-treated ewes raises the possibility that GnRHR gene-expressing neurons in this structure may be involved in the control of GnRH cell activities. This suggests that the increase of GnRHR neurons in the POA may down-regulate GnRH gene expression. On the other hand, the decrease of GnRHR mRNA in the VM/SME in these animals may be due to an increase in the utilization of GnRHR mRNA in posttranscriptional processes in the biosynthesis of GnRHR. Increased activity of GnRHR in the VM may suppress GnRH release. It is generally 104 Regulation of GnRH secretion believed that the activation of GnRHR in the VM can exert an inhibitory [40, 167] effect on GnRH release in rats and an increase in GnRHR mRNA level in this structure [84, 124]. EFFECT OF MODULATION OF THE CRH-ERGIC ACTIVITY ON THE GNRH/LH SECRETION AND EXPRESSION OF GNRH AND GNRHR GENES IN THE HYPOTHALAMIC-ANTERIOR PITUITARY UNIT IN FOLLICULAR PHASE EWES CRH is one of numerous neurotransmitters/neurohormones in the central nervous system that interact together in various structures of the brain to alter the activity of GnRH neurons during certain challenges. Most in vivo and in vitro studies have shown that CRH can act directly on GnRH perikarya via synaptic contacts in the POA as well as on GnRH nerve terminals in the SME to suppress GnRH release [58, 75, 113, 130]. Evidence of CRH interferences with GnRH neural activity was also confirmed in experiments performed on rats in which CRH antagonist reversed or attenuated the inhibitory influence of CRH on GnRH/LH secretion [114, 158, 160]. However, the pathways and mode of action of CRH on GnRH cells is still not well recognized. CRH action on GnRH neurons may represent a local connection between CRH and GnRH cells in the POA as well as neural CRH projection from other hypothalamic and extrahypothalamic areas to GnRH neurons [43, 119, 158, 159]. A number of immunohistochemical and physiological studies have also shown that the mechanism through which CRH affects GnRH release involves the modulation of other pathways including catecholaminergic, serotoninergic, GABA-ergic, opioidergic systems. The concept that catecholaminergic systems are involved in CRHinduced suppression of GnRH/LH secretion is based on observations that systemic- or intracerebroventricularly (icv)-injected CRH inhibits GnRH/ LH secretion [158] and facilitates NA release in rats [47, 49] and NA output in sheep [151, 191]. CRH administered intraventricularly or directly into the locus coeruleus in rats increases the release of NA in areas receiving projections from this structure [43]. CRH containing axon terminals synapse onto Ciechanowska et al 105 catecholamine dendrites and may presynaptically modulate other afferents in the rostral pole of locus coeruleus in the rat brain [202]. With respect to serotonin, it has been presented that the administration of CRH inhibits 5-HT release and raphe neural activity [149]. In rats, the serotoninergic axon terminals from raphe nuclei project into the POA to enhance GnRH secretion [179]. Similarly, it has been documented that CRH synapse on GABA cells rising the opportunity to react indirectly with GnRH neurons [34, 43]. CRH stimulates GABA-ergic system; an infusion of CRH into the locus coeruleus resulted in a significant increase in the percentage of immunoreactive GAD67 (isoform of glutamic acid decarboxylase) within the POA [119]. Additionally, CRH cooperates with the opioidergic system in the hypothalamus in the control of GnRH release [131, 132, 133]; its effect is dependent upon sex steroids [19, 20] and may be species-specific [158]. It has been shown that CRH inhibits LH secretion in gonadectomized monkeys and rats through a mechanism that depends partially on opioid tone [8, 59, 139, 147]. However, a similar stimulation during follicular and luteal phases is totally ineffective in monkeys [134]. On the other hand, long-term gonadectomized rats do not respond along with the suppression of LH secretion to opioid [7] or CRH treatment [132]. In ovariectomized or intact sheep, peripheral or central administration of CRH during anestrus or breeding season has generally no effect on GnRH release [31, 46, 142]. However, during the preovulatory period the icv infused CRH does not affect tonic LH secretion but suppresses the preovulatory LH surge and prolongs the estrous cycle [148]. In contrast, GnRH/LH secretion is stimulated by CRH during non-breeding and breeding seasons in ovariectomized progesterone-estradiol-treated ewes in which LH pulse frequency is low [19]. The experiments on the immortalized neural cell line (Gn11) evidenced that CRH-like peptides affect GnRH gene expression acting through CRH receptors on these cells [186, 187]. The infusion of CRH or CRH antagonist (α helical CRH 9-41) into the third cerebral ventricle of ewes caused a decrease or increase in GnRH gene expression in the hypothalamus, respectively, and led to different responses in GnRHR gene expression in the discrete parts 106 Regulation of GnRH secretion of the hypothalamus [30]. CRH increased GnRHR mRNA in the POA and decreased it in the AH and VM, and SME (tab. 2). In CRH-treated animals GnRHR mRNA decreased significantly in the anterior pituitary gland and LH in blood. The blockade of CRH receptors exerted opposite effects on GnRHR gene expression to that induced by CRH. Presented results suggest that CRH acting through CRF-receptor mechanism(s) in the hypothalamus may decrease GnRH biosynthesis and its release through increased GnRHR expression in the hypothalamus. Altogether, the decrease of GnRH mRNA level in the hypothalamus after the stimulation of µ-opioidergic, GABAA-ergic and CRH-ergic receptors with a concomitant decrease in LH secretion suggests that the activation of these receptors has a suppressive effect on GnRH biosynthesis and consequently on GnRH release. Such an assumption seems to be supported by observation that the blockade of these receptors leads to an increase of GnRHR mRNA level in the anterior pituitary gland and LH secretion. The specific responses in GnRHR gene expression in the discrete parts of the hypothalamus to agonists and/or antagonists of μ-opioidergic, GABAA-ergic and CRH-ergic receptors in follicular phase ewes, suggests that the GnRHR in various parts of the hypothalamus are located on different neural systems. It is likely that the decrease or increase in GnRHR mRNA levels in the VM/ SME after the stimulation or inhibition of these receptors may result from the increase or decrease, respectively, in the utilization of GnRHR mRNA in the biosynthesis of GnRH-R in this structure. The increase of GnRHR activity in this structure of rats has a suppressive effect on GnRH release [40, 167]. IMPLICATION OF DOPAMINERGIC SYSTEM IN THE HYPOTHALAMUS OF ANESTROUS EWES ON LH RELEASE AS WELL AS GNRH- AND GNRH-R GENES EXPRESSION IN THE HYPOTHALAMIC-ANTERIOR PITUITARY UNIT It is generally accepted that the annual reproductive cycle in sheep and other seasonal breeders are primarily induced by photoperiodic-mediated Ciechanowska et al 107 changes in the activity of several neuroendocrine systems and their functions in controlling GnRH release from the hypothalamus [82, 110]. It has been proposed that melatonin signals, which encode photoperiods, act within the VM to affect GnRH secretion [115] through several neural pathways including dopaminergic [2, 61, 197, 198], noradrenergic [60, 62, 193], serotoninergic [102], opioidergic [36, 197] and GABA-ergic [169, 193] systems. It has been established that among these transsynaptic pathways regulating GnRH cell activity, the dopaminergic systems in the hypothalamus have a predominant inhibitory effect on GnRH release in the non-breeding season (long-day) but not in the breeding season (short-day; [2, 13, 61, 197]). Numerous studies support the notion that increased dopaminergic system activity in the hypothalamus of anestrous ewes suppresses GnRH release, whereas reduced activity of these systems during short day increases pulsatility in GnRH/LH secretion [63, 197, 198]. Pharmacological blockade of dopaminergic systems with specific dopaminergic antagonists [35, 73, 198] as well as surgical [143, 150] or chemical [188] disruption of dopaminergic pathways between ME and POA resulted in significant increases in GnRH/gonadotrophin secretion in sexually inactive animals during long days. Dopaminergic receptor agonists failing to suppress LH secretion during the non-breeding season in sheep are consistent with the views that inhibitory dopaminergic systems are strongly activated during long-day photoperiod [197, 198]. In contrast, the specific dopaminergic antagonists were ineffective in simulating pulsatile LH secretion when given to breeding ewes, whereas dopaminergic agonists displayed a suppressive effect on gonadotropins release [197, 198]. However, it is becoming obvious that there are a number of dopaminergic pathways which influence gonadotropin by either increasing or decreasing that secretion [9, 81, 88]. These include A12, A13 and A15 (brain dopaminergic nuclei), dopaminergic cell groups with each pathway apparently modulating the LH pulse generator independently [2, 9, 88, 104, 123]. The most likely location of dopaminergic cells that inhibit GnRH pulse frequency during anestrus is the A15 cell group [2]. Lesion studies have documented the participation of A15 dopaminergic neurons in 108 Regulation of GnRH secretion E2-mediated feedback action on GnRH secretion during anestrus [2]. Because dopaminergic neurons do not contain estrogen receptors [103, 177], estrogen sensitive neural afferents to A15 groups cells are likely to project from the ventromedial part of the POA [9] and retrochiasmatic structure [57, 69]. Thus, a neural circuit consisting of estrogen sensitive neurons in these areas which project to inhibitory A15 dopaminergic cells afferent to GnRH neurons mediates estrogen negative action in anestrus. The seasonal changes in morphology and the number of synapses on A15 neurons further support this concept [2]. The dopaminergic terminals which are located in the external layer of the ME establish synaptic contact with GnRH terminals suggesting that dopamine may presynaptically control GnRH release [101]. The morphological and functional observations revealed that the dopaminergic neurons are capable of affecting the GnRH cells indirectly through opioid systems in the hypothalamus [197]. It has been indicated that opiodergic systems in the hypothalamus of anestrous ewes display a minor role in the suppression of GnRH/LH release [77, 190, 197]. Recent studies have demonstrated that dopaminergic neurotransmission through dopaminergic DA-2 receptor affects not only GnRH release but also GnRH and GnRHR gene expression in the hypothalamus of anestrous ewes [28]. Indeed, the blockade of DA-2 receptors by the antagonist, sulpiride, significantly decreased GnRH mRNA level in the ventromedial hypothalamus; such treatment had no evident effect on GnRH gene expression in the preoptic/anterior hypothalamic area. The infusion of sulpiride in the third cerebral ventricle of the brain significantly increased GnRHR mRNA level in the POA/AH areas but decreased them in the VM/SME. All these changes in GnRH mRNA and GnRHR mRNA levels in selected parts of the hypothalamus were associated with an increase in GnRHR gene expression in the anterior pituitary gland and LH secretion. These results strongly suggest that the dopaminergic system acting through DA-2 receptors in the hypothalamus is involved in the regulation of GnRH and GnRHR gene expression in specific coordinated fashion, which leads to changes in the biosynthesis of GnRH and its release in anestrous ewes. It is likely that the decreased GnRH mRNA level in Ciechanowska et al 109 the VM after the blockade of dopaminergic DA-2 receptors is related to increased utilization of GnRH mRNA in biosynthetic processes of GnRH. Such an assumption is supported by increased expression of GnRHR gene in the anterior pituitary gland and LH secretion. A lack of significant changes in the GnRH mRNA level in the POA/AH areas of sulpiride-treated ewes suggests that the blockade of DA-2 receptors did not evidently affect transcriptional activity of the GnRH gene. The blockade of DA-2 receptor gene led to different responses in GnRHR mRNA level in various parts of the hypothalamus: increased GnRHR mRNA in the POA/AH areas and decreased GnRH mRNA content in the VM/SME. The regional specific differences of GnRHR expressing neurons in various parts of the hypothalamus to dopamine may be due to their different response to the blockade of dopaminergic receptors as well as to dopaminergic sensitive interneurons inputs. It could be presumed that the decrease in GnRHR mRNA level in the VM/SME is associated with a decreased biosynthesis of GnRHR protein which, in turn, allows an increase in GnRH release. EFFECT OF GNRH ON THE LEVELS OF GNRH mRNA AND GNRHR mRNA IN THE HYPOTHALAMUS OF ANESTROUS EWES It has been shown that the hypothalamic GnRH, among other neurohormones and/or neurotransmitters, is involved in the control of GnRH release through ultrashort loop feedback mechanism. It has been proven in vivo and in vitro studies that the activation of GnRH receptors in the ventromedial hypothalamus with GnRH exerts an inhibitory effect on GnRH release from the GnRH terminals in rats [40, 167]. In the anestrous ewes, the prolonged intermittent stimulation of GnRHR in the hypothalamus with the infusion of GnRH into the third ventricle increases GnRH mRNA in the VM and GnRHR mRNA in the entire hypothalamus. This suggests that GnRH at the genomic level in the hypothalamus may act on both GnRH and GnRHR synthesis and, consequently, alter GnRH release [112]. 110 Regulation of GnRH secretion REFERENCES 1. Adams BM, Sukarai H, Adams TE 1996 Concentration of gonadotropin-releasing hormone (GnRH) receptor messenger ribonucleic acid in pituitary tissue of orchidectomized sheep; effect of estradiol and GnRH. Biology of Reproduction 54 407412. 2. Adams VL, Goodman RL, Salm AK, Coolen LM, Karsch FJ, Lehman MN 2006 Morphological plasticity in the neural circuitry responsible for seasonal breeding in the ewe. Endocrinology 147 4843-4851. 3. Adler BA, Crowley WR 1986 Evidence for γ-aminobutyric acid modulation of ovarian hormonal effects on luteinizing hormone secretion and hypothalamic catecholamines activity in the female rat. Endocrinology 118 91-97. 4. Akema T, Kimura F 1990 The mode of GABAβ receptor-mediated inhibition of the preovulatory luteinizing hormone surge in female rats. Brain Research 562 169-172. 5. Akema T, Kimura F 1992 Modulation of pulsatile LH secretion by baclofen, a selective GABAβ receptor agonist, in ovariectomized rats. Neuroendocrinology 56 141-147. 6. Akema T, Kimura F 1993 Differential effects of GABAA and GABAβ receptor agonists on NMDA-induced and norepinephrine induced luteinizing hormone release in ovariectomized estrogen-primed rat. Neuroendocrinology 57 28-33. 7. Almeida OFX, Nikolarakis KE, Schulz R, Herz A 1987 A “window of time” during which testosterone determines the opiatergic control of LH release in the adult male rat. Journal of Reproduction and Fertility 39 299-305. 8. Almeida OFX, Nikolarakis KE, Herz A 1988 Evidence for the involvement of endogenous opioids in the inhibition of luteinizing hormone in the rat. Endocrinology 122 1034-1041. 9. Anderson ST, Walsh JP, Tillet Y, Clarke IJ, Curlevis JD 2001 Dopaminergic input to the ventromedial hypothalamus facilitates the estrogen induced luteinizing hormone surge in ewes. Neuroendocrinology 73 91-101. 10. Armstrong DT 1986 Environmental stress and ovarian function. Biology of Reproduction 34 29-34. 11. Attardi B, Klatt B, Hoffman GE, Smith MS 1997 Facilitation or inhibition of the estradiol-induced gonadotropin surge in the immature rat by progesterone regulation of GnRH and LH messenger RNAs and activation of GnRH neurons. Journal of Neuroendocrinology 9 589-600. 12. Bauer-Dantoin AC, Weiss J, Janneson JA 1995 Role of estrogen, progesterone, and gonadotropin-releasing hormone (GnRH) in the control of pituitary GnRH receptor gene expression at the time of the preovulatory gonadotropin surges. Endocrinology 136 1014-1019. 13. Bertrand F, Thiery JC, Picard S, Malpaux B 1999 Implication of DA2-like dopaminergic receptors in the median eminence during the establishment of long-day of LH secretion in the ewe. Journal of Endocrinology 163 243-254. 14. Bicknell RJ 1985 Endogenous opioid peptides and hypothalamic neuroendocrine neurones. Journal of Endocrinology 107 437-446. Ciechanowska et al 111 15. Bonavera JJ, Kalra SP, Kalra PS 1993 Mode of action of interleukin-1 suppression of pituitary LH in castrated male rats. Brain Research 612 1-8. 16. Breen KM, Oakley AE, Pytiak AV, Tilbrook AJ, Wagenmaker ER, Karsch FJ 2007 Does cortisol acting via the type II glucocorticoid receptor mediate suppression of pulsatile luteinizing hormone secretion in response to psychosocial stress? Endocrinology 148 1882-1890. 17. Breen KM, Davis TL, Doro LC, Nett TM, Oakley AE, Padmanabhan V, Rispoli LA, Wagenmaker EW, Karsch FJ 2008 Insight into the neuroendocrine site and cellular mechanism by which cortisol suppresses pituitary responsiveness to gonadotropinreleasing hormone. Endocrinology 149 767-773. 18. Caraty A, Skinner DC 1999 Progesterone priming is essential for the full expression of the positive feedback effect of estradiol in inducing the preovulatory gonadotropinreleasing hormone surge in the ewe. Endocrinology 140 165-170. 19. Caraty A, Miller DW, Delaleu B, Martin B 1997 Stimulation of LH secretion in sheep by central administration of corticotropin-releasing hormone. Journal Reproduction and Fertility 111 249-257. 20. Cates PC, Li XF, O’Byrne KT 2004 The influence of 17β estradiol on corticotropinreleasing hormone (CRF) induced suppression of luteinizing hormone (LH) pulses and the role of CRF in hypoglycaemic stress-induced suppression of pulsatile LH secretion in the female rat. Stress 7 113-118. 21. Cheng CK, Leung PCK 2005 Molecular biology of gonadotropin-releasing hormone (GnRH)-1, GnRH-II, and their receptors in humans. Endocrine Reviews 26 283-306. 22. Cheng KW, Ngan ESW, Kang SK, Chow BKC, Leung PCK 2000 Transcriptional downregulation of human gonadotropin-releasing hormone (GnRH) receptor gene by GnRH; role of protein kinase C and activating protein 1. Endocrinology 141 3611-3622. 23. Cho BN, Seong JY, Cho H, Kim K 1994 Progesterone stimulates GnRH gene expression in the hypothalamus of ovariectomized, estrogen treated adult rats. Brain Research 652 177-180. 24. Chomicka LK 1984 Effect of stress on the activity of the pituitary-gonadal axis and brain serotonin. Acta Physiologica Polonica 35 103-112. 25. Ciechanowska M, Łapot M, Malewski T, Misztal T, Mateusiak K, Przekop F 2007 Effect of stress on the expression of GnRH and GnRH receptor (GnRH-R) genes in the preoptic area-hypothalamus and GnRH-R gene in the stalk/median eminence and anterior pituitary gland in ewes during follicular phase of the estrous cycle. Acta Neurobiologiae Experimentalis 67 1-12. 26. Ciechanowska M, Łapot M, Malewski T, Mateusiak K, Misztal T, Przekop F 2008 Effects of GABAA receptor modulation on the expression of GnRH gene and GnRH receptor (GnRH-R) gene in the hypothalamus and GnRH-R gene in the anterior pituitary gland of follicular phase ewes. Animal Reproduction Science 111 235-248. 27. Ciechanowska M, Łapot M, Malewski T, Mateusiak K, Misztal T, Przekop F 2008 Expression of the GnRH and GnRH receptor (GnRH-R) genes in the hypothalamus and GnRH-R gene in the anterior pituitary gland of anestrous and luteal phase ewes. Animal Reproduction Science 108 345-355. 112 Regulation of GnRH secretion 28. Ciechanowska M, Łapot M, Malewski T, Mateusiak K, Misztal T, Przekop F 2008 Implication of dopaminergic systems on GnRH and GnRH-R genes expression in the hypothalamus and GnRH-R gene expression in the anterior pituitary gland of anestrous ewes. Experimental and Clinical Endocrinology & Diabetes 116 357-362. 29. Ciechanowska M, Łapot M, Malewski T, Mateusiak K, Misztal T, Przekop F 2008 The central effect of β-endorphin and naloxone on the expression of GnRH gene and GnRH receptor (GnRH-R) gene in the hypothalamus, and on GnRH-R gene in the anterior pituitary gland in follicular phase ewes. Experimental and Clinical Endocrinology & Diabetes 116 40-46. 30. Ciechanowska M, Łapot M, Malewski T, Mateusiak K, Misztal T, Przekop F (in press) The effect of CRF and its antagonist on the expression of GnRH gene and GnRH receptor (GnRH-R) gene in the hypothalamus and GnRH-R gene in the anterior pituitary gland of follicular phase ewes. Neuroendocrinology Letters. 31. Clarke IJ, Horton RJE, Doughton BW 1990 Investigation of the mechanism by which insulin induced hypoglycaemia decreases luteinizing hormone secretion in ovariectomized ewes. Endocrinology 127 1470-1476. 32. Collu R, Gibb W, Ducharme JR 1984 Effect of stress on gonadal function. Journal of Endocrinological Investigation 7 529-538. 33. Conover CD, Kuljis RO, Rabii J, Advis JB 1993 β-endorphin regulation of luteinizing hormone-releasing hormone release at the median eminence in ewes: immunocytochemical and physiological evidence. Neuroendocrinology 57 1182-1195. 34. Culliman WE 2000 GABAA receptor subunit expression within hypophysiotropic CRF neurons: a dual hybridization histochemical study. Journal of Comparative Neurology 419 344-351. 35. Curlevis JD, Naylor AM, Mac Neilly AS 1991 Evaluation of a possible role for dopamine D1 and D2 receptors in the steroid-dependent suppression of luteinizing hormone secretion in the seasonally anestrous ewes. Neuroendocrinology 3 387-391. 36. Curlevis JD, Naylor AM, Rhind SM, Mc Neilly AS 1991 Effect of beta-endorphin on pulsatile luteinizing hormone and prolactin secretion during follicular phase in the ewe. Journal of Neuroendocrinology 3 123-126. 37. Currie WD, Evans ACO, Ravindra JP, Rawlings NC 1993 Progesterone blockade of the positive feedback effects of 17-β estradiol in the ewe. Animal Reproduction Science 30 281-287. 38. Dantzer R, Hermide P 1995 Stress in farm animals: need for reevaluation. Journal of Animal Science 57 6-18. 39. De Fazio RA, Heger S, Ojeda SR, Moenter SM 2002 Activation of A-type gamma aminobutyric acid receptors excites gonadotropin-releasing hormone neurons. Molecular Endocrinology 16 2872-2891. 40. De Paolo LV, King RA, Carollo AJ 1987 In vivo and in vitro examination of autoregulatory mechanism for luteinizing hormone-releasing hormone. Endocrinology 120 272-279. 41. Diez-Gurra FJ, Augood S, Emson PC, Dyer RG 1987 Opioid peptides inhibit the release of noradrenaline from slices of rat medial preoptic area. Experimental Brain Research 66 378-384. Ciechanowska et al 113 42. Di Gregorio GB, Nett TM 1995 Estradiol and progesterone influence the synthesis of gonadotropins in the absence of gonadotropin-releasing hormone in the ewe. Biology of Reproduction 53 166-172. 43. Dobson H, Ghuman S, Prabhakat S, Smith R 2003 A conceptual model of the influence of stress on female reproduction. Reproduction 125 151-163. 44. Domański E, Chomicka LK, Ostrowska A, Gajewska A, Mateusiak K 1991 Release of luteinizing hormone-releasing hormone, β-endorphin and norepinephrine by the nucleus infundibularis/median eminence during periovulatory period in the sheep. Neuroendocrinology 54 151-158. 45. Domey JH, Gunn RG, Griffiths JG 1973 The effect of premating stress on the onset of estrus and ovulation rate in Scotish blackface ewes. Journal of Reproduction and Fertility 35 381-384. 46. Donald RA, Redekopp C, Cameron V, Nichols MG, Bolton J, Liversey J, Espiner EA, Rivier J, Vale W 1983 The hormonal actions of corticotropin-releasing factor in sheep: effects of intravenous and intracerebroventricular injection. Endocrinology 113 866-870. 47. Dunn AJ, Berridge CW 1990 Physiological and behavioral responses to corticotropin releasing factor administration; is CRF a mediator of anxiety of stress responses? Brain Reviews 15 71-100. 48. Eckersell CB, Micevych PE 1998 Colocolization of immunoreactivity for mu and delta opioid receptors with cholecystokinin, LHRH and limbic system of female rat. Social Neuroscience 28, 632. 49. Emoto H, Yokoo H, Yoshida M, Tanaka M 1993 Corticotropin-releasing factor enhances noradrenaline release in the rat hypothalamus assessed by intracerebral microdialysis. Brain Research 601 286-288. 50. Evans NP, Dahl GE, Glover BH, Karsch FJ 1994 Central regulation of pulsatile gonadotropin-releasing hormone (GnRH) secretion by estradiol during the period leading up to the preovulatory GnRH surge in the ewe. Endocrinology 134 1806-1811. 51. Evans NP, Dahl GE, Padmanabhan V, Thrun LA 1997 Estradiol requirements for induction and maintenance of the gonadotropin-releasing hormone surge: implication for neuroendocrine processing of the estradiol signal. Endocrinology 138 5408-5414. 52. Faletti AG, Mastronardi CA, Lomniczi A, Seilicovich A, Gimeno H, Mc Cann SM, Rettori V 1999 β-endorphin blocks luteinizing hormone-releasing hormone release by inhibiting the nitricoxidergic pathway controlling its release. Proceedings of the National Academy of Sciences of the United States of America 96 1722-1726. 53. Feleder C, Jarry H, Leonhardt S, Wuttke W, Moguilevsky JA 1996 The gabaergic control of gonadotropin releasing hormone secretion in male rats during sexual maturation involves effects on hypothalamic excitatory and inhibitory amino acids systems. Neuroendocrinology 64 305-312. 54. Ferreira SA, Scott CJ, Kuehl DE, Jackson GL 1995 Differential regulation of luteinizing hormone release by γ-aminobutyric acid receptor subtypes in the arcuate-ventromedial region of the castrated ram. Endocrinology 137 3453-3460. 55. Ferreira SA, Hilman SM, Kuehl DE, Jackson GL 1998 Effect of dialyzing γ-aminobutyric acid antagonists into the medial preoptic area and arcuate ventromedial region on luteinizing hormone release in male sheep. Biology of Reproduction 58 1038-1046. 114 Regulation of GnRH secretion 56. Fueshko SM, Key S, Wray S 1998 Luteinizing hormone-releasing hormone (LHRH) neurons maintained in nasal explants decrease LHRH messenger ribonucleic acid levels after activation of GABAA receptors. Endocrinology 139 2734-2740. 57. Gallegos-Sandez J, Delaleu B, Caraty A, Malpaux B, Thiery JE 1997 Estradiol acts locally within the retrochiasmatic area to inhibit pulsatile luteinizing hormone release in the female sheep during anestrus. Biology of Reproduction 56 1544-1549. 58. Gambacciani M, Yen SSC, Rasmussen DD 1986 GnRH release from the mediobasal hypothalamus: in vitro inhibition by corticotropin-releasing factor. Neuroendocrinology 43 533-536. 59. Gindoff PR, Ferin M 1987 Endogenous opioid peptides modulate the effect of corticotropin-releasing factor on gonadotropin release in primates. Neuroendocrinology 49 202-206. 60. Goodman RL 1996 Neural systems mediating the negative feedback actions of estradiol and progesterone in the ewe. Acta Neurobiologiae Experimentalis 56 727-741. 61. Goodman RL, Inskeep EK 2006 The neuroendocrine control of the ovarian cycle of sheep. In: Neill JD ed. Knobil and Neill’s Physiology and Reproduction 3 rd ed Amsterdam: Elsevier 2006 vol 2 2389-2447. 62. Goodman RL, Robinson JE, Kendrick KM, Dyer RG 1995 Is the inhibitory action of estradiol on luteinizing hormone pulse frequency in anestrous ewes mediated by noradrenergic neurons in the preoptic area? Neuroendocrinology 61 284-292. 63. Goodman RL, Thiery JC, Delaleu B, Malpaux B 2000 Estradiol increases multiunit activity in the A15 area of ewes exposed to inhibitory photoperiod. Biology of Reproduction 63 1352-1357. 64. Gore AC 2002 GnRH; the marker molecule of reproduction. Neuroanatomy of the GnRH-1 system, pp 9-28. Kluver Academic Publishers, Boston/Dordrecht, London. 65. Gruenewald DA, Matsumoto AM 1993 Reduced gonadotropin-releasing hormone gene expression with fasting in the male rat brain. Endocrinology 132 480-482. 66. Guillemin R 2005 Hypothalamic hormones a.k.a. hypothalamic releasing factors. Journal of Endocrinology 184 11-28. 67. Han SK, Abraham IM, Herbison AE 2002 Effect of GABA on GnRH neurons switches from depolarization to hyperpolarization at puberty in female mouse. Endocrinology 143 1459-1466. 68. Han SK, Todman MG, Herbison AE 2004 Endogenous GABA release inhibits the firing of adult gonadotropin-releasing hormone neurons. Endocrinology 145 495-499. 69. Hardy SL, Anderson GM, Valent M, Connors JM, Goodman RL 2003 Evidence that estrogen receptor alfa but not beta, mediates seasonal changes in the response of the ovine retrochiasmatic area to estradiol. Biology of Reproduction 68 846-852. 70. Harris TG, Robinson JE, Evans NP, Skinner DC, Herbison AE 1998 Gonadotropinreleasing hormone messenger ribonucleic acid expression changes before the onset of the estradiol-induced luteinizing hormone surge in the ewe. Endocrinology 139 57-64. 71. Harris TG, Dyer S, Robinson JE, Skinner DC, Evans NP 1999 Progesterone can block transmission of the estradiol-induced signal for luteinizing hormone surge generation during a specific period of time immediately after activation of the gonadotropinreleasing hormone surge-generating system. Endocrinology 140 827-834. Ciechanowska et al 115 72. Hartman RD, He JR, Barraclough CA 1990 γ-aminobutyric acid-A and B receptor antagonists increase luteinizing hormone-releasing hormone neuronal responsiveness to intracerebroventricular norepinephrine in ovariectomized estrogen treated rats. Endocrinology 127 1336-1345. 73. Havern RL, Whisnant CS, Goodman RL 1991 Hypothalamic sites of catecholamine inhibition of luteinizing hormone in the anestrous ewe. Biology of Reproduction 44 476-482. 74. Hayashi KT, Moberg GP 1987 Influence of acute stress and the adrenal axis on regulation of LH and testosterone in the male rhesus monkey (Macaca mulatta). American Journal of Primatology 12 263-273. 75. Herbison AE 1998 Multimodal influence of estrogen upon gonadotropin-releasing hormone neurons. Endocrine Reviews 19 302-330. 76. Herbison AE, Robinson JE, Skinner DC 1993 Distribution of estrogen receptor-immunoreactive cells in the preoptic area of the ewe: colocolization with glutamic acid decarboxylase but not luteinizing hormone releasing hormone. Neuroendocrinology 57 751-759. 77. Horvath TL, Naftolin F, Leranth C 1992 β-endorphin innervation of dopaminergic neurons in the rat hypothalamus: a light and electron microscopic double immunostaining study. Endocrinology 131 1547-1557. 78. Jackson GL, Kuehl DE 2002 The GABA (B) antagonist CGP 52432 attenuates the stimulatory effect of the GABA (B) agonist SKF 97541 on luteinizing hormone secretion in the male sheep. Experimental Biology and Medicine 227 315-320. 79. Jackson GL, Kuehl DE 2004 Effects of applying gamma aminobutyric acids drugs into the medial basal hypothalamus on basal luteinizing hormone concentration and on luteinizing hormone surges in the female sheep. Biology of Reproduction 70 334339. 80. Jackson GL, Wood SG, Kuehl DE 2000 A γ-aminobutyric acid B agonist reverses the negative feedback effect of testosterone on gonadotropin-releasing hormone and luteinizing hormone secretion in the male sheep. Endocrinology 141 3940-3945. 81. Jannes MD, Mac Kenzie FJ, Tuohy-Janes PA, Wilson CA 1987 Dopaminergic neurons in the zona incerta exert a stimulatory control of gonadotropin release via D1-dopamine receptors. Neuroendocrinology 45 348-355. 82. Jansen HT, Cutter C, Hardy S, Lehman MN, Goodman RL 2003 Seasonal plasticity within the gonadotropin-releasing hormone system of the ewe: changes in identified GnRH inputs and glial association. Endocrinology 144 3663-3676. 83. Jarry H, Leonhardt S, Wuttke W 1991 γ-aminobutyric acid neurons in the preoptic/ anterior hypothalamic area synchronize the phasic activity of the gonadotropin-releasing hormone pulse generator in ovariectomized rats. Neuroendocrinology 53 261-267. 84. Jennes L, MC Shane T, Brame B 1996 Dynamic changes in gonadotropin releasing hormone receptor mRNA content in the mediobasal hypothalamus during the rat estrous cycle. Journal of Neuroendocrinology 8 275-281. 85. Jung H, Shannon EM, Fritschy JM, Ojeda SR 1998 Several GABAA receptor subunits are expressed in LHRH neurons of juvenile female rats. Brain Research 780 218-229. 116 Regulation of GnRH secretion 86. Kaiser UB, Conn PM, Chin UW 1997 Studies of gonadotropin-releasing hormone (GnRH) action using GnRH receptor expressing pituitary cell lines. Endocrine Reviews 18 46-70. 87. Kaiser UB, Jakubowiak A, Steinberger A, Chin WW 1997 Differential effect of gonadotropin-releasing hormone (GnRH) pulse frequency on gonadotropin subunit and GnRH receptor messenger ribonucleic acid in vitro. Endocrinology 138 1224-1231. 88. Kalia V, Fenske C, Hole RD, Wilson CA 1999 Effect of gonadal steroid and gammaaminobutyric acid on LH release and dopamine expression and activity in the zona incerta in rats. Journal Reproduction and Fertility 117 189-197. 89. Kang SM, Seong JY, Cho S, Cho H, Kim K 1995 Acute increases of GABAergic neurotransmission exerts a stimulatory effect on GnRH gene expression in the preoptic/ anterior hypothalamus area of ovariectomized, estrogen and progesterone treated adult female rats. Neuroendocrinology 61 486-492. 90. Kang SS, Kim SR, Leonhardt S, Jarry H, Wuttke W, Kim K 2000 Effect of interleukin1β on gonadotropin releasing hormone (GnRH) and GnRH receptor gene expression in castrated male rats. Journal of Neuroendocrinology 12 421-429. 91. Kalra SP, Simpkins JW 1981 Evidence for noradrenergic mediation of opioid effects on luteinizing hormone secretion. Endocrinology 109 776-782. 92. Kalra SP, Kalra PS 1984 Opioid-adrenergic-steroid connection in the regulation of luteinizing hormone secretion in the rat. Neuroendocrinology 38 418-426. 93. Karges B, Karges W, de Roux N 2003 Clinical and molecular genetics of the human GnRH receptor. Human Reproduction Update 9 523-530. 94. Kasa-Vubu JZ, Dahl GE, Evans NP, Moenter SM, Padmanabhan V, Thrun LA, Karsch FJ 1992 Progesterone blocks the estradiol-induced gonadotropin discharge in the ewe by inhibiting the surge of gonadotropin-releasing hormone. Endocrinology 131 208212. 95. Kaupman K, Huggel K, Heid J 1997 Expression cloning of GABAB receptors uncovers similarity to metabotropic glutamate receptors. Nature 386 239-246. 96. Kerdelhue B, Parnet P, Lenoir V, Schihar A, Gaudoux F, Levaseur M, Palkovits M, Blacker C, Scholler R 1988 Interaction between 17-β estradiol and the hypothalamopituitary β-endorphin system in the regulation of the cyclic LH secretion. Journal Steroid Biochemistry 30 1-6. 97. Kim K, Lee BJ, Park J, Cho WK 1989 Progesterone increases messenger ribonucleic acid (mRNA) encoding luteinizing hormone releasing hormone (LHRH) level in the hypothalamus of ovariectomized estradiol-primed prepubertal rats. Molecular Brain Research 6 151-158. 98. Kim K, Lim IS, Cho BN, Kang SS, Lee BJ, Choi KH, Chung CH, Lee CC, Cho WK, Wuttke W 1993 A partial blockade of catecholaminergic neurotransmission with 6-hydroxydopamine decreases mRNA level of gonadotropin releasing hormone in the male rat hypothalamus. Neuroendocrinology 58 146-152. 99. Kim K, Lee BJ, Cho BN, Kang SS, Choi WS, Park SD, Lee CC, Cho WK, Wuttke W 1994 Blockade of noradrenergic neurotransmission with diethylodithiocarbonic acid decreases the mRNA level of gonadotropin-releasing hormone in the hypothalamus of ovariectomized, steroid treated prepubertal rats. Neuroendocrinology 59 539-544. Ciechanowska et al 117 100. Kirkpatrick BL, Esquivel E, Moss GE, Hamernik DL, Wise ME 1998 Estradiol and gonadotropin-releasing hormone (GnRH) interact to increase GnRH receptor expression in ovariectomized ewes after hypothalamic-pituitary disconnection. Endocrine 8 225-229. 101. Kuljis SRO, Advis JP 1989 Immunocytochemical and physiological evidence of a synapse between dopamine- and luteinizing hormone-releasing hormone-containing neurons in the ewe median eminence. Endocrinology 124 1579-1581. 102. Le Corre S, Chemineau P 1993 Control of photoperiodic inhibition of luteinizing hormone secretion by dopaminergic and serotoninergic systems in ovariectomized Ile-de-France ewes supplemented with estradiol. Journal of Reproduction and Fertility 97 367-373. 103. Lehman M, Karsch FJ 1993 Do gonadotropin-releasing hormone, tyrosine hydroxylase-, and β-endorphin-immunoreactive neurons contain estrogen receptors? A doublelabel immunocytochemical study in Soffolk ewe. Endocrinology 133 887-895. 104. Lehman MN, Durham DM, Jansen HT, Adrian B, Goodman RL 1996 Dopaminergic A14/15 neuron are activated during the estradiol negative feedback in anestrous, but not breeding season, ewes. Endocrinology 137 4443-4450. 105. Leonhardt S, Seong JY, Kim K, Thorun J, Wuttke W 1995 Activation of central GABAA but not GABAB receptors rapidly reduces pituitary LH release and GnRH gene expression in the preoptic/anterior hypothalamic area of ovariectomized rats. Neuroendocrinology 61 655-662. 106. Leranth C, MacLusky NJ, Shanabrough M, Naftolin F 1988 Immunocytochemical evidence for synaptic conections between proopiomelanocortin-immunoreactive axons and LHRH neurons is the preoptic area of the rat brain. Brain Research 449 167-176. 107. Li S, Pelletier G 1993 Chronic administration of muscimol and pentobarbital decreases gonadotropin-releasing hormone mRNA levels in the male rat hypothalamus determined by quantitative in situ hybridization. Neuroendocrinology 58 136-139. 108. Liaw J, Barraclough CA 1993 N-methyl-D,L-aspartic acid differentially affects LH release and LHRH mRNA levels in estrogen-treated ovariectomized control and androgen-sterilized rats. Molecular Brain Research 17 112-118. 109. Lin X, Conn M 1999 Transcriptional activation of gonadotropin-releasing hormone (GnRH) receptor gene by GnRH: involvement of multiple signals transduction pathways. Endocrinology 140 358-364. 110. Lincoln GA 2002 Neuroendocrine regulation of seasonal gonadotrophin and prolactin rhythms: lesson from the Soay ram model. Reproduction. Supplement 59 131-141. 111. Łapot M, Ciechanowska M, Malewski T, Misztal T, Mateusiak K, Przekop F 2007 The effect of stress on the expression of GnRH and GnRH receptor genes in the discrete regions of the hypothalamus and pituitary of anestrous ewes. Reproductive Biology 7 55-71. 112. Łapot M, Ciechanowska M, Malewski T, Mateusiak K, Misztal T, Przekop F 2008 Changes in the GnRH mRNA and GnRH receptor (GnRH-R) mRNA levels in the hypothalamic-anterior pituitary unit of anestrous ewes after infusion of GnRH into the third cerebral ventricle. Reproductive Biology 8 149-161. 118 Regulation of GnRH secretion 113. Mac Lusky NJ, Naftolin F, Leranth C 1988 Immunocytochemical evidence for direct synaptic connections between corticotropin releasing factor (CRF) and gonadotropinreleasing hormone (GnRH) – containing neurons in the preoptic area of the rat. Brain Research 439 391-395. 114. Maeda A, Cagampang FRA, Coen CW, Tsukamura H 1994 Involvement of the catecholaminergic input to the paraventricular nucleus and corticotropin-releasing hormone release in female rats. Endocrinology 134 1718-1722. 115. Malpaux B, Daveau A, Maurice-Mandon F, Duarte G, Chemineau P 1995 Evidence that melatonin acts in the premammilary hypothalamic area to control reproduction in the ewe: presence of binding sites and stimulation of luteinizing hormone by in situ microimplant delivery. Endocrinology 139 1508-1516. 116. Masoto C, Negro-Vilar A 1987 Activation of gamma-aminobutyric acid B-receptors abolishes naloxone-induced luteinizing hormone release. Endocrinology 121 22512255. 117. Millar RP 2005 GnRHs and GnRH receptors. Animal Reproduction Science 88 5-28. 118. Millar RP, Lu Z-L, Pawson AJ, Flanagan CA, Morgan K, Maudsley SR 2004 Gonadotropin-releasing hormone receptors. Endocrine Reviews 25 235-275. 119. Mitchell JC, Li XF, Breen L, Thalabard JC, O’Byrne KT 2005 The role of the locus coeruleus in corticotropin-releasing hormone and stress-induced suppression of pulsatile luteinizing hormone secretion in the female rat. Endocrinology 146 323-331. 120. Mitsushima D, Hei DL, Tarasawa E 1994 γ-aminobutyric acid is an inhibitory neurotransmitter restricting the release of luteinizing hormone-releasing hormone before the onset of puberty. Proceedings of the National Academy of Sciences of the United States of America 91 395-399. 121. Moberg PG 1976 Effect of environmental and menagement stress on the reproduction in the dairy cow. Journal of Dairy Science 59 1618-1624. 122. Moenter SM, De Fazio RA 2005 Endogenous γ-aminobutyric acid can excite gonadotropin-releasing hormone neurons. Endocrinology 146 5374-5379. 123. Moore KL, Lookingland KJ 1995 Dopaminergic neural systems in the hypothalamus. Bloom FE and Kupfer DJ editors. Psychopharmacology: The fourth generation of progress (Chapter 22) New York: Raven Press, Ltd 245-256. 124. Moragues N, Ciofi P, Lafon P, Tramu G, Gorret M 2003 GABA-A receptor epsilon subunit expression in identified peptidergic neurons of hypothalamus. Brain Research 967 285-289. 125. Morgan K, Sellar R, Pawson AJ, Lu Z-L, Millar RP 2006 Bovine and ovine gonadotropin-releasing hormone (GnRH) – II ligand precursors and type II GnRH receptor genes are functionally inactivated. Endocrinology 147 5041-5051. 126. Moss GE, Nett TM 1980 GnRH interaction with anterior pituitary gland IV. Effect of estradiol-17β on GnRH-mediated release of LH from ovine pituitary cells obtained during the breeding season, anestrus, and period of transition into or out of the breeding season. Biology of Reproduction 23 398-403. 127. Mott DD, Lewia DW 1994 The pharmacology and function of central GABAB receptors. International Review of Neurobiology 36 97-223. Ciechanowska et al 119 128. Naftolin F, Mor G, Horvath TL, Luquin S, Fajer AR, Kohen F, Garcia-Segura LM 1996 Synaptic remodeling in the arcuate nucleus during the estrous cycle is induced by estrogen and precedes the preovulatory gonadotropin surge. Endocrinology 137 3576-3580. 129. Nappi RE, Rivest S 1997 Effect of immune and metabolic challenges on the luteinizing hormone-releasing hormone neural system in cycling female rats: an evaluation at the transcriptional level. Endocrinology 138 1374-1384. 130. Nikolarakis KE, Almeida OFX, Herz A 1986 Corticotropin releasing factor (CRF) inhibits gonadotropin-releasing hormone (GnRH) release from superfused rat hypothalami in vitro. Brain Research 377 388-390. 131. Nikolarakis KE, Almeida OFX, Herz A 1986 Stimulation of hypothalamic β-endorphin and dynorphin release by corticotropin-releasing factor (in vitro). Brain Research 399 152-155. 132. Nikolarakis KE, Almeida OFX, Herz A 1988 Hypothalamic opioid receptors mediate the inhibitory actions of corticotropin-releasing hormone on luteinizing hormone release: further evidence from a morphin tolerant animal model. Brain Research 450 360-373. 133. Nikolarakis KE, Almeida OFX, Sirinathsighji DJS, Hertz A 1988 Concomitant changes in the in vitro and in vivo release of opioid peptides and luteinizing hormone-releasing hormone from the hypothalamus following blockade of receptors for corticotropinreleasing factor. Neuroendocrinology 47 545-550. 134. Norman RL 1994 Corticotropin-releasing hormone effects on luteinizing hormone and cortisol secretion in intact female rhesus macaques. Biology of Reproduction 50 949-955. 135. Oakley AE, Breen KM, Clarke IJ, Karsch FJ, Wagenmaker ER, Tilbrook AJ 2009 Cortisol reduces GnRH pulse frequency in follicular phase ewes: influence of ovarian steroids. Endocrinology 150 341-349. 136. Oakley AE, Breen KM, Tilbrook AJ, Wagenmaker ER, Karsch FJ 2009 Role of estradiol in cortisol-induced reduction of luteinizing hormone pulse frequency. Endocrinology 150 2775-2782. 137. Ojeda SR, Negro-Vilar A, McCann SM 1979 Release of prostaglandin Es by hypothalamic tissue: evidence for their involvement in catecholamine – induced luteinizing hormone-releasing hormone release. Endocrinology 104 617-620. 138. Ojeda SR, Negro Vilar A, McCann SM 1982 Evidence for involvement of α-adrenergic receptors in norepinephrine – induced prostaglandin E2 and luteinizing hormonereleasing hormone release from the median eminence. Endocrinology 110 409-411. 139. Olster DH, Ferin M 1987 Corticotropin releasing hormone inhibits gonadotropin secretion in ovariectomized rhesus monkey. Journal Clinical Endocrinology and Metabolism 65 262-267. 140. Pacak K, Palkovits M 2001 Stressor specificity of central neuroendocrine responses: implication for stress-related disorders. Endocrine Reviews 22 502-548. 141. Pape JR, Skynner MJ, Herbison AE 2001 Profiling γ-aminobutyric acid (GABAA) receptor subunit mRNA expression in postnatal gonadotropin-releasing hormone (GnRH) neurons of the male mouse with single cell RT-PCR. Neuroendocrinology 74 300-308. 120 Regulation of GnRH secretion 142. Parrott RF, Robinson JE, Thornton SN 1988 No change in LH pulse frequency or plasma oxytocin in castrated rams treated with corticotropin-releasing hormone (CRF). Journal of Endocrinology Supplement 119 p 130. 143. Pau K, Kuehl DE, Jackson GL 1982 Effect of frontal hypothalamic deafferentation on luteinizing hormone secretion and seasonal breeding in the ewe. Biology of Reproduction 27 999-1009. 144. Pawson AJ, Morgan K, Maudsley SR, Millar RP 2003 Type II gonadotropin releasing hormone (GnRH-II) in reproductive biology. Reproduction 126 271-278. 145. Petersen JE, Mc Crone S, Coy D, Adelman JP, Malven LC 1993 GABAA receptor subunit mRNA in cells of preoptic area: colocalization with LHRH mRNA using dual-label in situ hybridization histochemistry. Endocrine Journal 1 1-29. 146. Petersen SL, Mc Crone S, Keller M, Shores S 1995 Effects of estrogen and progesterone on luteinizing hormone-releasing hormone messenger ribonucleic acid levels: consideration of temporal and neuroanatomical variables. Endocrinology 136 3604-3610. 147. Phogat PB, Smith RF, Dobson H 1997 The influence of stress on neuroendocrine control of the hypothalamic-pituitary-ovarian axis. Veterinary Bulletin 67 553-567. 148. Polkowska J, Przekop F 1997 The effect of corticotropin-releasing factor (CRF) on the gonadotropin hormone-releasing hormone (GnRH) hypothalamic neuronal system during preovulatory period in the ewe. Acta Neurobiologiae Experimentalis 57 91-99. 149. Price ML, Curtis AL, Kirby LG, Valentino RJ, Lucki I 1998 Effect of corticotropinreleasing factor on brain serotoninergic activity. Neuropsychopharmacology 18 492-502. 150. Przekop F 1978 Effect of anterior deafferentation of the hypothalamus on the release of luteinizing hormone (LH) and reproductive function in sheep. Acta Physiologica Polonica 29 393-407. 151. Przekop F, Tomaszewska D 1996 Responses in hypothalamic monoaminergic systems activity in the ewes to β-endorphin, CRF and their antagonists. Acta Neurobiologiae Experimentalis 56 807-817. 152. Przekop F, Wolińska-Witort E, Mateusiak K, Sadowski B, Domański E 1984 The effect of prolonged stress on the estrous cycles and prolactin secretion in sheep. Animal Reproduction Science 7 333-342. 153. Rabow LE, Russek SJ, Farb DH 1995 From ion currents to genomic analysis: recent advances in GABAA receptors research. Synapse 21 189-274. 154. Rasmussen DD, Malven PV 1983 Effect of confinement stress on episodic secretion of LH in ovariectomized sheep. Neuroendocrinology 36 393-396. 155. Rettori V, Gimeno M, Lyson K, McCann SM 1992 Nitric oxide mediates norepinephrine -induced prostaglandin E2 release from the hypothalamus. Proceedings of the National Academy of the Sciences of United States of America 89 11543-11546. 156. Richter TA, Robinson JE, Evans NP 2001 Progesterone treatment that either blocks or augments the estradiol-induced gonadotropin-releasing hormone surge is associated with different patterns of hypothalamic neural activation. Neuroendocrinology 73 378-386. Ciechanowska et al 121 157. Rivest S, Rivier C 1993 Interleukin-1β inhibits the endogenous expression of the early gene C-fos located within the nucleus of LHRH neurons and interferes with hypothalamic LHRH release during proestrus in the rat. Brain Research 613 132-142. 158. Rivest S, Rivier C 1995 The role of corticotropin-releasing factor and interleukin-1 in the regulation of neurons controlling reproductive functions. Endocrine Reviews 16 177-199. 159. Rivier C, Rivest S 1991 Effect of stress on the activity of hypothalamic-pituitary-gonadal axis: peripheral and central mechanisms. Biology of Reproduction 45 523-532. 160. Rivier C, Rivier J, Vale W 1986 Stress-induced inhibition of the reproductive functions: role of endogenous corticotropin-releasing factor. Science 231 607-609. 161. Roberts JL, Dublow CM, Jakubowski M, Blum M, Millar RP 1989 Estradiol stimulates preoptica area-anterior hypothalamic pro GnRH-GAP gene expression in ovariectomized rats. Molecular Brain Research 6 127-134. 162. Robinson JE, Kendrick KM, Lambart CE 1991 Changes in the release of gammaaminobutyric acid and catecholamines in the preoptic/septal area prior to and during the preovulatory surge of luteinizing hormone in ewe. Journal of Reproduction 3 393-399. 163. Robinson JE, Healey AE, Harris TG, Messent EA, Skinner DC, Taylor JA, Evans NP 2000 The negative feedback action of progesterone on luteinizing hormone release is not associated with changes in GnRH mRNA expression in the ewe. Journal of Neuroendocrinology 12 121-129. 164. Rosie R, Thompson E, Fink G 1990 Oestrogen positive feedback stimulates the synthesis of LHRH mRNA in neurons of the rostral diencephalon of the rat. Journal of Endocrinology 124 285-289. 165. Roth C, Jung K, Kim K, Arias P, Moguilevsky J, Jarry H, Leonhardt S, Wuttke W 1997 Involvement of gamma amino butyric acid (GABA) in the postnatal function of the GnRH pulse generator as gene expression in the hypothalamus and pituitary. Experimental and Clinical Endocrinology & Diabetes 105 353-358. 166. Sakakibara H, Taga M, Iheda M, Kurogi K, Minaguchi H 1996 Continuous stimulation of gonadotropin-releasing hormone (GnRH) receptors by GnRH agonist decreases pituitary GnRH receptor messenger ribonucleic acid concentration in immature female rat. Endocrine Journal 43 115-118. 167. Sarkar DK 1987 In vitro secretion of LHRH in ovariectomized rats is regulated by a possible autofeedback mechanism. Neuroendocrinology 45 510-513. 168. Sarkar DK, Yen SSC 1985 Changes in β-endorphin-like immunoreactivity in pituitary portal blood during the estrous cycle and after ovariectomy in rats. Endocrinology 116 2075-2079. 169. Scott CJ, Clarke IJ 1993 Inhibition of luteinizing hormone secretion in ovariectomized ewes during the breeding season by γ-aminobutyric acid (GABA) is mediated by GABA-A receptors, but not GABA-Β receptors. Endocrinology 132 1789-1796. 170. Scott CJ, Clarke IJ 1993 Evidence that changes in the function of the subtypes of the receptors for γ-aminobutyric acid may be involved in the seasonal changes in the negative-feedback effects of estrogen on gonadotropin-releasing hormone secretion and plasma luteinizing hormone levels in the ewe. Endocrinology 133 2904-2912. 122 Regulation of GnRH secretion 171. Scott CJ, Rawson JA, Pereira AM, Clarke IJ 1999 Oestrogen receptors in the brain stem of the female sheep: relationship to noradrenergic cells and cells projecting to the medial preoptic area. Journal of Neuroendocrinology 11 745-756. 172. Sealfon SC, Weinstein H, Millar RP 1997 Molecular mechanisms of ligand interaction with the gonadotropin-releasing hormone receptor. Endocrine Reviews 18 180-205. 173. Seong JY, Jarry H, Kühnemuth S, Leonhardt S, Wuttke W, Kim K 1995 Effect of GABA-ergic compounds on gonadotropin-releasing hormone receptor gene expression in the rat. Endocrinology 136 2587-2593. 174. Seong JY, Kang SS, Kam K, Han Y-G, Kwon HB, Ryu K, Kim K 1998 Different regulation of gonadotropin-releasing hormone (GnRH) receptor expression in the posterior mediobasal hypothalamus by steroid hormones: implication of GnRH neural activity. Molecular Brain Research 53 226-235. 175. Sieghart W 1995 Structure and pharmacology of gamma-aminobutyric acid A receptor subtypes. Pharmacological Reviews 47 181-234. 176. Silverman A-J, Livne I, Witkin JW 1994 The gonadotropin-releasing hormone (GnRH), neuronal systems: immunocytochemistry and in situ hybridization. The Physiology of Reproduction Second Edition (Chapter 28), pp 1683-1709. Eds Knobil E and Neill JD, Raven Press Ltd, New York. 177. Skinner DC, Herbison AE 1997 Effects of photoperiod on estrogen receptors, tyrosine hydroxylase (TH), neuropeptide Y and β-endorphin immunoreactivity in the ewe hypothalamus. Endocrinology 138 2585-2595. 178. Sliwowska JH, Billingo HJ, Goodman RL, Lehman MN 2006 Immunocytochemical colocalization of GABA-Β receptor subunit in gonadotropin-releasing hormone neurons of the sheep. Neuroscience 141 311-319. 179. Smith MJ, Jennes L 2001 Neural signals that regulate GnRH neurons directly during the estrous cycle. Reproduction 122 1-10. 180. Stackpole CA, Clarke IJ, Breen KM, Turner AI, Karsch FJ, Tilbrook AJ 2006 Sex differences in the suppressive effect of cortisol on pulsatile secretion in luteinizing hormone in sheep. Endocrinology 147 5921-5931. 181. Stoebel DP, Moberg PG 1982 Repeated acute stress during follicular phase and luteinizing hormone surge of dairy heifer. Journal Dairy Science 65 92-96. 182. Stojikovic SS, Reinhart J, Catt KJ 1994 Gonadotropin-releasing hormone receptors: structure and signal transduction pathways. Endocrine Reviews 15 462-499. 183. Sullivan SD, Moenter SM 2004 γ-aminobutyricacid neurons integrate and rapidly transmit permissive and inhibitory metabolic cuts to gonadotropin-releasing hormone neurons. Endocrinology 145 1194-1202. 184. Sullivan SD, Moenter SM 2005 GABA-ergic integration of progesterone and androgen feedback to gonadotropin-releasing hormone neurons. Biology of Reproduction 72 33-41. 185. Tanebe K, Nishijo A, Muraguchi A, Ono T 2000 Effects of chronic stress on hypothalamic interleukin-1β, interleukin-2, and gonadotropin-releasing hormone gene expression in ovariectomized rats. Journal of Neuroendocrinology 12 13-21. 186. Tellam DJ, Perone MJ, Dunn IC, Radovick S, Brennand JE, Rivier JE, Castro MG, Lovejoj DA 1998 Direct regulation of GnRH transcription by CRF-like peptides in an immortalized neuronal cell line. Neuro Report 9 3135-3140. Ciechanowska et al 123 187. Tellam DJ, Mohammad YN, Lovejoj DA 2000 Molecular integration of hypothalamopituitary-adrenal axis related neurohormones on the GnRH neuron. Biochemistry and Cell Biology 78 205-216. 188. Thiery J-C, Martin GB, Tillet Y, Caldani M, Quentin M, Jamain C, Revault JP 1989 Role of hypothalamic catecholamines in the regulation of luteinizing hormone and prolactin secretion in the ewe during seasonal anestrus. Neuroendocrinology 49 80-87. 189. Thind KK, Goldsmith PC 1988 Infundibular gonadotropin releasing hormone neurons are inhibited by direct opioid and autoregulatory synapses in juvenile monkeys. Neuroendocrinology 47 203-216. 190. Tomaszewska D, Przekop F 1999 Catecholaminergic activity in the medial preoptic area and nucleus infundibularis-median eminence of anestrus ewes in normal physiological state and under stress condition. Journal of Neural Transmission 106 1031-1043. 191. Tomaszewska D, Mateusiak K, Przekop F 1997 Extracellular monoamines and their metabolites in the mediobasal hypothalamus-median eminence of anestrous ewes during CRF treatment. Experimental and Clinical Endocrinology & Diabetes 105 175-181. 192. Tomaszewska D, Mateusiak K, Przekop F 1998 Extracellular concentration of monoamines and their metabolites in the mediobasal hypothalamus-median eminence of ewes during β-endorphin treatment. Neuroendocrinology Letters 19 43-53. 193. Tomaszewska-Zaremba D, Przekop F 2005 Effects of GABAB receptor modulation on gonadotropin-releasing hormone and β-endorphin release, and on catecholaminergic activity in the ventromedial hypothalamus-infundibular nucleus region of anestrous ewes. Journal of Neuroendocrinology 17 44-56. 194. Tomaszewska-Zaremba D, Mateusiak K, Przekop F 2003 The role of GABAA receptors in the neural systems of the medial preoptic area in the control of GnRH release in ewes during follicular phase. Animal Reproduction Science 77 71-81. 195. Tomaszewska-Zaremba D, Mateusiak K, Przekop F 2003 The role of GABAA receptors in the neural systems of the ventromedial hypothalamus-nucleus infundibularis region in the control of GnRH release in ewes during follicular phase. Experimental and Clinical Endocrinology & Diabetes 111 335-340. 196. Toranzo D, Dupont E, Simard J, Labrie C, Couet J, Labrie F, Pelletier G 1989 Regulation of pro-gonadotropin-releasing hormone gene expression by sex steroids in the brain of male and female rats. Molecular Endocrinology 3 1748-1756. 197. Tortonese DJ 1999 Interaction between hypothalamic dopaminergic and opioidergic systems in the photoperiodic regulation of pulsatile luteinizing hormone secretion in sheep. Endocrinology 140 750-757. 198. Tortonese DJ, Lincoln GA 1994 Photoperiodic modulation of the dopaminergic control of pulsatile LH secretion in sheep. Journal of Endocrinology 143 25-32. 199. Turzillo AM, Juengel JL, Nett TM 1995 Pulsatile gonadotropin-releasing hormone (GnRH) increases concentrations of GnRH receptor messenger ribonucleic acid and numbers of GnRH receptors during luteolysis in the ewe. Biology of Reproduction 53 418-423. 200. Turzillo AM, Nolan TE, Nett TM 1998 Regulation of luteinizing hormone-releasing hormone (GnRH) receptor gene expression in sheep: interaction of GnRH and estradiol. Endocrinology 139 4890-4994. 124 Regulation of GnRH secretion 201. Ubuka T, Kim S, Huang Y-C, Reid J, Jiang R, Osugi T, Chowdhury VS, Tsutsui K, Bentley GE 2008 Gonadotropin-inhibitory neurons interact directly with gonadotropin releasing hormone-I and -II neurons in European stowling brain. Endocrinology 149 268-278. 202. Van Bockstaelc EJ, Colago EE, Valentino RJ 1996 Corticotropin-releasing factorcontaining axon terminals synapse onto catecholamines dendrites and may presynaptically modulate other afferents in the rostral pole of the nucleus locus coeruleus in the rat brain. Journal of Comparative Neurology 419 344-351. 203. Wagenmaker ER, Breen KM, Oakley AE, Tilbrook AJ, Karsch FJ 2009 Psychosocial stress inhibits amplitude of gonadotropin-releasing hormone pulses. Independent of cortisol action on the type II glucocorticoid receptor. Endocrinology 150 762-769. 204. Walker CD, Dallman MF, Palmer AA, Steele MK 1993 Involvement of central corticotropin-releasing factor (CRF) in suckling-induced inhibition of luteinizing hormone secretion in lactating rats. Journal of Neuroendocrinology 5 451-459. 205. Weesner GD, Harms PG, McArthur NH, Wilson JM, Forrest DW, Wu TJ, Pfaff DW 1993 Luteinizing hormone-releasing hormone gene expression in the bovine brain: anatomical localization and regulation by ovarian state. Biology of Reproduction 49 431-436. 206. Whisnant CS, Curto K, Goodman RL 1992 Immunocytochemical localization of beta endorphin and gonadal steroid regulation of proopiomelanocortin messenger ribonucleic acid in the ewe. Neuroendocrinology 56 812-821. 207. Wizcarra JA, Wattermann RP, Bruden VD, Turzillo AM, Nett TM 1997 Effect of gonadotropin-releasing hormone (GnRH) pulse frequency on serum and pituitary concentrations of luteinizing hormone and follicle stimulating hormone, GnRH receptor and messenger ribonucleic acid for gonadotropin subunits in cows. Endocrinology 138 594-601. 208. Yasin M, Dalkin AC, Haisenleder DJ, Kerrigan JR, Marshall JC 1995 Gonadotropinreleasing hormone (GnRH) pulse pattern regulates GnRH receptor gene expression: augmentation by estradiol. Endocrinology 136 1559-1564.