* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Phenol - Macmillan Academy
Woodward–Hoffmann rules wikipedia , lookup
Homoaromaticity wikipedia , lookup
Hydroformylation wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Marcus theory wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Macrocyclic stereocontrol wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
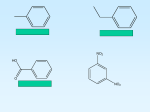
Phenol Draw the following structures Phenol 2. 2-ethylphenol 3. 3,4,4-trichlorobiphenyl 4. 2-phenylethanol Extension: Why is no. 4 not a phenol? 1. Learning Outcomes Explain what PCBs are Describe the reactions of phenol Discuss the uses of phenols PCB’s (Polychlorinated biphenyls) 3,4,4-trichlorobiphenyl Learning Outcomes Explain what PCBs are Describe the reactions of phenol Discuss the uses of phenols Diploma Phenol Phenol cannot be made directly from benzene, the benzene ring is inert to attack by the nucleophile OH-. CH(CH3)2 AlCl3 +CH3CH=CH2 heat benzene propene OH i)O2 +CH3COCH3 ii)H+ (propan-2-yl) benzene ‘cumene’ This is called the ‘cumene’ process phenol propanone PHENOL Structure phenol is an aromatic alcohol with formula C6H5OH the OH group is attached directly to the benzene ring it is an almost colourless crystalline solid Preparation You cannot put an OH group directly onto a benzene ring by electrophilic substitution, so phenol has to be synthesised in a multi-stage process 1 Step 1 - Nitration of benzene reagents conc. nitric acid and conc. sulphuric acid (catalyst) conditions reflux at 55°C equation C6H6 + HNO3 ——> C6H5NO2 + H2O mechanism electrophilic substitution PHENOL Structure phenol is an aromatic alcohol with formula C6H5OH the OH group is attached directly to the benzene ring it is an almost colourless crystalline solid Preparation You cannot put an OH group directly onto a benzene ring by electrophilic substitution, so phenol has to be synthesised in a multi-stage process 2 Step 2 - Reduction of nitrobenzene reagents tin and conc. hydrochloric acid conditions reflux equation C6H5NO2 + 6 [H] ——> C6H5NH2 mechanism reduction + 2H2O PHENOL Structure phenol is an aromatic alcohol with formula C6H5OH the OH group is attached directly to the benzene ring it is an almost colourless crystalline solid Preparation You cannot put an OH group directly onto a benzene ring by electrophilic substitution, so phenol has to be synthesised in a multi-stage process 3 Step 3 - Diazotisation of phenylamine reagents nitrous acid and hydrochloric acid (use sodium nitrite) conditions keep below 10°C equation C6H5NH2 + HNO2 + HCl ——> C6H5N2+ Cl¯ + 2H2O reaction type diazotisation PHENOL Structure phenol is an aromatic alcohol with formula C6H5OH the OH group is attached directly to the benzene ring it is an almost colourless crystalline solid Preparation You cannot put an OH group directly onto a benzene ring by electrophilic substitution, so phenol has to be synthesised in a multi-stage process 4 Step 4 - Substitution of diazo group by OH reagents water conditions warm above 10°C equation C6H5N2+ Cl¯ + H2O ——> C6H5OH reaction type hydrolysis / substitution + N2 + HCl PHENOL - REACTIONS OF THE OH GROUP Water phenol is a weak acid it dissolves very slightly in water to form a weak acidic solution it is a stronger acid than aliphatic alcohols the ring helps weaken the O-H bond and stabilises the resulting anion C6H5OH(aq) NaOH C6H5O¯(aq) + H+(aq) phenol reacts with sodium hydroxide to form a salt - sodium phenoxide it is ionic and water soluble C6H5OH(aq) + NaOH(aq) ——> C6H5O¯ Na+(aq) + H2O(l) Sodium phenol reacts with sodium to form an ionic salt - sodium phenoxide hydrogen is also produced this reaction is similar to that with aliphatic alcohols such as ethanol 2C6H5OH(s) + 2Na(s) ——> 2C6H5O¯ Na+(s) + H2(g) PHENOL - REACTIONS OF THE AROMATIC RING ELECTROPHILIC SUBSTITUTION Ease the OH group is electron releasing it increases the electron density of the delocalised system it makes substitution much easier compared to benzene a p orbital on the oxygen overlaps with the p orbitals in benzene p orbitals in the system the p orbital on the O overlaps with the p orbitals in the ring PHENOL - REACTIONS OF THE AROMATIC RING ELECTROPHILIC SUBSTITUTION Bromine the OH group is electron releasing it increases the electron density of the delocalised system it makes substitution much easier compared to benzene the electron density is greatest at the 2,4 and 6 positions substitution takes place at the 2,4 and 6 positions phenol reacts readily with bromine water WITHOUT A CATALYST it is so easy that multiple substitution takes place other electrophiles such as NO2+ react in a similar way The OH group is electron releasing Electron pair donation takes place from a p orbital on oxygen It increases the electron density of the delocalised system It makes substitution much easier compared to benzene The electron density is the greatest at the 2,4 and 6 positions Substitution takes place at the 2,4 and 6 positions Phenol reacts readily with bromine water without a catalyst It is so easy that multiple substitution takes place. Other electrophiles such as NO2+ react in a similar way Learning Outcomes Explain what PCBs are Describe the reactions of phenol Discuss the uses of phenols Summary sheet Phenol summary sheet.doc Exam questions benzene and phenols.rtf