* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Atomic Number
Survey
Document related concepts
Transcript
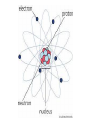
Protons and neutrons are responsible for most of the atomic mass of an atom, while electrons contribute a very small amount of mass(9.108 X 10-28 grams). The number of _________ in the nucleus of an atom determines an element's atomic number. Atomic number. Atomic symbol 6 Element name C Carbon 12.011 Average atomic mass *To find the number of protons, see the atomic number. *To find the number of electrons (see the atomic number, adjust for charge if there is one) *To find the number of neutrons, subtract the atomic number from the atomic mass. Use whole number. Atomic mass -- 12 Atomic # -6 6 neutrons An atom that changes the number of electrons is called an __________. The number of ________ and ________ stays the same. No change in atomic number or ____________ ______________. If the atom loses electrons, the atom becomes ____________ charged. If an atom gains electrons, the atom becomes ____________ charged. (4 +) + (3-) = 1+ (4 +) + (5-) = 1- Changing the number of neutrons in an atom makes an ________________. Have the same _______ _____________ Have different mass numbers (number of __________ + ___________) Changing the number of protons causes ________________- changing from one element to another. The _________________and __________________ changes What is the atom’s net charge? What is the atomic number of the atom? What is the mass number? This is an atom of what element? What is the atom’s net charge? What is the atomic number of the atom? What is the mass number? This is an atom of what element? 79. Democritus 80. John Dalton 81. Atomic theory 82. J.J. Thomason 83. Ernest Rutherford 84. Niels Bohr 85.James Chadwick 86. Electron clouds 87. Nucleus 88. Protons 89. Neutrons 90. Electrons 91. Atomic mass 92. Atomic number 93. Ion 94. Isotope 95. Transmutation