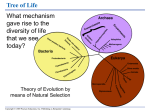
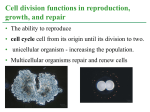
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Biological energy
Microbial metabolism wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthesis wikipedia , lookup
Biochemistry wikipedia , lookup
Citric acid cycle wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
An Introduction to Metabolism ENERGY AND ENERGY TRANSFER Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Overview • Metabolism • Exothermic/Endothermic reactions • ATP • Energy pyramids and ecosystems Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Overview: The Energy of Life • The living cell – Is a miniature factory where thousands of reactions occur – Converts energy in many ways Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Metabolism – Is the totality of an organism’s chemical reactions – Arises from interactions between molecules Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Organization of the Chemistry of Life into Metabolic Pathways • A metabolic pathway has many steps – That begin with a specific molecule and end with a product – That are each catalyzed by a specific enzyme Enzyme 1 A Enzyme 2 D C B Reaction 1 Enzyme 3 Reaction 2 Starting molecule Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Reaction 3 Product • Catabolic pathways – Break down complex molecules into simpler compounds – Release energy • Anabolic pathways – Build complicated molecules from simpler ones – Consume energy Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Thermodynamics – Is the study of energy transformations An organism’s metabolism transforms matter and energy, subject to the laws of thermodynamics Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings The First and Second Laws of Thermodynamics • According to the first law of thermodynamics – Energy can be transferred and transformed – Energy cannot be created or destroyed • According to the second law of thermodynamics – Spontaneous changes that do not require outside energy increase the entropy, or disorder, of the universe Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Concept 8.2: The free-energy change of a reaction tells us whether the reaction occurs spontaneously Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Free-Energy Change, G • A living system’s free energy – Is energy that can do work under cellular conditions • The change in free energy, ∆G during a biological process – Is related directly to the enthalpy change (∆H) and the change in entropy – ∆G = ∆H – T∆S Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Free Energy, Stability, and Equilibrium • Organisms live at the expense of free energy • During a spontaneous change – Free energy decreases and the stability of a system increases Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Exergonic and Endergonic Reactions in Metabolism • An exergonic reaction – Proceeds with a net release of free energy and is spontaneous Reactants Free energy Amount of energy released (∆G <0) Energy Products Progress of the reaction Figure 8.6 (a) Exergonic reaction: energy released Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • An endergonic reaction – Is one that absorbs free energy from its surroundings and is nonspontaneous Free energy Products Energy Reactants Progress of the reaction Figure 8.6 (b) Endergonic reaction: energy required Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Amount of energy released (∆G>0) Equilibrium and Metabolism • Reactions in a closed system – Eventually reach equilibrium ∆G < 0 Figure 8.7 A ∆G = 0 (a) A closed hydroelectric system. Water flowing downhill turns a turbine that drives a generator providing electricity to a light bulb, but only until the system reaches equilibrium. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Cells in our body – Experience a constant flow of materials in and out, preventing metabolic pathways from reaching equilibrium (b) An open hydroelectric system. Flowing water keeps driving the generator because intake and outflow of water keep the system from reaching equlibrium. Figure 8.7 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings ∆G < 0 • An analogy for cellular respiration ∆G < 0 ∆G < 0 ∆G < 0 Figure 8.7 (c) A multistep open hydroelectric system. Cellular respiration is analogous to this system: Glucoce is brocken down in a series of exergonic reactions that power the work of the cell. The product of each reaction becomes the reactant for the next, so no reaction reaches equilibrium. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Concept 8.3: ATP powers cellular work by coupling exergonic reactions to endergonic reactions • A cell does three main kinds of work – Mechanical – Transport – Chemical Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings The Structure and Hydrolysis of ATP • ATP (adenosine triphosphate) – Is the cell’s energy shuttle – Provides energy for cellular functions Adenine N O O -O O - O - O O C C N HC O O O NH2 - Phosphate groups Figure 8.8 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings N CH2 O H N H H H OH CH C OH Ribose Structure of ATP • Phosphate – phosphate bonds – Negative charges repel, unstable – “high transferable energy” – C-C ~400 KJ/mol while P-P 7.3 KJ/mol – Right amount for most chemical reactions • Each cell contains one billion ATP • Short term storage • Controlled production Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Energy is released from ATP – When the terminal phosphate bond is broken P P P Adenosine triphosphate (ATP) H2O P i + Figure 8.9 Inorganic phosphate P P Adenosine diphosphate (ADP) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Energy • The three types of cellular work – Are powered by the hydrolysis of ATP P i P Motor protein Protein moved (a) Mechanical work: ATP phosphorylates motor proteins Membrane protein ADP + ATP P P Solute P i Solute transported (b) Transport work: ATP phosphorylates transport proteins P Glu + NH3 Reactants: Glutamic acid and ammonia Figure 8.11 NH2 Glu + P i Product (glutamine) made (c) Chemical work: ATP phosphorylates key reactants Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings i The Regeneration of ATP • Catabolic pathways – Drive the regeneration of ATP from ADP and phosphate ATP hydrolysis to ADP + P i yields energy ATP synthesis from ADP + P i requires energy ATP Energy from catabolism (exergonic, energy yielding processes) Figure 8.12 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Energy for cellular work (endergonic, energyconsuming processes) ADP + P i • Concept 8.4: Enzymes speed up metabolic reactions by lowering energy barriers • A catalyst – Is a chemical agent that speeds up a reaction without being consumed by the reaction Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • An enzyme – Is a catalytic protein Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • The activation energy, EA – Is the initial amount of energy needed to start a chemical reaction – Is often supplied in the form of heat from the surroundings in a system Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • The effect of enzymes on reaction rate Course of reaction without enzyme EA without enzyme Free energy EA with enzyme is lower Reactants ∆G is unaffected by enzyme Course of reaction with enzyme Products Progress of the reaction Figure 8.15 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • The active site can lower an EA barrier by – Orienting substrates correctly – Straining substrate bonds – Providing a favorable microenvironment – Covalently bonding to the substrate Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Concept 8.5: Regulation of enzyme activity helps control metabolism • A cell’s metabolic pathways – Must be tightly regulated Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Energy – Flows into an ecosystem as sunlight and leaves as heat Light energy ECOSYSTEM Photosynthesis in chloroplasts Organic CO2 + H2O + O2 Cellular molecules respiration in mitochondria ATP powers most cellular work Figure 9.2 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Heat energy • Some redox reactions – Do not completely exchange electrons – Change the degree of electron sharing in covalent bonds Products Reactants becomes oxidized + CH4 CO 2O2 2 Energy + 2 H2O becomes reduced O O C O H O O H H H C + H H Figure 9.3 Methane (reducing agent) Oxygen (oxidizing agent) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Carbon dioxide Water Oxidation of Organic Fuel Molecules During Cellular Respiration • During cellular respiration – Glucose is oxidized in a series of steps and oxygen is reduced becomes oxidized C6H12O6 + 6O2 6CO2 + 6H2O + Energy becomes reduced Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Electrons from organic compounds – Are usually first transferred to NAD+, a coenzyme – + NAD+ O NH2 H C O OCH O 2 O O P – O OH P O HO – CH HO 2 NH2 N N H O H HO 2 N+ Nicotinamide (oxidized form) H OH N H OH 2e +2H – 2 e + NADH H+ Dehydrogen Reduction of H HO ase 2[H] NH +(from + + C NAD Oxidation of N Nicotinamide food) NADH (reduced form) N H Figure 9.4 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • NADH, the reduced form of NAD+ – Passes the electrons to the electron transport chain • At the end of the chain – Electrons are passed to oxygen, forming water Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • There are three main processes in this metabolic enterprise Electron shuttles span membrane CYTOSOL MITOCHONDRION 2 NADH or 2 FADH2 2 NADH 2 NADH Glycolysis Glucose 2 Pyruvate 6 NADH Citric acid cycle 2 Acetyl CoA + 2 ATP by substrate-level phosphorylation Maximum per glucose: + 2 ATP 2 FADH2 Oxidative phosphorylation: electron transport and chemiosmosis + about 32 or 34 ATP by substrate-level by oxidative phosphorylation, depending on which shuttle transports electrons phosphorylation from NADH in cytosol About 36 or 38 ATP Figure 9.16 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Excitation of Chlorophyll by Light • When a pigment absorbs light – It goes from a ground state to an excited state, which is unstable e– Excited state Heat Photon (fluorescence) Photon Chlorophyll molecule Figure 10.11 A Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Ground state • Produces NADPH, ATP, and oxygen H2O CO2 Light NADP+ ADP CALVIN CYCLE LIGHT REACTIONS ATP NADPH O2 [CH2O] (sugar) Primary acceptor Primary acceptor Fd 2 2 H+ + O2 Pq e H2O e NADP+ NADP+ + 2 H+ reductase 3 NADPH PC e– 5 + H+ P700 P680 Light 6 ATP Figure 10.13 8 e– Cytochrome complex e– Light 1 7 4 Photosystem II (PS II) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Photosystem-I (PS I) A Comparison of Chemiosmosis in Chloroplasts and Mitochondria • Chloroplasts and mitochondria – Generate ATP by the same basic mechanism: chemiosmosis – But use different sources of energy to accomplish this Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Concept 10.3: The Calvin cycle uses ATP and NADPH to convert CO2 to sugar • The Calvin cycle – Is similar to the citric acid cycle – Occurs in the stroma Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • The Calvin cycle Light H2 O CO2 Input 3 (Entering one CO2 at a time) NADP+ ADP CALVIN CYCLE LIGHT REACTION ATP Phase 1: Carbon fixation NADPH O2 Rubisco [CH2O] (sugar) 3 P 3 P P Short-lived intermediate P Ribulose bisphosphate (RuBP) P 6 3-Phosphoglycerate 6 ATP 6 ADP CALVIN CYCLE 3 ADP 3 ATP 6 P P 1,3-Bisphoglycerate 6 NADPH Phase 3: Regeneration of the CO2 acceptor (RuBP) 6 NADPH+ 6 P P 5 (G3P) 6 P Glyceraldehyde-3-phosphate (G3P) P 1 Figure 10.18 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings G3P (a sugar) Output Glucose and other organic compounds Phase 2: Reduction • Overview: Ecosystems, Energy, and Matter • An ecosystem consists of all the organisms living in a community – As well as all the abiotic factors with which they interact Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Concept 54.1: Ecosystem ecology emphasizes energy flow and chemical cycling • Ecosystem ecologists view ecosystems – As transformers of energy and processors of matter Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Ecosystems and Physical Laws • The laws of physics and chemistry apply to ecosystems – Particularly in regard to the flow of energy • Energy is conserved – But degraded to heat during ecosystem processes Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • Energy flows through an ecosystem – Entering as light and exiting as heat Tertiary consumers Microorganisms and other detritivores Detritus Secondary consumers Primary consumers Primary producers Heat Key Chemical cycling Energy flow Figure 54.2 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Sun • Concept 54.2: Physical and chemical factors limit primary production in ecosystems • Primary production in an ecosystem – Is the amount of light energy converted to chemical energy by autotrophs during a given time period • The extent of photosynthetic production – Sets the spending limit for the energy budget of the entire ecosystem Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Gross and Net Primary Production • Total primary production in an ecosystem – Is known as that ecosystem’s gross primary production (GPP) • Not all of this production – Is stored as organic material in the growing plants • Net primary production (NPP) – Is equal to GPP minus the energy used by the primary producers for respiration • Only NPP – Is available to consumers Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Pyramids of Production • This loss of energy with each transfer in a food chain – Can be represented by a pyramid of net production Tertiary consumers Secondary consumers Primary consumers Primary producers Figure 54.11 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 10 J 100 J 1,000 J 10,000 J 1,000,000 J of sunlight Pyramids of Numbers • A pyramid of numbers – Represents the number of individual organisms in each trophic level Trophic level Tertiary consumers Number of individual organisms 3 Secondary consumers 354,904 Primary consumers 708,624 Primary producers Figure 54.13 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 5,842,424 • Worldwide agriculture could successfully feed many more people – If humans all fed more efficiently, eating only plant material Trophic level Secondary consumers Primary consumers Primary producers Figure 54.14 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings • In biological magnification – Toxins concentrate at higher trophic levels because at these levels biomass tends to be lower Concentration of PCBs Herring gull eggs 124 ppm Figure 54.23 Lake trout 4.83 ppm Smelt 1.04 ppm Zooplankton 0.123 ppm Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Phytoplankton 0.025 ppm