* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 01 physical, technological and organizational bases of radiation
Survey
Document related concepts
Transcript
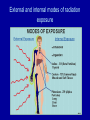
Physical, technological and organizational bases of radiation medicine. Prof. Igor Y. Galaychuk, MD Head, Department of Oncology & Radiology Ternopil State Medical University Physics of Radiation Proton - positive charge, in the nucleus. Neutron - neutral (no) charge, in the nucleus. Electron - negative charge, orbits the nucleus. Physics of Radiation – Stable atoms do not contain excess energy. – Unstable atoms contain excess energy. This is caused by an imbalance in the ratio of protons to neutrons in the nucleus of the atom. These atoms release their excess energy during the process known as radioactive decay. The energy released in the process is called ionizing radiation. Physics of Radiation Radiation - Radiation is energy in the form of waves or particles given off during radioactive decay, or as a consequence of certain physical processes that we can control. Examples of these are x-ray machines and particle accelerators. Wave radiations include gamma and x-rays. A common term used to describe this type of radiation is photon radiation. Particle radiation can consist of charged or uncharged particles which are emitted with very high velocity. Physics of Radiation Radioactive contamination - contamination is radioactive material that is in a form or location which may allow it to be spread to unwanted locations. Many radioactive sources are sealed or are in a form that isolates the material from potential spread. Contamination may be Fixed, Transferable (loose), or Airborne. Physics of Radiation Radioactivity - Radioactivity is the process of unstable (or radioactive) atoms becoming stable by emitting radiation. The radioactive decay process involves fundamental physical constants which enable us to characterize and measure radioactive materials very accurately. • Radioactive half-life - Radioactive half-life is the time is takes for one half of the radioactive atoms present in a given sample to decay. The half-life of a particular isotope is a constant, and depending on the isotope it may range from a fraction of a second to millions of years. After seven half-lives the activity will be less than 1% of the original activity Physics of Radiation • Ionization - The process of ionization is important in understanding radiation, because it is this process that differentiates ionizing radiation from other types. Ionization is the process of removing electrons from atoms. If enough energy is supplied to remove electrons from the atom the remaining atom has a + charge. The positive charges atom and the negatively charges electron are called an ion pair. Ionization should not be confused with radiation. Ions (or ion pairs) can be the result of radiation exposure and allow the detection of radiation. • Typically, we classify types of radiation as ionizing or non ionizing radiations depending on whether or not the radiation can form ion pairs in common material such as air or tissue. Physics of Radiation – Ionizing radiation - Radiation which has enough energy to ionize an atom is called ionizing radiation. The four basic types of ionizing radiation that are of primary concern to us are alpha particles, beta particles, gamma rays (includes x -rays) and neutron particles. – Non-ionizing radiation - Radiation that doesn't have the amount of energy needed to ionize an atom. Examples of non-ionizing radiation are ultraviolet rays, microwaves and visible light. TYPES OF IONIZING RADIATION • Alpha particles are emitted during the decay of certain types of radioactive materials. Compared to other types, the alpha particle has a relatively large mass. It consists of two protons and two neutrons. (Positive charge of plus two.) • The alpha particle deposits a large amount of energy in a short distance of travel. This large energy deposit limits the penetrating ability of the alpha particle to a very short distance. This range in air is about one to two inches. – Shielding • Most alpha particles are stopped by a few centimeters of air, a sheet of paper, or the dead layer (outer layer) of skin on our bodies. – Biological hazard • Alpha particles are not considered an external radiation hazard. This is because they are easily stopped by the dead layer of skin. If alpha emitting radioactive material is inhaled to ingested, it becomes a source of internal exposure. Internally, the source of the alpha radiation is in close contact with body tissue and can deposit large amounts of energy in a small volume of body tissue. • Beta particles • The beta particle is an energetic electron emitted during radioactive decay. Compared to an alpha particle, a beta particle is nearly 8000 times less massive and has half the electrical charge. Beta radiation causes ionization by the same forces at work with alpha radiation mainly electrical interactions with atoms which are encountered as it travels. However, because it is not as highly charged, the beta particle is not as effective at causing ionization. Therefore, it travels further before giving up all its energy and finally coming to rest. • The beta particle has a limited penetrating ability. Its typical range in air is up to about 10 feet. In human tissue, the same beta particle would travel only a few millimeters. – Shielding • Beta particles are easily shielded by relatively thin layers of plastic, glass, aluminum, or wood. Dense materials such as leas should be avoided when shielding beta radiation sue to the increase in production of x-rays in the shield. – Biological Hazard • Externally, beta particles are potentially hazardous to the skin and eyes. They cannot penetrate to deep tissue such as the bone marrow or other internal organs. We call this type of external exposure shallow dose. When taken into the body, materials that emit beta radiation can be a hazard in a similar way to that described from alpha emitters - although comparatively less damage is done in the tissue exposed to the beta emitter. • Gamma rays /x-rays • Gamma /x-ray radiation is an electromagnetic wave or photon and has no electrical charge. Gamma rays and x-rays can be thought of as physically identical. The only difference is in the place of origin. These photons have no mass or charge but can ionize matter as a result of direct interactions with orbital electrons. Like all electromagnetic radiations, gamma rays travel at the speed of light. • Because gamma /x-ray radiation had no charge and no mass, it has a very high penetrating power (said another way, the radiation has a low probability of interacting in matter). Gamma rays have no specific "range" but are characterized by their probability of interacting in a given material. There is no distinct maximum range in matter, but the average range in a given material can be used to compare materials for their shielding ability. • Gamma /x-ray radiation are best shielded by very dense materials, such as lead, concrete, or steel. Shielding is often expressed by thicknesses that provide a certain shielding factor, such as a "half-value layer" (HVL). An HVL is the thickness of a given material required to reduce the dose rate to one half the unshielded dose rate. – Biological hazard • Due to the high penetrating power, gamma /x-ray radiation can result in radiation exposure to the whole body rather than a small area of tissue near the source. Therefore, a photon radiation has the same ability to cause dose to tissue whether the source is inside or outside the body. This is in contrast to alpha radiation for example which must be received internally to be a hazard. Gamma radiation is considered an external hazard. Refer to the definition of "whole body" in the glossary. • Neutron particles. • Physical characteristics: • Neutron radiation consists of neutrons that are ejected from the nuclei of atoms. A neutron has no electrical charge. Due to their charge, neutrons do not interact directly with electrons in matter. A direct interaction occurs as the result of a "collision" between a neutron and the nucleus of an atom. A charges particle or other radiation which can cause ionization may be emitted during these interactions. This is called indirect ionization. • Because neutrons do not experience electrostatic forces, they have a relatively high penetrating ability and are difficult to stop. Like gamma radiation, the range is not absolutely defined. The distance they travel depends on the probability for interaction in a particular material. You can think of neutrons as being "scattered" as they travel through material, with some energy being lost with each scattering event. • Moderate to low energy neutron radiation is best shielded by materials with a high hydrogen content, such as water (H2O) or polyethylene plastic (CH2-CH2-X). High energy neutrons are best shielded by more dense materials such as steel or lead. Sometimes a multi-layered shield will be used to first slow down very 'fast' neutrons, and then absorb the 'slow' neutrons. – Biological hazard • Like gamma radiation, neutrons are an external "whole body" hazard due to their high penetrating ability. … from history of ionizing radiation: • Doctors and scientists paid a price or even lost their lives in their early work with x-rays and radionuclides. Marie Curie, who discovered radium, died of a malignant blood disease probably because of the radiation exposure to her bone marrow during her lengthy research work with the radionuclide. The widespread use of x-ray in medical diagnosis and treatment for some diseases in the early 30's without realizing its harmful effects led to cases of radiation dermatitis and chronic ulceration, eventually resulting in radiation induced cancers. Relative penetrating power of different radiations Different ionization rate in tissue due to different penetrating power of the radiations External and internal modes of radiation exposure Radiation sources • There are many different radiation exposure scenarios that can be evaluated. Some examples follow: • External exposure from relatively distant radiation sources (e.g., neutrons and/or gamma rays) • External exposure from nearby radioactive soil • External exposure from radioactive contamination on the outside of the body • Internal exposure from inhaled radioactive substances • Internal exposure from ingested radioactive substances • Combinations of the above Human exposure pathways associated with radioactive isotopes in the environment Natural sources of radiation • All humans were born slightly radioactive because all living tissue contains radioactive substances. The radioactive characteristic is maintained throughout life. However, the normal radioactivity found in humans is nothing to worry about. Furthermore, nothing can be done to eliminate it. • The main radioactive materials in rocks are potassium-40, rubidium-87, and two series of radioactive elements arising from the decay of uranium-238 and thorium-232. Uranium-238 and thorium-232 are long-lived radioactive isotopes that have remained on Earth since its origin. The levels of terrestrial radiation differ from place to place around the world because the concentrations of these materials in the Earth’s crust vary. • The naturally occurring isotopes carbon-14 and tritium are produced by cosmic radiation. Other naturally occurring isotopes of interest include potassium-40, lead-210, polunium-210, radon-222, and radon-220. Nuclear Reactor Accidents/Destruction • Nuclear accidents, such as occurred in 1979 at Three Mile Island in the U.S. and in 1986 at Chernobyl in Ukraine, lead to public and worker exposures to radiation. Unlike the Three Mile Island accident, the Chernobyl accident led to the loss of many lives. Such accidents can lead to the release of large amounts of alpha-, beta-, and gammaemitting radionuclides (i.e., radioactive isotopes) into the environment. • As with nuclear weapons, fallout can arise from nuclear power plant accidents. The terms "cloud shine" and "ground shine" are therefore also used in describing radiation sources associated with nuclear accidents. • Nuclear facilities can also be destroyed by weapons leading to similar radiation sources such as those that arise from nuclear accidents. HOW ARE DIFFERENT AMOUNTS OF RADIATION EXPRESSED? • The units often used follow: • roentgen (R), rad, gray (Gy), rem, sievert (Sv) • These units relate to radiation exposure, radiation dose, or radiation protection. • Other units (curie, becquerel) relate to radioactivity. The International System of Units (or SI units) includes the gray, sievert, and becquerel: • The gray is the SI unit used for absorbed dose. • The sievert is the SI unit used for the dose equivalent and for the effective dose equivalent. • The becquerel is the SI unit used for radioactivity. Radiation exposure units • The roentgen describes the amount of xrays or gamma rays to which a target (e.g., mouse, rat, human, cow, etc.) is exposed. The roentgen relates to the ability of x-rays and gamma rays to remove electrons from atoms in air. • One roentgen corresponds to 2.58 x 10-4 coulombs per kilogram of air. Radiation absorbed dose units • The absorbed dose relates to how much radiation energy gets put into a given target mass (e.g., lung, eye, thyroid gland). • Different absorbed doses can arise in different organs or tissue of the body for the same exposure in R. Thus, if a person were exposed to 10 R of gamma rays, the eye, the thyroid, and the lung would have different absorbed doses. Special computer programs can calculate such doses. • Units of absorbed dose often used are the rad and gray (an SI unit). • The rad is a relatively old unit of absorbed dose. One rad corresponds to 100 ergs of radiation energy per gram of target substance. • The gray unit represents 1 joule of radiation energy put into a kilogram mass. Thus, 1 gray equals 1 joule per kilogram. • The gray and rad apply to all types of ionizing radiation, unlike the roentgen unit, which only applies to x-rays and gamma rays. • Some useful conversion factors that relate to absorbed dose follow: 1 gray (Gy) = 100 rad, 1 milligray (mGy) = 0.1 rad, 1 rad = 1 centigray (cGy) Radioactivity Units • Radioactivity arises from the disintegration of unstable atoms and is expressed in units like the becquerel (Bq) and curie (Ci). • One becquerel (Bq) corresponds to 1 disintegration (transformation) per second. • One curie represents 37,000,000,000 (i.e., 3.7 x 10x10) disintegrations per second. A curie is a very large amount of radioactivity. • Some useful conversions follow: 1 microcurie (µCi) = 37,000 becquerels (Bq) Radiation-Protection Units • Special dosimetric units are used in radiation protection to limit radiation exposure of nuclear workers and the public. These units include the rem and sievert (SI unit), which apply to single and mixed radiations and are measures of potential harm to humans. • 1 sievert (Sv) = 100 rem. • One rem of alpha radiation would be expected to pose the same risk of harm as 1 rem of gamma rays or as 1 rem of combined exposure to neutrons and gamma rays. • The rem and Sv were developed to account for different efficiencies of different types of radiation in producing harm. Because these units apply to single and mixed radiations, it follows that: • 1 rem of alpha radiation = 1 rem of gamma rays. • 1 rem of gamma rays = 1 rem of neutrons + gamma rays. Types of doses expressed in rem or Sv include: • dose equivalent (applies to single organ) • committed dose equivalent (applies to single organ) • effective dose equivalent (applies to total body) • committed effective dose equivalent (applies to total body) Dose equivalent (applies to single organ) • The dose equivalent is a quantity that accounts for the different efficiencies of different external radiations in causing harm to a given organ or tissue. • Special weighting factors (e.g., quality factor) are used to account for differences in radiation quality. The term "external radiation" simply means that radiation originates from outside the body. Examples are gamma rays from contaminated soil or from a radioactive cloud. Committed dose equivalent (applies to single organ) • The committed dose equivalent is similar to the dose equivalent but applies to doses from radionuclides taken inside the body. • Quality factors are used to account for different efficiencies of different radiations in producing biological damage internally. Committed doses are evaluated to some future time (e.g., to 50 years) after intake of radionuclides. Effective dose equivalent (applies to total body) • Effective dose equivalents account for different biological sensitivities of different organs and tissue and apply to the total body. They also account for different doses (dose equivalents) to different organs. Effective dose equivalents specifically apply to external radiation sources (i.e., sources outside the body). • An effective dose equivalent of 1 Sv for a nonuniform, combined exposure to neutrons and gamma rays would represent the theoretical dose of gamma rays uniformly distributed over the body that would incur the same risk of harm from stochastic effects (mainly cancer) as for the actual nonuniform exposure to neutrons and gamma rays. Committed effective dose equivalent (applies to total body) • Committed effective dose equivalents also account for different biological sensitivities of different organs and tissue but apply only to radionuclides that enter the body (e.g., via inhalation or ingestion). • A committed dose equivalent of 1 Sv from inhaled alpha particle emitters would represent the theoretical committed dose of gamma rays to the total body that would yield the same risk of harm from stochastic effects such as cancer. Committed dose equivalents are evaluated to a fixed time in the future (e.g., 50 years) after intake of radionuclides. Radiation weighting factors • The equivalent dose is obtained by multiplying the absorbed dose by special factors called radiation weighting factors (WR) that are intended to account for different efficiencies of the different radiations in producing biological damage. • Like its predecessor the quality factor, the weighting factor WR was designed to protect against radiation-induced harm (mainly from cancer induction). • Radiation weighting factors, WR, currently used for alpha radiation, beta radiation, gamma rays, and x-rays follow: • WR for alpha radiation = 20 • WR for beta radiation = 1 • WR for gamma radiation = 1 • WR for x-rays = 1 What is the equivalent dose for combined exposure of the cornea (of the eye) to 0.1 Gy of alpha radiation plus 0.2 Gy of gamma rays? The answer follows: Equivalent dose in Sv = WR (alpha radiation)*0.1 Gy + WR (gamma rays)*0.2 Gy. Since WR (alpha radiation) equals 20 and WR (gamma rays) equal 1, you get Equivalent dose in Sv = 20 x 0.1 + 1 x 0.2 = 2.2 Sv. WHAT CAN BE DONE TO PROTECT FROM RADIATION HARM? WHAT CAN BE DONE TO PROTECT FROM RADIATION HARM? • Physical protection is used to prevent radiation exposure of individuals from occurring. • Chemical protection is provided to protect when a possible radiation exposure is anticipated or after an exposure has occurred. • Physiological protection is provided after a radiation incident has occurred to lessen the harm from the radiation exposure. The end?