* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Fatty acids
Survey
Document related concepts
Transcript
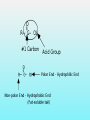
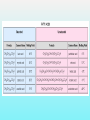
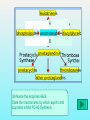
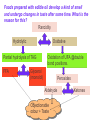
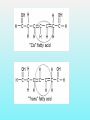
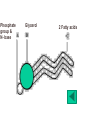
Department of Biochemistry Foundation Module Phase - 1 Lipids Soluble in non-polar solvents and insoluble in polar solvents. Lipids are not polymers. Fatty acids •They are basic building blocks of lipids(simplest lipids). •Fatty acids are carboxylic acids. Q.Fatty Acids are amphipathic molecules. Why? A.Because they have both polar(hydrophilic) and nonpolar(hydrophobic) portions in their structure Classification of fatty acids Fatty acids saturated unsaturated monounsaturated polyunsaturated Q. What is the chemical difference between saturated and unsaturated fatty acids? A. Saturated fatty acids contain only single bonds between carbons, whereas unsaturated fatty acids contain at least one double bond. Corn oil contains 86% polyunsaturated fatty acids Therefore is a liquid at room temperature. Olive oil contains monounsaturated fatty acids. Therefore is a liquid at room temperature, likely to solidify when refrigerated. Saturated fatty acids (tropical oils, animal fats) are solids, or nearly solids at room temperature. Unsaturated fats are preferred over saturated fatty acids for nutritional reasons. Q.Would you expect a deep sea fish (living in cold water) or a shallow water fish (living in warmer water) to have a higher percentage of unsaturated fatty acids in its tissues? A. deep sea fish Q1.Give some examples for simple lipids Fatty acids + Alcohol Q2.Find out some common examples for complex lipids Fatty acids + Alcohol+ X The degree of saturation of a fatty acid affects particularly the fatty acid melting point. Q3.Unsaturated fatty acids have lower melting points than saturated fatty acids. Why is that? Melting Points and Solubility in Water of Fatty Acids Melting Point Solubility in H2O Chain Length Q. What are essential fatty acids? A. Fatty acids which have to be obtained from food, as human cells have no appropriate enzymes required to produce them internally. Q4.What are the two families of essential fatty acids? Good sources of ‘omega-3 fatty acids’ • Oily fish (salmon, mackerel, sardines, tuna) • Pumpkin seeds, sesame seeds • soybean oil Good sources of ‘omega-6 fatty acids’ •Most vegetable oil, Sunflower oil, Corn oil, Soybean oil •Cotton seeds oil •Pumpkin seeds •Nuts and cereals •Poultry, eggs •Avocado Essential fatty acids are used by every cell in your body. Q5.Why are essential fatty acids good for you? Lipids have four major functions which are physiologically important for human. Serve as vitamins and hormones. Structural components of biological membranes. synthesize bile acids that aid lipid solubilization Provide energy Glycerides Glycerides (acylglycerols) are esters formed from glycerol and fatty acids. Glycerides Monoglycerides Diglycerides Triglycerides Q6.What elements are found in lecithin and sphingolipids? Q7. Identify a-j Q8.What are Eicosanoids? A B Q9.Name the enzymes A& B. State the mechanisms by which aspirin and ibuprofen inhibit PGH2 Synthesis. Steroids •General structure of a steroid – four rings Q10.What are the derivatives of steroids? Cholesterol • Most abundant steroid in the body • Add methyl CH3- groups, alkyl chain, and -OH to steroid nucleus Cholesterol in the Body, • Cellular membranes • Myelin sheath, brain, and nerve tissue • Bile salts • Hormones • Vitamin D Q11 . Identify each lipid as: (1) phospholipid (2) steroid (3) triglyceride (4) sphingolipid A. Cholesterol B. Glycerol, 2 fatty acids, phosphate, and choline C. Sphingosine, fatty acid, phosphate, and choline D. Estradiol E. Bile salts F. Most of plasma membranes The diagram represents a phospholipid molecule Q12.Name the following parts of the molecule (i) A (ii) B (iii) C 1.Saponification Value What are the main ingredients in your soap? • • • • • One test tube is filled with Sunflower oil (A). Coconut oil is placed in a second test tube.(B) Add few drops of Hubl’s iodine for each solution. Test tubes are shaken. After some minutes a spatula full of starch powder is added to each of the two test tubes. • The test tubes are again shaken. A Q13.what is the observation? B Foods prepared with edible oil develop a kind of smell and undergo changes in taste after some time. What is the reason for this? Rancidity Hydrolytic Oxidative Partial hydrolysis of TAG FFA Oxidation of UFA @double bond positions. Glycerol (mono/di) Peroxides Aldehyde Objectionable odour + Taste Ketones Health issues associated with lipids Trans fat •Unsaturated fat with trans-isomer fatty acid. •Not essential fatty acids •Naturally found in animal products where they are produced by bacteria in the gut of ruminant animals (eg: cow, goat, sheep) •Therefore some trans fatty acids are present in meat, milk and other dairy products. •These are mostly C18 monounsaturated fatty acid (vaccenic acid). • Daily intake of 5g trans fat increases the risk of heart disease by 25% Trans fatty acids are commonly found in….. How are trans fats made artificially? Trans fats are made through the chemical process called hydrogenation of vegetable oils. Hydrogenation solidifies liquid oils and increases the shelf life and the flavour stability of oils and foods that contain them. Sun flower oil Margarine ANSWERS a.Fats: Fatty acids + Glycerol b. Waxes: Fatty acids + Long chain alcohol Example a.Phospholipid Fatty acids Alcohol Fatty acids Alcohol Fatty acids Glycerol ex: 1.Glycero phospholipid ex: Lecithin Fatty acids 2.Sphingophospho lipid Fatty acids b.Glycolipids(Glycosphi ngo lipid) Glycerol X Phosphoric acid residue Phosphoric acid residue Choline Sphingosin Carbohydrat e e Presence of one or more double bonds in hydrocarbon chain of unsaturated fatty acids results “ BENDS” in the molecules. These molecules do not stack well. As a result of that inter-molecular interactions become much weaker than saturated fatty acids leading to lower melting points. 1. ω-3 (or omega-3 or n-3) 2. ω-6 (omega-6, n-6.) • Alpha-linolenic acid – a polyunsaturated omega-3 fatty acid • Linoleic acid -an unsaturated omega-6 fatty acid. • Control immune system, blood pressure, nervous system, inflammatory reactions, blood clotting and hormonal functions. • Regulate a large number of mechanisms including increasing the fluidity of cell membranes and improving the ability of selective permeability and help to keep toxin out and bring nutrients into cells. • Influence the activation of cell genes and act as second messengers and produce good eicosanoids. • Furthermore, a diet rich in EFAs can be helpful in many diseases( Rheumatoid arthritis, Inflammatory bowel disease) Lecithin • • • • • • • • • • a: Oligosaccharide b: Glycoprotein c: Glycolipids d: Head of phospholipid e:Tail of phospholipid f: Phospholipid bilayer g: Filament h:Peripheral protein i: Cholesterol j: Transmembrane protein Eicosanoids are a family of powerful, hormone-like compounds produced in the body from essential fatty acids . . . • They have specific effects on target cells close to their site of formation. • They are rapidly degraded, so they are not transported to distal sites within the body. • Examples: prostaglandins, prostacyclins, thromboxanes, leukotrienes, epoxyeicosatrienoic acids (EETs). • Roles in inflammation, fever, regulation of blood pressure, blood clotting, immune system modulation, control of reproductive processes & tissue growth, sleep/wake cycle regulation. A: Lipoxygenase B:PGH2 synthase Aspirin and Ibuprofen (a Non-steroid anti inflammatory drug) Inhibit PGH2 Synthase enzyme. Note: Aspirin irreversibly acetylates and thus inactivates the enzyme whereas the action of Ibuprofen is reversible. • • • • Steroid hormones Cholesterol Vitamin D Bile A. B. C. D. E. F. steroid phospholipid sphingolipid steroid steroid phospholipid Phosphate group & N- base Glycerol 2 Fatty acids Observation: The sample containing Coconut oil turns intensely blue (iodine-starch complex). In Sunflower oil all the iodine will be consumed. Consequently no colour change can be observed after the addition of starch. Discussion and background: Sunflower oil contains unsaturated fatty acids. Iodine adds across the double bonds in an unsaturated bond. Coconut oil consists mainly of glycerides from saturated fatty acids (e.g. palmitic and lauric acid).