* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download File - Mrs. Henderson
Survey
Document related concepts
Transcript
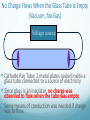
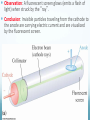
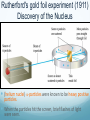
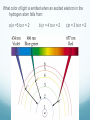
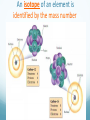
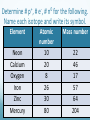
Chapter 4 Atomic Structure Alpharetta High School Dr. Sonha Payne Why do scientists use models? Models may be used to represent things that are difficult to visualize. Scaled-down models allow you to see something too large to see all at once, (solar system) or something that hasn’t been built yet. Scaled-up models are used to visualize things that are too small to see. (atoms) 1st Model of the Atom Just as a brick is basic to the structure of a wall, an atom is basic to the structure of matter. Democritus (400 BC) atomos, meaning uncuttable, or cannot be divided Foundations of Atomic Theory Law of Conservation of Mass (Lavoisier) Mass is neither created nor destroyed during ordinary chemical reactions. Mass products = mass reactants Foundations of Atomic Theory Law of Definite Proportions (Proust) A chemical compound contains the same elements in the same percent by mass regardless of the size of the sample or the source of the sample. Ex: H2O will always have the same percent by mass, 11.2% H and 88.8% O. Foundations of Atomic Theory Law of Multiple Proportions (Dalton) When elements combine, they do so in the ratio of small whole numbers. CO2 and CO H2O and H2O2 Principles of Dalton’s Atomic Theory of Matter (1808) 1. All matter is composed of extremely small, indivisible particles called atoms. 2. All atoms of a given element have identical properties that differ from those of other elements. 3. Atoms cannot be created, destroyed, or transformed into atoms of another element. 4. Compounds are formed when atoms of different elements combine with one another in small wholenumber ratios. 5. The relative numbers and kinds of elements are constant in a given compound. Dalton’s Billiard Ball Model of the Atom Billiard Ball Model because the atom is likened to a billiard ball - it is a single, complete unit of matter. A solid, indivisible, indestructible sphere No Charge Flows When the Glass Tube is Empty (Vacuum, No Gas) Voltage source Cathode Ray Tube: 2 metal plates sealed inside a glass tube connected to a source of electricity Since glass is an insulator, no charge was observed to flow when the tube was empty. Some means of conduction was needed if charge was to flow. Charge Flows (a Ray is observed) in the Presence of ANY GAS Voltage source - + When a small amount of any gas was placed in the glass tube and the power source was turned on, a ray was observed striking the phosphor-coated end of the tube and emitting a flash of light. For current to flow, mobile, charged particles are required. - Voltage source + The current carriers in the gas are “invisible” and are visualized by the phosphor-coated screen which fluoresces as the current carriers strike it. Observation: A fluorescent screen glows (emits a flash of light) when struck by the “ray”. Conclusion: Invisible particles traveling from the cathode to the anode are carrying electric current and are visualized by the fluorescent screen. Observation: When a tiny object is placed in the middle of the tube, a shadow is cast on the screen at the anode Conclusion: the particles travel in a straight line. Cathode Rays Originate at the Cathode (Negative Electrode) The rays were called cathode rays because they originated at the negative electrode (aka the cathode) and moved to the positive electrode (aka the anode). The entire apparatus is now known as a cathode ray tube (CRT). Observation: In the presence of a magnetic field, the cathode rays bend. Conclusion: The cathode ray consists of charged particles. Observation: In the presence of an external electric field, the cathode rays bent towards the positive plate. Conclusion: The particles are negatively charged. ALL gases used in the tube were found to produce identical rays, so the negatively charged particles of the cathode rays were determined to be part of all matter. Observation: A paddle wheel in the middle of the cathode ray tube turns. Conclusion: The particles have mass. JJ Thomson (1897) Credited with discovery of the electron Found the ratio of the mass of the particles to the charge of the particles in cathode rays m/e = -5.6856 x 10-9 g/C Conclusion: all cathode rays are composed of identical, negatively-charged particles. Conclusion: these negative particles are fundamental particles of matter. (electrons) In 1897, JJ Thomson discovered that negatively charged electrons were part of all matter. Dalton Postulate 1: All matter is composed of extremely small, indivisible particles called atoms. This is no longer valid. Atoms are NOT indivisible particles, but CONTAIN ELECTRONS. Millikan Determined the Charge on an Electron by Examining the Motion of Tiny Oil Drops (1909) Small drops of oil with a negative charge are examined. The diameter of the3 drop is measured. From this, the volume is determined (4/3pr ). The mass is calculated using the known density of the oil. By adjusting the electric field to balance the force of gravity, the charge on the drop is calculated. Charge in Coulombs 13.458 x 10-19 = 7 x (1.92 x 10-19) 15.373 x 10-19 = 8 x (1.92 x 10-19) 17.303 x 10-19 = 9 x (1.92 x 10-19) 15.378 x 10-19 = 8 x (1.92 x 10-19) 17.308 x 10-19 = 9 x (1.92 x 10-19) 28.844 x 10-19 = 15 x (1.92 x 10-19) 11.545 x 10-19 = 6 x (1.92 x 10-19) 19.214 x 10-19 = 10 x (1.92 x 10-19) Observation The charge of each drop is a whole number multiple of some common number. Conclusion Each number is divisible by 1.92 x 10-19. This is the charge of an electron. (modern value = 1.602 x 10-19 C) Millikan Determined the Mass of an Electron Knowing the charge, Millikan was able to use Thomson’s chargeto-mass ratio to determine the mass of an electron. charge 1.6022 1019 C 28 mass of an electron = = = 9.10 10 g 8 charge / mass 1.76 10 C/g Inferences from the Properties of Electrons Atoms are neutral, so there must be positively charged particles to balance the negatively charged electrons. Electrons have a tiny mass, so atoms must contain other particles that account for most of their mass. Even the lightest atom, H, has a mass of 1.7 x 10-24 g, compared to a mass of only 9.1 x 10-28 g for an electron. That is, the lightest atom is almost 10,000 x heavier than an electron. Thus, most of the mass of an atom had to come from somewhere else. Thomson’s Plum Pudding Model of the Atom •Since electrons are negatively charged, and atoms are neutral, atoms must also contain a positively charged substance. •Thomson’s model of the atom: the positively charged substance fills the atom and the electrons are embedded throughout the substance like “raisons in plum pudding”. Rutherford's gold foil experiment (1911) Discovery of the Nucleus (helium nuclei) a-particles were known to be heavy positive particles. When the particles hit the screen, brief flashes of light were seen. Rutherford’s Expected and Actual Results If plum-pudding model was correct. Actual results. Rutherford’s Expected and Actual Results • Observation: Most particles went straight through the foil undeflected. • Conclusion: The atom is mostly empty space. • Observation: Some particles bounced back! • Conclusion • To deflect the energetic a-particles, most of the mass and all of the positive charge must be in a dense central region which he called the nucleus. • For the fraction deflected to be small, the nucleus must be small, relative to the overall size of the atom. • Since atoms are neutral particles, the charge of the nucleus must be equal to the sum of the negative charges of the electrons. 1911 Rutherford’s Gold Foil Experiment Conclusion: An atom is mostly empty space occupied by electrons, and centrally located within that space lies a tiny region, which he called the nucleus, that contains all the positive charge and essentially all the mass of the atom. Rutherford proposed that positive particles lay within the nucleus and called them protons. Summary: The Rutherford Experiment (1911) Alpha particles (helium nuclei) fired at a thin sheet of gold Assumed that the positively charged alpha-particles were bounced back if they approached a positively charged atomic nucleus head on (Like charges repel one another) Very few particles were greatly deflected back from the gold sheet Atoms contain very small, very dense, positively-charged nuclei. The electrons are in clouds surrounding the nucleus at relatively large distances. Most of the atom is empty space, and if the nucleus were the size of a ladybug, it would be in an atom the size of the Georgia Dome. Moseley,1913 X-rays, Atomic Number and the Proton A beam of electrons shot at a sample of an element gives off x-rays. Observation The frequency of x-rays given off is unique to that element. Higher energy rays are given off when the nuclear charge is higher. Conclusion The atomic number can be determined from the x-ray data Each element on the periodic chart differs from the next by having one more positive charge in the nucleus These fundamental positive charge units are the protons Bohr’s Theory of the Hydrogen Atom Visible Light Spectrum When white light is passed through a prism, a continuous spectrum of colors results which contains all of the wavelengths of visible light The Hydrogen Line Emission Spectrum When a sample of hydrogen gas is excited by electricity and passed through a prism, only a few lines are seen, each of which corresponds to a discrete wavelength. Every Element Has Its Own Unique Emission Line Spectrum Bohr Attributed the Emission of Radiation to the Electron Dropping From a Higher Energy Orbit to a Lower One 1. Electrons orbit the nucleus in circular orbits. 2. Only orbits of certain radii are permitted. 3. An electron in a permitted orbit has a specific energy. 4. Energy is emitted or absorbed as a photon when the electron changes its orbit. What color of light is emitted when an excited electron in the hydrogen atom falls from: a) n =5 to n = 2 b) n = 4 to n = 2 c)n = 3 to n = 2 The Hydrogen e- Visualized as a Standing Wave Around the Nucleus Only certain circular orbits have a circumference into which a whole number of wavelengths of the standing electron wave will “fit”. Circular orbits with any other circumference produce destructive interference of the standing electron wave and are not allowed. Electron Diffraction Experiments Demonstrated that Electrons Exhibit the Wave Property of Interference Experiments have shown that electrons do indeed possess wavelike properties: X-ray diffraction pattern of aluminum foil Electron diffraction pattern of aluminum foil. Quantum Mechanics The Heisenberg uncertainty principle states that it is impossible to know simultaneously both the momentum p and the position x of a particle with certainty. Δx is the uncertainty in position in meters Δp is the uncertainty in momentum Δu is the uncertainty in velocity in m/s m is the mass in kg If the position of a particle is known more precisely, then it’s velocity measurement must become less precise In the picture, we know the exact location of the cars, but we have no idea how fast they are moving. If the velocity of a particle is measured more precisely then the position must become correspondingly less precise In the picture, we know the speed of the cars, but we have no idea exactly where they are. Which of the following statements regarding Dalton’s atomic theory are still believed to be true? I. Elements are made of tiny particles called atoms. II. All atoms of a given element are identical. III. A given compound always has the same relative numbers and types of atoms. IV. Atoms are indestructible. 43 Modern Atomic Theory 1. All matter is made up of very tiny particles called atoms. 2. Atoms of the same element are chemically alike. 3. Individual atoms of an element may not all have the same mass. However, the atoms of an element have a definite average mass that is characteristic of the element. 4. Atoms of different elements have different average masses. 5. Atoms are not subdivided, created, or destroyed in chemical reactions. Modern Atomic Theory An atom is an electrically neutral, spherical entity composed of a positively charged central nucleus surrounded by one or more negatively charged electrons. The “cloud” of rapidly moving, negatively charged electron occupies virtually all of the atom’s volume and surrounds the tiny nucleus. The nucleus is very dense as it contributes 99.97% of the atom’s mass but occupies only about one ten-trillionth of its volume -10 m) is about 10,000 times the diameter of An atom’s diameter (~10 -14 it’s nucleus (~10 m). The Modern Model of the Atom The precise paths of electrons cannot be determined accurately. Instead, the PROBABILITY of finding electrons in a specific location can be determined. The location and energy of electrons can be specified using three terms: Shell (aka level) Subshell (aka sublevel) Orbital An additional fourth term, spin number, indicates whether the electron is spinning clockwise or What are the particles that make up an atom and where are they located? Particle Symbol Charge Relative Mass (amu) Actual Mass (g) Proton p+ +1 1 1.7 x 10-24 Neutron n 0 1 1.7 x 10-24 Electron e- -1 0 9.1 x 10-28 Atomic Mass Unit (amu): the Unit Used For Masses of Atoms and Subatomic Particles Atoms have an very small mass (e.g. hydrogen 1.67 x 10–24 g) It is hard to work with these small numbers so a relative mass scale was introduced: atomic mass unit (amu or u) 1 u = 1/12 the mass of a carbon-12 atom Particle Symbol Charge Relative Mass (amu) Actual Mass (g) Proton p+ +1 1 1.7 x 10-24 Neutron n 0 1 1.7 x 10-24 Electron e- -1 0 9.1 x 10-28 # Protons Defines an Atom The number of protons distinguishes atoms of one element from atoms of all of the other elements. Atomic number = # protons in the nucleus What Distinguishes One Element from Another Element? The # of Protons! For a Neutral Atom, # protons = # electrons For a neutral atom, the positive charges (+1 per proton) and negative charges (-1 per electron) must add up to zero. Complete the table. Element Pb Atomic number 82 Protons Electrons 8 30 Mass Number = protons + neutrons Always a whole number The mass number is NOT on the periodic table! Carbon-12 Nuclear Symbol and Hyphen Notation are Interchangeable Hydrogen-2 Atoms of the Same Element That Have Different # of Neutrons are ISOTOPES Isotopes of elements have the same # of protons but different # of neutrons. Magnesium-24 Magnesium-25 Magnesium-26 An isotope of an element is identified by the mass number How many protons, electrons, and neutrons do each of these isotopes have? Determine # p+, # e-, # n0 for the following. Name each isotope and write its symbol. Element Neon Calcium Oxygen Iron Zinc Mercury Atomic number 10 20 8 26 30 80 Mass number 22 46 17 57 64 204 Complete each of the following isotope symbols: 206 ? a. 84 197 Au c. ? 224 Ra b. ? d. 84 ? 36 Average Atomic Mass Atomic mass is the mass of an atom in atomic mass units (amu). 1 amu = 1/12 the mass of a carbon-12 atom The average atomic mass on the periodic table represents the average mass of the naturally occurring mixture of isotopes. Isotope Isotopic mass (amu) Natural abundance (%) 12C 12.00000 98.93 13C 13.003355 1.07 = 12.01 amu Average mass (C) = (0.9893)(12.00000 amu) + (0.0107)(13.003355 amu) Isotopes Used in Medicine Some radioactive isotopes are useful in the medical field. 131 53 I Used to detect thyroid problems 60 27 Co Used in cancer therapy to kill cancerous tissue 32 15 P Used in leukemia therapy 137 55 Cs Used to irradiate food to kill bacteria and other organisms Isotopes Is it possible to have a hydrogen isotope with 3 neutrons, hydrogen-4? No, only certain combinations of protons and neutrons are possible; the others are unstable. Not all elements have the same number of isotopes. Every element up to lead (Z = 82) has at least 1 stable isotope. Isotope Abundance Some elements have only 1 isotope, while others have many more. Not all isotopes are present in equal amounts. Oxygen-16 99.762% Oxygen-17 0.038% Oxygen-18 0.200% Total 100.00% Chlorine-35 75.77% Chlorine-37 24.23% Total 100.00% Note: the sum of the percentages is 100% The atomic mass of an element (the number on the periodic table) is the weighted average mass of all its isotopes expressed in atomic mass units. Chlorine (Cl) How to determine the atomic mass of an element What is the atomic weight of chlorine? List each isotope, it’s mass in atomic mass units, and it’s abundance in nature. Isotope Mass (amu) Isotopic Abundance Cl-35 34.97 75.78% = 0.7578 Cl-37 36.97 24.22% = 0.2422 How to determine the atomic mass of an element Multiply the isotopic abundance by the mass of each isotope, and add up the products. The sum is the atomic weight of the element. 34.97 x 0.7578 = 26.5003 amu 36.97 x 0.2422 = 8.9541 amu 35.4544 amu = 4 sig. figs. 35.45 amu Answer 4 sig. figs. Calculating Atomic Mass Using Isotopes Average atomic mass = å(isotope %)(isotope mass) Example average atomic mass of Cl: Chlorine-35 75.77% Chlorine-37 24.23% Total 100.00% Most elements have two or more isotopes that contribute to the atomic mass of that element Average Atomic Mass of Mg Calculating Atomic Mass Using Isotopes Average atomic mass = Example average atomic mass of O å(isotope %)(isotope mass) Oxygen-16 99.762% Oxygen-17 0.038% Oxygen-18 0.200% Total 100.00% = (99.762%)(15.99491464) + (0.038%)(16.9991306) + (0.200%)(17.99915939) = 16.00 u Calculating the Average Mass of an Element Oxygen is the most abundant element in both Earth’s crust and the human 17 16 body. The atomic masses of its three stable isotopes, O (99.757 percent), O 8 8 18 (0.038 percent), O (0.205 percent), are 15.9949, 16.9991, and 17.9992 amu, 8 respectively. Calculate the average atomic mass of oxygen using the relative abundances given in parentheses. Strategy Each isotope contributes to the average atomic mass based on its relative abundance. Multiplying the mass of each isotope by its fractional abundance (percent value divided by 100) will give its contribution to the average atomic mass. Solution (0.99757)(15.9949 amu) + (0.00038)(16.9991 amu) + (0.00205)(17.992 amu) = 15.9994 amu Think About It The average atomic mass should be closest to the atomic mass of the most abundant isotope (in this case, oxygen-16) and, to four significant figures, should be the same number that appears in the periodic table on the inside front cover of your textbook (in this case, 16.00 amu). The Modern Periodic Table Groups contain elements with similar properties and are arranged in vertical columns. Periods are the horizontal rows of elements. Two Numbering Systems for Groups The old method uses the letter A for the representative elements (1A to 8A) and the letter B for the transition elements. The new method numbers groups 1–18 from left to right. Alkali Metals Special Names of Groups Halogens Shape of the Periodic Table with Lanthanides and Actinides Inserted Into the Main Section Metals, Nonmetals, and Metalloids The heavy zigzag line separates metals and nonmetals. Metals are located to the left. Nonmetals are located to the right. Metalloids are located along the heavy zigzag line between the metals and nonmetals (except for Al) Metals solids at room temperature, except Hg reflective surface (shiny) conduct heat and electricity Malleable (can be shaped) Ductile (can be pulled into wires) lose electrons and form cations in reactions About 75% of the elements are metals. 79 Nonmetals Exist as s, l, g poor conductors of heat and electricity Solids are brittle. gain electrons in reactions to become anions upper right on the table except H Sulfur, S(s) Chlorine, Cl2(g Bromine, Br2(l) 80 Metalloids show some properties of metals and some of nonmetals also known as semiconductors Properties of Silicon shiny conducts electricity does not conduct heat Properties of Metals, Nonmetals, and Metalloids Metals Shiny (s) Ductile, malleable Good conductors Metalloids Nonmetals Dull (s, l, g) Brittle Better conductors than Good insulators nonmetals, but not as good as metals