Reflectivity measurements of a quantum well
... aquisition time, grating and entrance slit size; • by moving focusing lens 2 in horizontal direction find the position when the spot of white light is focused on the sample surface; • by moving focusing lens 3 in all three directions find the maximum of the signal from the sample (first using the br ...
... aquisition time, grating and entrance slit size; • by moving focusing lens 2 in horizontal direction find the position when the spot of white light is focused on the sample surface; • by moving focusing lens 3 in all three directions find the maximum of the signal from the sample (first using the br ...
Lecture 3 Teaching notes
... wavefunctions for one particle, and the total wavefunction for all the electrons of a many-electron system. I will try to distinguish these two senses by speaking of a “configuration” of a many-particle system when there is a chance of confusion. The lowest-energy configuration state of the system i ...
... wavefunctions for one particle, and the total wavefunction for all the electrons of a many-electron system. I will try to distinguish these two senses by speaking of a “configuration” of a many-particle system when there is a chance of confusion. The lowest-energy configuration state of the system i ...
Spectrophotometry and its Applications in Microbiology
... “turbid”. Thus we are measuring the turbidity of the sample (some instruments used for this purpose are called turbidimeters). Since the reading we obtain when using the “absorbance” setting on the spectrophotometer is not truly absorbance, another term we use to describe this value is Optical Densi ...
... “turbid”. Thus we are measuring the turbidity of the sample (some instruments used for this purpose are called turbidimeters). Since the reading we obtain when using the “absorbance” setting on the spectrophotometer is not truly absorbance, another term we use to describe this value is Optical Densi ...
Conduction and Semiconductors
... 3. He bands are bent, or curved, where the built-in electric fields exist at the junction 4. A potential energy step, qVc, due to contact potential Vc, develops at the junction. It is equal in magnitude to EF(n)-EF(p) or, equivalently qF(p)-qF(n) 5. Externally applied electric potentials displace th ...
... 3. He bands are bent, or curved, where the built-in electric fields exist at the junction 4. A potential energy step, qVc, due to contact potential Vc, develops at the junction. It is equal in magnitude to EF(n)-EF(p) or, equivalently qF(p)-qF(n) 5. Externally applied electric potentials displace th ...
paper 1 - ResearchGate
... 4) Porphyrin rings like the one in the figure at the right appear in myoglobin and chlorophyll. A porphyrin ring contains 26 π electrons which may be treated as particles in a thin square box a) What is the expression for the energies of a particle in a two dimensional square box as a function of t ...
... 4) Porphyrin rings like the one in the figure at the right appear in myoglobin and chlorophyll. A porphyrin ring contains 26 π electrons which may be treated as particles in a thin square box a) What is the expression for the energies of a particle in a two dimensional square box as a function of t ...
ppt
... Nanostructures on the sample surface are found to be entirely dotlike, while capped nanostructures are predominantly ringlike. Moreover, an in situ anneal process applied after thinly capping the dots is shown to enhance the severity of the rings and relax the strain in the matrix in the proximity o ...
... Nanostructures on the sample surface are found to be entirely dotlike, while capped nanostructures are predominantly ringlike. Moreover, an in situ anneal process applied after thinly capping the dots is shown to enhance the severity of the rings and relax the strain in the matrix in the proximity o ...
Chapt. 5: Quantum Theory of the Hydrogen Atom
... Solutions to the wave equation are called: wave functions (ψ) For hydrogen, get the same electron energies as Bohr did The square of the wave function (ψ2) gives a probability density for an electron in a specified energy state ...
... Solutions to the wave equation are called: wave functions (ψ) For hydrogen, get the same electron energies as Bohr did The square of the wave function (ψ2) gives a probability density for an electron in a specified energy state ...
Atomic Orbitals and quantum numbers
... •Therefore, on any given energy level, there can be up to 1s orbital, 3p orbitals, 5d orbitals, and 7f orbitals. ...
... •Therefore, on any given energy level, there can be up to 1s orbital, 3p orbitals, 5d orbitals, and 7f orbitals. ...
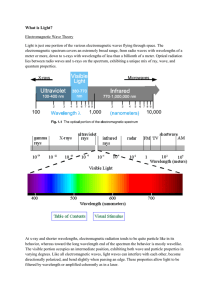
What is Light?
... intensity and angle of incidence of each LED must match and mix appropriately in order to create the white light correctly. The combination of blue and yellow light also gives the impression of white light since the yellow light stimulates the sensors of the eye that are sensitive to red and green, ...
... intensity and angle of incidence of each LED must match and mix appropriately in order to create the white light correctly. The combination of blue and yellow light also gives the impression of white light since the yellow light stimulates the sensors of the eye that are sensitive to red and green, ...
Pre-Knowledge: Chemistry and Physics Vocabulary Atomic Number
... The sum of the number of neutrons and protons in the nucleus of an atom. Nucleus The small “core” of the atom, where most of its mass and all of its positive charge is concentrated. Except for ordinary hydrogen (which has only a proton), atomic nuclei consist of protons and neutrons. For this reason ...
... The sum of the number of neutrons and protons in the nucleus of an atom. Nucleus The small “core” of the atom, where most of its mass and all of its positive charge is concentrated. Except for ordinary hydrogen (which has only a proton), atomic nuclei consist of protons and neutrons. For this reason ...
Quantum Mechanics
... When UV light is shone on a metal plate in a vacuum, it emits charged particles (Hertz 1887), which were later shown to be electrons by J.J. Thomson (1899). Light, frequency ν ...
... When UV light is shone on a metal plate in a vacuum, it emits charged particles (Hertz 1887), which were later shown to be electrons by J.J. Thomson (1899). Light, frequency ν ...
chapter40
... He assumed the cavity radiation came from atomic oscillations in the cavity walls Planck made two assumptions about the nature of the oscillators in the cavity walls ...
... He assumed the cavity radiation came from atomic oscillations in the cavity walls Planck made two assumptions about the nature of the oscillators in the cavity walls ...
I. Physical Biochemistry Protein Interactions Ultraviolet(UV
... to an acceptor molecule without emission of a photon. ...
... to an acceptor molecule without emission of a photon. ...
Atoms, electrons, nuclei J.J. Thomson discovered the electron (1897
... no energy loss and anode current (I) will increase; if Ugrid = U*, collisions might become inelastic: electrons may transfer their energy to a Mercury atom and anode current will decrease Coclusion: energy of Mercury atom cannot change continuously, but only by certain discrete values, so-called qua ...
... no energy loss and anode current (I) will increase; if Ugrid = U*, collisions might become inelastic: electrons may transfer their energy to a Mercury atom and anode current will decrease Coclusion: energy of Mercury atom cannot change continuously, but only by certain discrete values, so-called qua ...
Atomic Structure
... properties of moving particles are insignificant in our everyday world. A moving object such as a car or a tennis ball has an incredibly small wavelength associated with it. It is on the atomic scale that the dual nature of particles and light become significant. ...
... properties of moving particles are insignificant in our everyday world. A moving object such as a car or a tennis ball has an incredibly small wavelength associated with it. It is on the atomic scale that the dual nature of particles and light become significant. ...
Document
... • If the right amount of energy is added, the electron moves up. To move down, a certain amount of energy must be released. ...
... • If the right amount of energy is added, the electron moves up. To move down, a certain amount of energy must be released. ...
ki̇mya
... • the electrons in an atom move at a certain distance from nucleus and their motions are stable . Each stationery state has a definite energy. • Electrons move in each stationary energy state in a circular orbital. These circular orbitals are called energy levels or shells. The possible states for t ...
... • the electrons in an atom move at a certain distance from nucleus and their motions are stable . Each stationery state has a definite energy. • Electrons move in each stationary energy state in a circular orbital. These circular orbitals are called energy levels or shells. The possible states for t ...
Abstract
... on a generalized Boltzmann-type equation, including energy-drift and anti-diffusion effects, interband excitation, Coulomb scattering, thermal exchange with the lattice, etc. The kinetic Fokker-Planck equation describes the physics of these processes and can be used to explain how they lead to a las ...
... on a generalized Boltzmann-type equation, including energy-drift and anti-diffusion effects, interband excitation, Coulomb scattering, thermal exchange with the lattice, etc. The kinetic Fokker-Planck equation describes the physics of these processes and can be used to explain how they lead to a las ...
chem 1411- chapter 7
... get excited to higher energy levels by absorbing energy. This is the excited state of an atom, which is unstable. The electrons then start falling from higher levels to lower levels, releasing energy. This energy when resolved through a spectroscope, we get different lines of specific wavelengths. T ...
... get excited to higher energy levels by absorbing energy. This is the excited state of an atom, which is unstable. The electrons then start falling from higher levels to lower levels, releasing energy. This energy when resolved through a spectroscope, we get different lines of specific wavelengths. T ...
The Photoelectric Effect
... Many people are aware that Einstein was awarded the Nobel Prize in Physics in 1921 for his work on the photoelectric effect in 1905, but fewer people are aware of the work that preceded his explanation. In 1887, Heinrich Hertz first observed the photoelectric effect when investigating “spark gap” ra ...
... Many people are aware that Einstein was awarded the Nobel Prize in Physics in 1921 for his work on the photoelectric effect in 1905, but fewer people are aware of the work that preceded his explanation. In 1887, Heinrich Hertz first observed the photoelectric effect when investigating “spark gap” ra ...
Matter
... – Collection of the combination same type of atom elements and/or – Cannot be compounds or decomposed both. • Compound • USUALLY – 2 or more different heterogeneous atoms chemically bonded together. ...
... – Collection of the combination same type of atom elements and/or – Cannot be compounds or decomposed both. • Compound • USUALLY – 2 or more different heterogeneous atoms chemically bonded together. ...
File - SPHS Devil Physics
... the work done in moving the electrons from the cathode to the collecting plate, which is the same as the maximum kinetic energy of the electrons, ...
... the work done in moving the electrons from the cathode to the collecting plate, which is the same as the maximum kinetic energy of the electrons, ...
Final Exam 2004
... two atoms. For large R, the dipole-dipole interaction can be considered as a small perturbation. Show that the energy of the dipole-dipole interaction of the two atoms in their ground states is zero in the first order of the perturbation theory. [Hint: Since the ground state is nondegenerate, you ca ...
... two atoms. For large R, the dipole-dipole interaction can be considered as a small perturbation. Show that the energy of the dipole-dipole interaction of the two atoms in their ground states is zero in the first order of the perturbation theory. [Hint: Since the ground state is nondegenerate, you ca ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.