ON POSSIBILITY OF MEASUREMENT OF THE
... aspects. In particular, it is found that the kinematical restrictions on the photon absorption process lead to interesting selection effects in angles of propagation of photons which can be absorbed by electrons. This circumstance is crucial for finding the energy of electrons. The electron beam ene ...
... aspects. In particular, it is found that the kinematical restrictions on the photon absorption process lead to interesting selection effects in angles of propagation of photons which can be absorbed by electrons. This circumstance is crucial for finding the energy of electrons. The electron beam ene ...
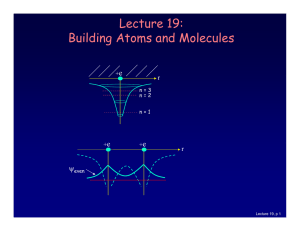
Lecture 19: Building Atoms and Molecules
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
Read PDF - Physics
... frequency. As expected, the power spectrum shows peaks at all frequencies, and the peaks at frequencies fE and fF are higher than the rest. The surprising result is obtained when the interferometer is modified to be a which-path experiment, using the nested MZI as a switch. By slightly shifting mirr ...
... frequency. As expected, the power spectrum shows peaks at all frequencies, and the peaks at frequencies fE and fF are higher than the rest. The surprising result is obtained when the interferometer is modified to be a which-path experiment, using the nested MZI as a switch. By slightly shifting mirr ...
Materials processing with a tightly focused femtosecond laser vortex
... high-power broadband laser vortex beams. The traditional methods utilizing spiral phase plates and holograms are inherently chromatic and therefore require the introduction of correcting elements in order to compensate for the topological charge dispersion occurring in polychromatic pulses. Uncompen ...
... high-power broadband laser vortex beams. The traditional methods utilizing spiral phase plates and holograms are inherently chromatic and therefore require the introduction of correcting elements in order to compensate for the topological charge dispersion occurring in polychromatic pulses. Uncompen ...
The Transactional Interpretation of Quantum Mechanics http://www
... 1. we have a quantum beam-splitter in superposition of being present or absent, the interferometer is in a superposition of being closed or open. This forces the quantum entity to be in a superposition of particle and wave at the same time. 2. The proposal, explicitly elaborates that both particle a ...
... 1. we have a quantum beam-splitter in superposition of being present or absent, the interferometer is in a superposition of being closed or open. This forces the quantum entity to be in a superposition of particle and wave at the same time. 2. The proposal, explicitly elaborates that both particle a ...
Photoelectric Effect
... The photoelectric effect is the emission of electrons when electromagnetic radiation falls on an object. It was first theorized by a German physicist named Max Planck and later expanded upon by Albert Einstein. Light consists of discrete bundles of energy called photons. The amount of energy in a ph ...
... The photoelectric effect is the emission of electrons when electromagnetic radiation falls on an object. It was first theorized by a German physicist named Max Planck and later expanded upon by Albert Einstein. Light consists of discrete bundles of energy called photons. The amount of energy in a ph ...
Chemistry (CP) Final Exam Study Guide 1
... ____ 27. Which of the following is a chemical property? a. color c. freezing point b. hardness d. ability to react with oxygen ____ 28. Which of the following is a chemical property of water at 4 C? a. its color b. its state c. its temperature d. its ability to decompose into hydrogen and oxygen ___ ...
... ____ 27. Which of the following is a chemical property? a. color c. freezing point b. hardness d. ability to react with oxygen ____ 28. Which of the following is a chemical property of water at 4 C? a. its color b. its state c. its temperature d. its ability to decompose into hydrogen and oxygen ___ ...
PDF: Aspden et al 2016 b
... Young’s double slit experiment, which falls into a small class of experiments that have a special place in both the history and development of physics. Generically, illumination of a double slit with spatially coherent light (usually an expanded laser beam) creates sinusoidal fringes in the far fiel ...
... Young’s double slit experiment, which falls into a small class of experiments that have a special place in both the history and development of physics. Generically, illumination of a double slit with spatially coherent light (usually an expanded laser beam) creates sinusoidal fringes in the far fiel ...
1. Atomic Structure
... www.sakshieducation.com The orbital with the lowest (n + l) value is filled first. When two or more orbitals have the same (n + l) value, the one with the lowest ‘n’ value is preferred in filling. Consider two orbitals 3d and 4s. The n + l value of 3d = 3 + 2 = 5 and of 4s = 4 + 0 = 4. Since 4s has ...
... www.sakshieducation.com The orbital with the lowest (n + l) value is filled first. When two or more orbitals have the same (n + l) value, the one with the lowest ‘n’ value is preferred in filling. Consider two orbitals 3d and 4s. The n + l value of 3d = 3 + 2 = 5 and of 4s = 4 + 0 = 4. Since 4s has ...
Nuclear(1).
... Radiation comes from the nucleus of an atom. Unstable nucleus emits a particle or energy alpha ...
... Radiation comes from the nucleus of an atom. Unstable nucleus emits a particle or energy alpha ...
Electro Optic Sampling of Ultrashort Mid
... Fourier analysis. Reflective properties of materials can be observed by comparison of measurements made with and without a sample, or measurements made at various temperatures, probe beam delay times, etc. ...
... Fourier analysis. Reflective properties of materials can be observed by comparison of measurements made with and without a sample, or measurements made at various temperatures, probe beam delay times, etc. ...
a) What is the difference between a semiconductor and a
... resistance of the material will increase linear while the temperature of the material is increasing (therefore the atom lattice vibrations) and with a negative temperature coefficient vice versa. This effect happens, due to the scattering of the free moving electrons with the atom lattice. Now phono ...
... resistance of the material will increase linear while the temperature of the material is increasing (therefore the atom lattice vibrations) and with a negative temperature coefficient vice versa. This effect happens, due to the scattering of the free moving electrons with the atom lattice. Now phono ...
Chapter7 Exercises - Berkeley City College
... possible allowed values for ml also indicate the number of degenerate orbitals within each sublevel. (Degenerate orbitals are orbitals having the same energy). For example, each subshell s has only one orbital; subshell p has three orbitals; subshell d has five orbitals; subshell f has seven (7) orb ...
... possible allowed values for ml also indicate the number of degenerate orbitals within each sublevel. (Degenerate orbitals are orbitals having the same energy). For example, each subshell s has only one orbital; subshell p has three orbitals; subshell d has five orbitals; subshell f has seven (7) orb ...
BRIEF REPORTS
... resonances!; the rate of phase change is largest on resonance. ~iv! The diameter of the ellipse is related to the product of the oscillator strength of the two resonances separated by energy « ~i.e., the larger the oscillator strength of the resonance, the larger the ellipse!. It is clear from Fig. ...
... resonances!; the rate of phase change is largest on resonance. ~iv! The diameter of the ellipse is related to the product of the oscillator strength of the two resonances separated by energy « ~i.e., the larger the oscillator strength of the resonance, the larger the ellipse!. It is clear from Fig. ...
Lecture 19: Quantization of the simple harmonic oscillator Phy851 Fall 2009
... cε=0 – Conclusion: either δ = 0, or there is a state lower than the ground state The second option is obviously a contradiction – For |ε0〉 to be the ground state requires δ = 0 ...
... cε=0 – Conclusion: either δ = 0, or there is a state lower than the ground state The second option is obviously a contradiction – For |ε0〉 to be the ground state requires δ = 0 ...
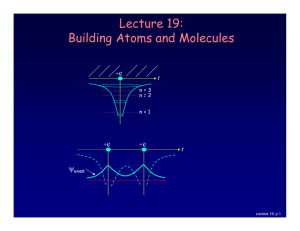
Lecture 19: Building Atoms and Molecules
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
Electrons in the Atom
... So scientists agreed to limit these calculations to locations where there was at least a 90% chance of finding an electron. Think of orbitals as sort of a "border” for spaces around the nucleus inside which electrons are allowed. No more than 2 electrons can ever be in 1 orbital. The orbital j ...
... So scientists agreed to limit these calculations to locations where there was at least a 90% chance of finding an electron. Think of orbitals as sort of a "border” for spaces around the nucleus inside which electrons are allowed. No more than 2 electrons can ever be in 1 orbital. The orbital j ...
Soft x-ray laser holography with wavelength resolution *
... temporal and spatial coherence of the illuminating source, the resolution of the medium in which the hologram is recorded, and the digitization process used are also factors that influence the ultimate resolution. The experimental setup described in this paper was optimized to limit the degrading ef ...
... temporal and spatial coherence of the illuminating source, the resolution of the medium in which the hologram is recorded, and the digitization process used are also factors that influence the ultimate resolution. The experimental setup described in this paper was optimized to limit the degrading ef ...
Worksheet - 1 - International Indian School, Riyadh
... 7. How many unpaired electrons are present in N? Name the principle which explains the presence of these unpaired electrons. 2 or more marks Question: 8. Write a short note on Plank’s Quantum theory. 9. Calculate the wavelength of an electron that has been accelerated in a particle accelerator throu ...
... 7. How many unpaired electrons are present in N? Name the principle which explains the presence of these unpaired electrons. 2 or more marks Question: 8. Write a short note on Plank’s Quantum theory. 9. Calculate the wavelength of an electron that has been accelerated in a particle accelerator throu ...
Document
... Hydrogen atoms in states of high quantum number have been created in the laboratory and observed in space. (a) Find the quantum number of the Bohr orbit in a hydrogen atom whose radius is 0.0199 mm. (b) What is the energy of a hydrogen atom in this case? (Beiser, ...
... Hydrogen atoms in states of high quantum number have been created in the laboratory and observed in space. (a) Find the quantum number of the Bohr orbit in a hydrogen atom whose radius is 0.0199 mm. (b) What is the energy of a hydrogen atom in this case? (Beiser, ...
CHEMISTRY 102B Name Hour Exam II March 19, 2015 Signature
... Which of the following best evaluates the statement “The 1st ionization energy for an oxygen atom is lower than the 1st ionization energy for a nitrogen atom”? a) It is consistent with the general trend relating changes in ionization energy across a period from left to right because it is easier to ...
... Which of the following best evaluates the statement “The 1st ionization energy for an oxygen atom is lower than the 1st ionization energy for a nitrogen atom”? a) It is consistent with the general trend relating changes in ionization energy across a period from left to right because it is easier to ...
No Slide Title
... light bends as the rays travel through a substance. Therefore, this property is key to optical research. Currently, refractive index measurements are performed primarily with refractrometric instruments. These instruments measure the angle of refraction of light through a material. The refractive in ...
... light bends as the rays travel through a substance. Therefore, this property is key to optical research. Currently, refractive index measurements are performed primarily with refractrometric instruments. These instruments measure the angle of refraction of light through a material. The refractive in ...
Review for Chapter 3: Atoms, Electrons and Periodic Trends Text
... predict where the electrons will be found most of the time. He developed mathematical equations called wave functions to allow him to predict the space around the nucleus where the electrons could be found 95% of the time, and he called these spaces orbitals. 6. Pauli’s exclusion principle states th ...
... predict where the electrons will be found most of the time. He developed mathematical equations called wave functions to allow him to predict the space around the nucleus where the electrons could be found 95% of the time, and he called these spaces orbitals. 6. Pauli’s exclusion principle states th ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.