Chapter 1 Homework - due Tuesday, Sept
... the production of ATP from ADP and Pi is catalyzed, and oxygen is reduced, forming water 4. What are the roles of NAD+ and FAD in aerobic respiration? NAD+ and FAD receive electrons at varying steps during glycolysis (NAD+ only) and the citric acid cycle (both NAD+ and FAD), to form NADH and FADH2, ...
... the production of ATP from ADP and Pi is catalyzed, and oxygen is reduced, forming water 4. What are the roles of NAD+ and FAD in aerobic respiration? NAD+ and FAD receive electrons at varying steps during glycolysis (NAD+ only) and the citric acid cycle (both NAD+ and FAD), to form NADH and FADH2, ...
Respiration
... transferred to carrier molecule NAD (nicotinamide adenine dinucleotide) to form reduced NAD (NADH) • Each NADH molecule can be used to transfer energy to other molecules during respiration • The end product of glycolysis, pyruvate (3C), still contains chemical potential energy ...
... transferred to carrier molecule NAD (nicotinamide adenine dinucleotide) to form reduced NAD (NADH) • Each NADH molecule can be used to transfer energy to other molecules during respiration • The end product of glycolysis, pyruvate (3C), still contains chemical potential energy ...
Reading Guide for Week 4
... 10. From section 6.4, know the difference between substrate-level phosphorylation and oxidative phosphorylation. What are the two sequential processes of oxidative phosphorylation? Make sure to review how the proton motive force is generated. Be able to explain Figure 6.18. 11. From section 6.4, kno ...
... 10. From section 6.4, know the difference between substrate-level phosphorylation and oxidative phosphorylation. What are the two sequential processes of oxidative phosphorylation? Make sure to review how the proton motive force is generated. Be able to explain Figure 6.18. 11. From section 6.4, kno ...
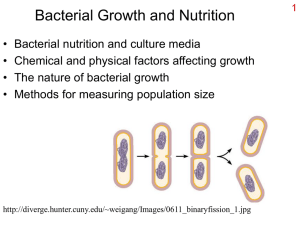
Bacterial Growth and Nutrition
... • Not all bacteria can use the same things – Some molecules cannot be transported in – Enzymes for metabolizing it might not be present – Chemical may be used, but more expensive – These differences are used for identification ...
... • Not all bacteria can use the same things – Some molecules cannot be transported in – Enzymes for metabolizing it might not be present – Chemical may be used, but more expensive – These differences are used for identification ...
doc 3.5.4 cycles gap fills Gap fills for carbon and nitrogen
... Nutrient cycles These consider how inorganic nutrients cycle through the various trophic levels and remain constantly available. The carbon cycle Carbon dioxide in the atmosphere and …………………………………… carbon dioxide in the oceans provide the major source of ……………………………… carbon for organisms. The carbon ...
... Nutrient cycles These consider how inorganic nutrients cycle through the various trophic levels and remain constantly available. The carbon cycle Carbon dioxide in the atmosphere and …………………………………… carbon dioxide in the oceans provide the major source of ……………………………… carbon for organisms. The carbon ...
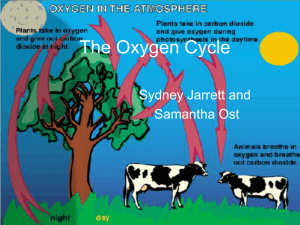
The Oxygen Cycle - EDHSGreenSea.net
... off in plants. • Plants use photosynthesis to produce carbohydrates and oxygen • Plants “breathe” in carbon dioxide and “breathe” out oxygen ...
... off in plants. • Plants use photosynthesis to produce carbohydrates and oxygen • Plants “breathe” in carbon dioxide and “breathe” out oxygen ...
cellular energies - Fairfield Public Schools
... • converts solar energy into chemical energy (ATP) ...
... • converts solar energy into chemical energy (ATP) ...
-The oxygen consumed during cellular respiration is involved
... -The primary role of oxygen in cellular respiration is to _____. -During aerobic respiration, H2O is formed. Where does the oxygen atom for the formation of the water ...
... -The primary role of oxygen in cellular respiration is to _____. -During aerobic respiration, H2O is formed. Where does the oxygen atom for the formation of the water ...
Cell Respiration ch. 9
... If molecular oxygen & mitochondria are present……. Each pyruvate is converted into acetyl CoA (begin w/ 2): CO2 is released; NAD+ ---> NADH; In each turn 2 C atoms enter (Acetyl CoA) and 2 exit (carbon dioxide) Oxaloacetate is regenerated (the “cycle”) For each pyruvate that enters: 3 NAD+ reduced to ...
... If molecular oxygen & mitochondria are present……. Each pyruvate is converted into acetyl CoA (begin w/ 2): CO2 is released; NAD+ ---> NADH; In each turn 2 C atoms enter (Acetyl CoA) and 2 exit (carbon dioxide) Oxaloacetate is regenerated (the “cycle”) For each pyruvate that enters: 3 NAD+ reduced to ...
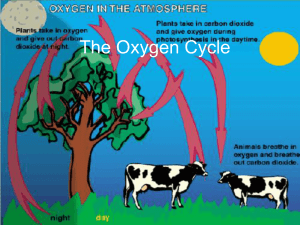
The Oxygen Cycle
... • The oxygen cycle starts off in plants. • Plants use photosynthesis to produce carbohydrates and oxygen • Plants “breathe” in carbon dioxide and “breathe” out oxygen ...
... • The oxygen cycle starts off in plants. • Plants use photosynthesis to produce carbohydrates and oxygen • Plants “breathe” in carbon dioxide and “breathe” out oxygen ...
Ch. 4: ATP and Cellular Respiration
... Energy • Stored in chemical bonds of compounds. • Compounds that store energy: ATP, NADH and FADH2. • When bonds are broken, energy is released. ...
... Energy • Stored in chemical bonds of compounds. • Compounds that store energy: ATP, NADH and FADH2. • When bonds are broken, energy is released. ...
Characteristics of Life PPT
... Materials and Energy Take in materials and energy to survive •Plants use light energy and make food •Animals eat food to get energy • Fungus digest dead material to get energy ...
... Materials and Energy Take in materials and energy to survive •Plants use light energy and make food •Animals eat food to get energy • Fungus digest dead material to get energy ...
Biogeochemical Cycling In The Ecosystem pp. 371
... Warm- up 3/17: •What is a trophic level? •How much energy is transferred between trophic levels? • Where does the lost energy go? ...
... Warm- up 3/17: •What is a trophic level? •How much energy is transferred between trophic levels? • Where does the lost energy go? ...
Sample Exam 2 Questions
... A acetyl-CoA. B. citric acid. C. ADP D. oxidized electron carriers. E. reduced electron carriers. 8. How many ATP molecules are synthesized directly in the Krebs cycle if you supply aerobically respiring cells with 10 pyruvate molecules? A. 2 B. 5 C. 10 D. 20 E. 300 9. In cellular metabolism, O2 is ...
... A acetyl-CoA. B. citric acid. C. ADP D. oxidized electron carriers. E. reduced electron carriers. 8. How many ATP molecules are synthesized directly in the Krebs cycle if you supply aerobically respiring cells with 10 pyruvate molecules? A. 2 B. 5 C. 10 D. 20 E. 300 9. In cellular metabolism, O2 is ...
Respiration Notes (chapter 8)
... 3.at the end of the chain is oxygen -the energy derived from the ETC is used to power the proton pump which services ATP synthase (Chemiosmosis). =Oxidative phosphorylation --686 Cal in Glucose --7.3 Cal in ATP x 36 = 263 Cal out of 686 Cal are conserved =39% Efficiency Is that Good? ...
... 3.at the end of the chain is oxygen -the energy derived from the ETC is used to power the proton pump which services ATP synthase (Chemiosmosis). =Oxidative phosphorylation --686 Cal in Glucose --7.3 Cal in ATP x 36 = 263 Cal out of 686 Cal are conserved =39% Efficiency Is that Good? ...
Answers to the RI and UC questions
... 2. Where in a cell does each part of cell respiration take place? Describe how the location of each part of the process is different in bacteria an din more complex cells. The cytosol is the site of glycolysis in both prokaryotic and eukaryotic cells. The mitochondrion is the site of the Krebs cycle ...
... 2. Where in a cell does each part of cell respiration take place? Describe how the location of each part of the process is different in bacteria an din more complex cells. The cytosol is the site of glycolysis in both prokaryotic and eukaryotic cells. The mitochondrion is the site of the Krebs cycle ...
Microbial physiology. Microbial metabolism. Enzymes. Nutrition
... the TCA cycle Circuit of organic acids – series of oxidations and reductions ...
... the TCA cycle Circuit of organic acids – series of oxidations and reductions ...
File - SBI
... 8. Why isn't anaerobic respiration effective for larger organisms? a. The energy yield is too small b. It causes too much glucose to be burned up c. It results in products that may be toxic to the organism d. NAD+ is lost over time because it can't be regenerated e. Only d is false 9. More ATP is pr ...
... 8. Why isn't anaerobic respiration effective for larger organisms? a. The energy yield is too small b. It causes too much glucose to be burned up c. It results in products that may be toxic to the organism d. NAD+ is lost over time because it can't be regenerated e. Only d is false 9. More ATP is pr ...
Cellular Respiration 2
... Glycolysis 2 ATP, Krebs 2 ATP, Electron Transport & oxidative phosphorylation 34 ATP Total of 38 ATP/glucose Photosynthesis and Respiration Complementary processes Plants perform both, heterotrophs only use respiration Each uses waste product of other process (O2 and CO2) Fermentation ...
... Glycolysis 2 ATP, Krebs 2 ATP, Electron Transport & oxidative phosphorylation 34 ATP Total of 38 ATP/glucose Photosynthesis and Respiration Complementary processes Plants perform both, heterotrophs only use respiration Each uses waste product of other process (O2 and CO2) Fermentation ...
Ecological Networks - ChaosAndComplexity
... • Study of the interactions between organisms and their environment • Study of ecosystems – Ecosystem- web/network of relationships of organisms to each other and their environment ...
... • Study of the interactions between organisms and their environment • Study of ecosystems – Ecosystem- web/network of relationships of organisms to each other and their environment ...
Respiration Cellular respiration Redox Various Ways of Harvesting
... ◦ Use of inorganic molecules (other than O2) as final electron acceptor ◦ Many prokaryotes use sulfur, nitrate, carbon dioxide or even inorganic ...
... ◦ Use of inorganic molecules (other than O2) as final electron acceptor ◦ Many prokaryotes use sulfur, nitrate, carbon dioxide or even inorganic ...
Microbial metabolism
Microbial metabolism is the means by which a microbe obtains the energy and nutrients (e.g. carbon) it needs to live and reproduce. Microbes use many different types of metabolic strategies and species can often be differentiated from each other based on metabolic characteristics. The specific metabolic properties of a microbe are the major factors in determining that microbe’s ecological niche, and often allow for that microbe to be useful in industrial processes or responsible for biogeochemical cycles.== Types of microbial metabolism ==All microbial metabolisms can be arranged according to three principles:1. How the organism obtains carbon for synthesising cell mass: autotrophic – carbon is obtained from carbon dioxide (CO2) heterotrophic – carbon is obtained from organic compounds mixotrophic – carbon is obtained from both organic compounds and by fixing carbon dioxide2. How the organism obtains reducing equivalents used either in energy conservation or in biosynthetic reactions: lithotrophic – reducing equivalents are obtained from inorganic compounds organotrophic – reducing equivalents are obtained from organic compounds3. How the organism obtains energy for living and growing: chemotrophic – energy is obtained from external chemical compounds phototrophic – energy is obtained from lightIn practice, these terms are almost freely combined. Typical examples are as follows: chemolithoautotrophs obtain energy from the oxidation of inorganic compounds and carbon from the fixation of carbon dioxide. Examples: Nitrifying bacteria, Sulfur-oxidizing bacteria, Iron-oxidizing bacteria, Knallgas-bacteria photolithoautotrophs obtain energy from light and carbon from the fixation of carbon dioxide, using reducing equivalents from inorganic compounds. Examples: Cyanobacteria (water (H2O) as reducing equivalent donor), Chlorobiaceae, Chromatiaceae (hydrogen sulfide (H2S) as reducing equivalent donor), Chloroflexus (hydrogen (H2) as reducing equivalent donor) chemolithoheterotrophs obtain energy from the oxidation of inorganic compounds, but cannot fix carbon dioxide (CO2). Examples: some Thiobacilus, some Beggiatoa, some Nitrobacter spp., Wolinella (with H2 as reducing equivalent donor), some Knallgas-bacteria, some sulfate-reducing bacteria chemoorganoheterotrophs obtain energy, carbon, and reducing equivalents for biosynthetic reactions from organic compounds. Examples: most bacteria, e. g. Escherichia coli, Bacillus spp., Actinobacteria photoorganoheterotrophs obtain energy from light, carbon and reducing equivalents for biosynthetic reactions from organic compounds. Some species are strictly heterotrophic, many others can also fix carbon dioxide and are mixotrophic. Examples: Rhodobacter, Rhodopseudomonas, Rhodospirillum, Rhodomicrobium, Rhodocyclus, Heliobacterium, Chloroflexus (alternatively to photolithoautotrophy with hydrogen)