Chapter 23 (Section 3) Pregnancy, Birth, and
... 1. The basic UNIT of all MATTER is the __________ 2. ATOM is the SMALLEST particle of an ___________ retaining all the chemical _____________ of that ELEMENT 3. Democritus’s GREEK term “____________,” which means “not able to be divided,” is the ORIGIN of the word, ___________ *4. Individual ATOMS ...
... 1. The basic UNIT of all MATTER is the __________ 2. ATOM is the SMALLEST particle of an ___________ retaining all the chemical _____________ of that ELEMENT 3. Democritus’s GREEK term “____________,” which means “not able to be divided,” is the ORIGIN of the word, ___________ *4. Individual ATOMS ...
MASS-INDEPENDENT ISOTOPE FRACTIONATION OF CHROMIUM
... For example 54Cr anomalies were observed by one laboratory [9] but not by the other [7] in the same materials thought to come from Vesta. Whether 53Cr anomalies reflect heterogeneous distribution of 53Mn in the solar nebula, Mn/Cr fractionation, or 53Cr heterogeneity is still to be understood [7]. H ...
... For example 54Cr anomalies were observed by one laboratory [9] but not by the other [7] in the same materials thought to come from Vesta. Whether 53Cr anomalies reflect heterogeneous distribution of 53Mn in the solar nebula, Mn/Cr fractionation, or 53Cr heterogeneity is still to be understood [7]. H ...
Periodic Table of Elements
... Periodic Table of Elements - History During the nineteenth century, chemists began to categorize the elements according to similarities in their physical and chemical properties. The end result of these studies was our modern periodic table. In 1829, J. Doebereiner classified some elements into gro ...
... Periodic Table of Elements - History During the nineteenth century, chemists began to categorize the elements according to similarities in their physical and chemical properties. The end result of these studies was our modern periodic table. In 1829, J. Doebereiner classified some elements into gro ...
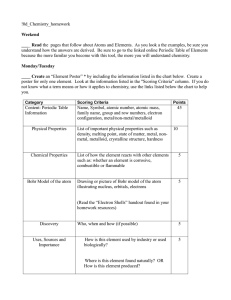
10_Chemistry homework
... ____ Create an “Element Poster” * by including the information listed in the chart below. Create a poster for only one element. Look at the information listed in the "Scoring Criteria" column. If you do not know what a term means or how it applies to chemistry, use the links listed below the chart t ...
... ____ Create an “Element Poster” * by including the information listed in the chart below. Create a poster for only one element. Look at the information listed in the "Scoring Criteria" column. If you do not know what a term means or how it applies to chemistry, use the links listed below the chart t ...
Elements and Compounds checklist for web
... Niels Bohr to our understanding of the structure of atoms. • Identify the elements named after the above scientists. • Identify as many elements named after other scientists or places. Extension work: -‐ D ...
... Niels Bohr to our understanding of the structure of atoms. • Identify the elements named after the above scientists. • Identify as many elements named after other scientists or places. Extension work: -‐ D ...
pdf.format - San Diego Mesa College
... Elements are made from atoms having the same atomic number, protons Are all atoms of one particular atom the same or are they mixtures? 1) All atom nuclei for an element have the same number of protons. 2) Every atom in an element has the same number of protons & electrons. 3) However, elements are ...
... Elements are made from atoms having the same atomic number, protons Are all atoms of one particular atom the same or are they mixtures? 1) All atom nuclei for an element have the same number of protons. 2) Every atom in an element has the same number of protons & electrons. 3) However, elements are ...
I. Atoms are the smallest forms
... elements – Atoms changing identity • Chemical reactions do not effect the nucleus of an atom • Certain conditions can change the number of protons • Each atom has isotopes with different numbers of neutrons • Stability of nucleus depends on the right number of protons and neutrons • Too few or too m ...
... elements – Atoms changing identity • Chemical reactions do not effect the nucleus of an atom • Certain conditions can change the number of protons • Each atom has isotopes with different numbers of neutrons • Stability of nucleus depends on the right number of protons and neutrons • Too few or too m ...
atoms
... How then are atoms of one element different from another element? Elements are different because they contain different numbers of PROTONS The “atomic number” of an element is the number of protons in the nucleus # protons in an atom = # electrons in a neutral ...
... How then are atoms of one element different from another element? Elements are different because they contain different numbers of PROTONS The “atomic number” of an element is the number of protons in the nucleus # protons in an atom = # electrons in a neutral ...
PowerPoint
... • Identify the structural differences between the isotopes of elements. • Determine the weighted average mass of an element when given the appropriate data. ...
... • Identify the structural differences between the isotopes of elements. • Determine the weighted average mass of an element when given the appropriate data. ...
Chemistry Atoms Learning Objectives Atoms Essential knowledge
... The average atomic mass for each element is the weighted average of that element’s naturally occurring isotopes. The mass number of an element is the sum of the number of protons and neutrons. It is different for each element’s isotopes. An isotope is an atom that has the same number of protons as a ...
... The average atomic mass for each element is the weighted average of that element’s naturally occurring isotopes. The mass number of an element is the sum of the number of protons and neutrons. It is different for each element’s isotopes. An isotope is an atom that has the same number of protons as a ...
File - Mrs. Dawson`s Classroom
... Example: Hydrogen has 3 isotopes: protium, deuterium, tritium (radioactive Example: Tin has 10 stable isotopes (the most of any element) ...
... Example: Hydrogen has 3 isotopes: protium, deuterium, tritium (radioactive Example: Tin has 10 stable isotopes (the most of any element) ...
Subatomic Particles - Ciencias Esmeralda
... If you round the atomic mass it gives you the mass number for the most common isotope. Unit is amu (atomic mass unit) 1 amu is 1/12 the mass of C-12 Gram atomic mass= amu but in grams ...
... If you round the atomic mass it gives you the mass number for the most common isotope. Unit is amu (atomic mass unit) 1 amu is 1/12 the mass of C-12 Gram atomic mass= amu but in grams ...
Laws
... • During a chemical reaction, a group combines 5.00 grams of sodium and 7.72 grams of chlorine. The result of the reaction was 12.72 grams of sodium chloride. Which law does this support? ...
... • During a chemical reaction, a group combines 5.00 grams of sodium and 7.72 grams of chlorine. The result of the reaction was 12.72 grams of sodium chloride. Which law does this support? ...
I. Atoms
... III. Distinguishing Between Atoms • Atomic number: ─ equal to the number of protons in an atom ─ in a neutral atom: # protons = # electrons How to find on the periodic table: the WHOLE number ...
... III. Distinguishing Between Atoms • Atomic number: ─ equal to the number of protons in an atom ─ in a neutral atom: # protons = # electrons How to find on the periodic table: the WHOLE number ...
Nuclide, Atomic Number, mass number - Chemwiki
... Elements can also have isotopes with the same atomic number, but different numbers of neutrons. There may be a few more or a few less neutrons, and so the mass is increased or decreased. On the periodic table, the mass number is usually located below the element symbol. The mass number listed is th ...
... Elements can also have isotopes with the same atomic number, but different numbers of neutrons. There may be a few more or a few less neutrons, and so the mass is increased or decreased. On the periodic table, the mass number is usually located below the element symbol. The mass number listed is th ...
Chapter 4 and 25 Study Guide
... radiation? Atomic number decreases by 2; increases by 1; stays the same 32. What stops each type of radiation? Alpha-paper; beta-wood or metal; gamma-lots of lead or concrete 33. What happens in a chain reaction? the products of the first reaction start the later reactions ...
... radiation? Atomic number decreases by 2; increases by 1; stays the same 32. What stops each type of radiation? Alpha-paper; beta-wood or metal; gamma-lots of lead or concrete 33. What happens in a chain reaction? the products of the first reaction start the later reactions ...
Homework #1 Atoms
... 1. Dalton theorized that atoms are indivisible, but the discovery of _______________ particles changed this theory. Scientists now know that atoms are made up of electrons, which have a _____________ charge; _____________, which have a positive charge; and _____________, which are neutral. The latte ...
... 1. Dalton theorized that atoms are indivisible, but the discovery of _______________ particles changed this theory. Scientists now know that atoms are made up of electrons, which have a _____________ charge; _____________, which have a positive charge; and _____________, which are neutral. The latte ...
1 | Page Chemistry Lecture #19: Atomic Number, Isotopes, and
... You can also find and print one off the internet. Try and find the element nitrogen on the periodic chart. near the top right hand side. ...
... You can also find and print one off the internet. Try and find the element nitrogen on the periodic chart. near the top right hand side. ...
Unit 2 Review Game
... • During a chemical reaction, a group combines 5.00 grams of sodium and 7.72 grams of chlorine. The result of the reaction was 12.72 grams of sodium chloride. Which law does this support? ...
... • During a chemical reaction, a group combines 5.00 grams of sodium and 7.72 grams of chlorine. The result of the reaction was 12.72 grams of sodium chloride. Which law does this support? ...
atom
... • Use atomic mass and percent of each isotope to calculate the contribution of each isotope to the weighted average. Atomic mass 35Cl x % abundance = Atomic mass 37Cl x % abundance = • Sum is atomic mass of Cl is ...
... • Use atomic mass and percent of each isotope to calculate the contribution of each isotope to the weighted average. Atomic mass 35Cl x % abundance = Atomic mass 37Cl x % abundance = • Sum is atomic mass of Cl is ...
Chem Ch. 4.3
... • The atomic mass of an element is the weighted average mass of the isotopes of that element. • Atomic mass, therefore, takes into account the percent abundance of each isotope as well as the mass of an atom of that isotope. • The isotope in greater abundance will have a greater effect on determinin ...
... • The atomic mass of an element is the weighted average mass of the isotopes of that element. • Atomic mass, therefore, takes into account the percent abundance of each isotope as well as the mass of an atom of that isotope. • The isotope in greater abundance will have a greater effect on determinin ...
Honors Chemistry Name Julien Period _____ Date Atoms and
... b. All atoms of a given element are identical to one another and different from atoms of other elements. c. Atoms of two or more different elements combine to form compounds. A particular compound is always made up of the same kinds of atoms and the same number of each kind of atom. d. A chemical r ...
... b. All atoms of a given element are identical to one another and different from atoms of other elements. c. Atoms of two or more different elements combine to form compounds. A particular compound is always made up of the same kinds of atoms and the same number of each kind of atom. d. A chemical r ...
Atom - Malibu High School
... Nucleus: the center of the atom; composed of neutrons and protons. Because the mass of the proton and the neutron is much larger than that of electrons, almost all the mass is located in the nucleus. Ion: a charged particle; # protons ≠ # electrons Electrons occupy most of the volume of an ato ...
... Nucleus: the center of the atom; composed of neutrons and protons. Because the mass of the proton and the neutron is much larger than that of electrons, almost all the mass is located in the nucleus. Ion: a charged particle; # protons ≠ # electrons Electrons occupy most of the volume of an ato ...
EPSc 446 STABLE ISOTOPE GEOCHEMISTRY Instructor: Bob Criss
... Found that a few α particles were deflected through large angles- up to 180°. ...
... Found that a few α particles were deflected through large angles- up to 180°. ...
NUCLEAR CHEMISTRY AND RADIOACTIVE DECAY
... 17. Sr-90 is a common waste product of nuclear fission reactors. How many half-lives of Sr-90 will pass after 144 years (half life = 28.8 yrs)? ...
... 17. Sr-90 is a common waste product of nuclear fission reactors. How many half-lives of Sr-90 will pass after 144 years (half life = 28.8 yrs)? ...
Einsteinium

Einsteinium is a synthetic element with symbol Es and atomic number 99. It is the seventh transuranic element, and an actinide.Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952, and named after Albert Einstein. Its most common isotope einsteinium-253 (half life 20.47 days) is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but at much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside of basic scientific research. In particular, einsteinium was used to synthesize, for the first time, 17 atoms of the new element mendelevium in 1955.Einsteinium is a soft, silvery, paramagnetic metal. Its chemistry is typical of the late actinides, with a preponderance of the +3 oxidation state; the +2 oxidation state is also accessible, especially in solids. The high radioactivity of einsteinium-253 produces a visible glow and rapidly damages its crystalline metal lattice, with released heat of about 1000 watts per gram. Difficulty in studying its properties is due to einsteinium-253's conversion to berkelium and then californium at a rate of about 3% per day. The isotope of einsteinium with the longest half life, einsteinium-252 (half life 471.7 days) would be more suitable for investigation of physical properties, but it has proven far more difficult to produce and is available only in minute quantities, and not in bulk. Einsteinium is the element with the highest atomic number which has been observed in macroscopic quantities in its pure form, and this was the common short-lived isotope einsteinium-253.Like all synthetic transuranic elements, isotopes of einsteinium are very radioactive and are considered highly dangerous to health on ingestion.