1 TEST DATE:
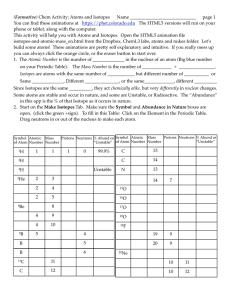
... mass ______________________________ of an atom. The number of neutrons in an atom can be found by subtracting the atomic number from the ____________________________ number. The mass of the atom is so small that there is a measure called the atomic _________________________ unit with a symbol of “µ. ...
... mass ______________________________ of an atom. The number of neutrons in an atom can be found by subtracting the atomic number from the ____________________________ number. The mass of the atom is so small that there is a measure called the atomic _________________________ unit with a symbol of “µ. ...
Atomic Theoryx
... • -all matter is made of atoms • -atoms of one element are identical, atoms of different elements are different • -atoms form compounds in simple whole number ratios • -chemical reactions are rearrangements of atoms, the atoms are not changed • -compounds with different ratios of atoms are different ...
... • -all matter is made of atoms • -atoms of one element are identical, atoms of different elements are different • -atoms form compounds in simple whole number ratios • -chemical reactions are rearrangements of atoms, the atoms are not changed • -compounds with different ratios of atoms are different ...
Make a large atom with p:95, n:146, e:95 - TSDCurriculum
... number of protons plus neutrons in the nucleus). An atom with 8 protons and 8 neutrons is called "Oxygen - 16", or 16O for short. The 16 in 16O is a superscript on the left side of the element symbol. You can also write "O-16 ". 6. DO and WRITE: 16O is one of the isotopes of oxygen. a. Write the nam ...
... number of protons plus neutrons in the nucleus). An atom with 8 protons and 8 neutrons is called "Oxygen - 16", or 16O for short. The 16 in 16O is a superscript on the left side of the element symbol. You can also write "O-16 ". 6. DO and WRITE: 16O is one of the isotopes of oxygen. a. Write the nam ...
Atom Internet Scavenger Hunt
... The Atom and Its Structure Answer Key In the 5th century B.C., a Greek philosopher named Democritus proposed that matter was made up of smaller particles and was the first person to write it down. But he had no experimental proof. A number of scientists after this attempted to prove Democritus’ theo ...
... The Atom and Its Structure Answer Key In the 5th century B.C., a Greek philosopher named Democritus proposed that matter was made up of smaller particles and was the first person to write it down. But he had no experimental proof. A number of scientists after this attempted to prove Democritus’ theo ...
Atomic structure and periodic table review questions What is an
... 3. Another name for the two sub-atomic particles found in the nucleus is ____________. 4. What are the sub-atomic particles found outside of the nucleus? 5. Which particle has a positive charge? 6. Which particle has a neutral charge? 7. Which particle has a negative charge? 8. An element’s atomic n ...
... 3. Another name for the two sub-atomic particles found in the nucleus is ____________. 4. What are the sub-atomic particles found outside of the nucleus? 5. Which particle has a positive charge? 6. Which particle has a neutral charge? 7. Which particle has a negative charge? 8. An element’s atomic n ...
Help us improve Wikipedia by supporting it financially
... nucleosynthesis during the first 20 minutes of the universe[10] in a ratio of around 3:1 by mass (approximately 12:1 by number of atoms). Almost all other elements found in nature, including some further hydrogen and helium created since then, were made by various natural or (at times) artificial me ...
... nucleosynthesis during the first 20 minutes of the universe[10] in a ratio of around 3:1 by mass (approximately 12:1 by number of atoms). Almost all other elements found in nature, including some further hydrogen and helium created since then, were made by various natural or (at times) artificial me ...
Atoms and nukes packet 2016
... something in your backyard! They’re searching for 30 million year old fish fossils, 2000 year old cups and billion year old rocks! Radioactive dating will help us estimate the age of some objects. But objects of different ages need isotopes with different length half lives. After about 6 half lives, ...
... something in your backyard! They’re searching for 30 million year old fish fossils, 2000 year old cups and billion year old rocks! Radioactive dating will help us estimate the age of some objects. But objects of different ages need isotopes with different length half lives. After about 6 half lives, ...
1 - Atomic Theory - Crestwood Local Schools
... 2. Atoms of the same element are identical. 3. The atoms of one element are different from the atoms of another element. 4. Atoms combine in simple whole-number ratios. 5. Atoms are separated, joined or rearranged in chemical reactions. Atoms of one element are never changed into atoms of another el ...
... 2. Atoms of the same element are identical. 3. The atoms of one element are different from the atoms of another element. 4. Atoms combine in simple whole-number ratios. 5. Atoms are separated, joined or rearranged in chemical reactions. Atoms of one element are never changed into atoms of another el ...
Periodic Table Extra Practice ANSWER KEY 2014
... third isotope is the radioactive hydrogen-3. The Bohr diagrams for the isotopes of hydrogen are shown below. Notice that only the mass number and number of neutrons are different. ...
... third isotope is the radioactive hydrogen-3. The Bohr diagrams for the isotopes of hydrogen are shown below. Notice that only the mass number and number of neutrons are different. ...
Chapter 4, Lesson 2: The Periodic Table
... Each student should find and present some basic information about their element to the class. The presentation can be in the form of a poster, pamphlet, PowerPoint presentation or other form. The presentations should be short and can include: atom name, atomic number, derivation of name, when and wh ...
... Each student should find and present some basic information about their element to the class. The presentation can be in the form of a poster, pamphlet, PowerPoint presentation or other form. The presentations should be short and can include: atom name, atomic number, derivation of name, when and wh ...
ch03 - earthjay science
... actual geologic dating (30): The actual age, expressed in years, of a geologic material or event. alpha particle (37): A particle equivalent to the nucleus of a helium atom, emitted from an atomic nucleus during radioactive decay. Archean Eon (30): Pertaining to the division of Precambrian beginning ...
... actual geologic dating (30): The actual age, expressed in years, of a geologic material or event. alpha particle (37): A particle equivalent to the nucleus of a helium atom, emitted from an atomic nucleus during radioactive decay. Archean Eon (30): Pertaining to the division of Precambrian beginning ...
Understanding the Atom
... covered a photographic plate with black paper, this energy would pass through the paper and expose the film. An image of the mineral appeared on the plate. One day, Becquerel left the mineral in a drawer next to a wrapped, unexposed plate. Later, he unwrapped the plate and found that it contained an ...
... covered a photographic plate with black paper, this energy would pass through the paper and expose the film. An image of the mineral appeared on the plate. One day, Becquerel left the mineral in a drawer next to a wrapped, unexposed plate. Later, he unwrapped the plate and found that it contained an ...
The Periodic Table of the Elements
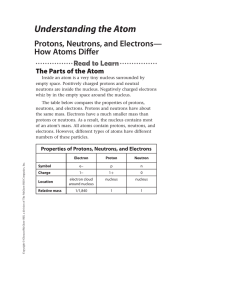
... C. How many electrons are in the atom? An atom is neutral, which means that the number of electrons is equal to the number of protons. An atom of Pb has 82 protons and ...
... C. How many electrons are in the atom? An atom is neutral, which means that the number of electrons is equal to the number of protons. An atom of Pb has 82 protons and ...
Chapter 4, Lesson 2: The Periodic Table
... or neutron). The majority of the atomic mass is contributed by the protons and neutrons. For any element in the periodic table, the number of electrons in an atom of that element always equals the number of protons in the nucleus. But this is not true for neutrons. Atoms of the same element can have ...
... or neutron). The majority of the atomic mass is contributed by the protons and neutrons. For any element in the periodic table, the number of electrons in an atom of that element always equals the number of protons in the nucleus. But this is not true for neutrons. Atoms of the same element can have ...
Chemistry Chapter 4 (Due October 24) [Test
... ____ 36. Which of the following equals one atomic mass unit? a. the mass of one electron b. the mass of one helium-4 atom c. the mass of one carbon-12 atom d. one-twelfth the mass of one carbon-12 atom ____ 37. Which of the following statements is NOT true? a. Protons have a positive charge. b. Ele ...
... ____ 36. Which of the following equals one atomic mass unit? a. the mass of one electron b. the mass of one helium-4 atom c. the mass of one carbon-12 atom d. one-twelfth the mass of one carbon-12 atom ____ 37. Which of the following statements is NOT true? a. Protons have a positive charge. b. Ele ...
Atomic Theory PPT
... Atomic Mass o The atomic mass of an element represents the average mass of all the isotopes found in nature. No element exists with only one possible isotope. Hydrogen has the smallest number of isotopes: 1H protium, 2H deuterium, 3H tritium. Its atomic mass is 1.0079 amu (atomic mass units). The a ...
... Atomic Mass o The atomic mass of an element represents the average mass of all the isotopes found in nature. No element exists with only one possible isotope. Hydrogen has the smallest number of isotopes: 1H protium, 2H deuterium, 3H tritium. Its atomic mass is 1.0079 amu (atomic mass units). The a ...
Name
... Copper consists of 69.15% copper-63, which has an atomic mass of 62.929 601 amu, and 30.85% copper-65, which has an atomic mass of 64.927 794 amu. The average atomic mass of copper can be calculated by multiplying the atomic mass of each isotope by its relative abundance (expressed in decimal form) ...
... Copper consists of 69.15% copper-63, which has an atomic mass of 62.929 601 amu, and 30.85% copper-65, which has an atomic mass of 64.927 794 amu. The average atomic mass of copper can be calculated by multiplying the atomic mass of each isotope by its relative abundance (expressed in decimal form) ...
Chapter 3—Time and Geology
... actual geologic dating (28): The actual age, expressed in years, of a geologic material or event. alpha particle (35): A particle equivalent to the nucleus of a helium atom, emitted from an atomic nucleus during radioactive decay. Archean Eon (28): Pertaining to the division of Precambrian time begi ...
... actual geologic dating (28): The actual age, expressed in years, of a geologic material or event. alpha particle (35): A particle equivalent to the nucleus of a helium atom, emitted from an atomic nucleus during radioactive decay. Archean Eon (28): Pertaining to the division of Precambrian time begi ...
Materials Required
... 5. Indicate the number of protons and neutrons in the nucleus of a particular element or isotope of an element 6. Define isotope as having a different number of neutrons Standards Ohio Standard: Physical Sciences 9-10 Benchmark A: Describe that matter is made of minute particles called atoms and ato ...
... 5. Indicate the number of protons and neutrons in the nucleus of a particular element or isotope of an element 6. Define isotope as having a different number of neutrons Standards Ohio Standard: Physical Sciences 9-10 Benchmark A: Describe that matter is made of minute particles called atoms and ato ...
PowerPoint for Ch 2 Part 2 - Dr. Samples` Chemistry Classes
... are H-2 (there are basically 0% H-3). This is called the natural abundance or %-abundance of an isotope. • So shouldn’t the “average” H atom look a lot like H-1, and shouldn’t the average atomic mass of H be very close to the mass of the H-1 isotope? • Because the different isotopes do not have equa ...
... are H-2 (there are basically 0% H-3). This is called the natural abundance or %-abundance of an isotope. • So shouldn’t the “average” H atom look a lot like H-1, and shouldn’t the average atomic mass of H be very close to the mass of the H-1 isotope? • Because the different isotopes do not have equa ...
Introduction to Atoms
... • Atoms are the building blocks of all materials • An atom is made of 3 parts: – Protons and Neutrons are in the nucleus (center) – Electrons orbit around the nucleus ...
... • Atoms are the building blocks of all materials • An atom is made of 3 parts: – Protons and Neutrons are in the nucleus (center) – Electrons orbit around the nucleus ...
Document
... element. The percentage by mass of each isotope in the mixture does not change from sample to sample. The atomic masses of the elements listed on the periodic table do not show the atomic mass of any one particular isotope of an element. Rather, the periodic table shows an average mass of all the na ...
... element. The percentage by mass of each isotope in the mixture does not change from sample to sample. The atomic masses of the elements listed on the periodic table do not show the atomic mass of any one particular isotope of an element. Rather, the periodic table shows an average mass of all the na ...
Key - Seattle Central College
... called atomos (meaning “not to cut) or atoms. Empedocles (490-430 B.C.): suggested all matter was composed of four basic elements: air, water, fire, and earth. Aristotle (384-321 B.C.): accepted Empedocles idea and added a fifth element, heavenly ether, which is perfect, eternal, and incorruptible. ...
... called atomos (meaning “not to cut) or atoms. Empedocles (490-430 B.C.): suggested all matter was composed of four basic elements: air, water, fire, and earth. Aristotle (384-321 B.C.): accepted Empedocles idea and added a fifth element, heavenly ether, which is perfect, eternal, and incorruptible. ...
Vocabulary Review
... e. positively charged particle that exists in the nucleus of an atom f. tiny bits of matter that are the building blocks of an atom Set Two----------------------------------------------------------------------------------------------------------1. _____group of elements a. atoms of the same element ...
... e. positively charged particle that exists in the nucleus of an atom f. tiny bits of matter that are the building blocks of an atom Set Two----------------------------------------------------------------------------------------------------------1. _____group of elements a. atoms of the same element ...
Einsteinium

Einsteinium is a synthetic element with symbol Es and atomic number 99. It is the seventh transuranic element, and an actinide.Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952, and named after Albert Einstein. Its most common isotope einsteinium-253 (half life 20.47 days) is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but at much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside of basic scientific research. In particular, einsteinium was used to synthesize, for the first time, 17 atoms of the new element mendelevium in 1955.Einsteinium is a soft, silvery, paramagnetic metal. Its chemistry is typical of the late actinides, with a preponderance of the +3 oxidation state; the +2 oxidation state is also accessible, especially in solids. The high radioactivity of einsteinium-253 produces a visible glow and rapidly damages its crystalline metal lattice, with released heat of about 1000 watts per gram. Difficulty in studying its properties is due to einsteinium-253's conversion to berkelium and then californium at a rate of about 3% per day. The isotope of einsteinium with the longest half life, einsteinium-252 (half life 471.7 days) would be more suitable for investigation of physical properties, but it has proven far more difficult to produce and is available only in minute quantities, and not in bulk. Einsteinium is the element with the highest atomic number which has been observed in macroscopic quantities in its pure form, and this was the common short-lived isotope einsteinium-253.Like all synthetic transuranic elements, isotopes of einsteinium are very radioactive and are considered highly dangerous to health on ingestion.