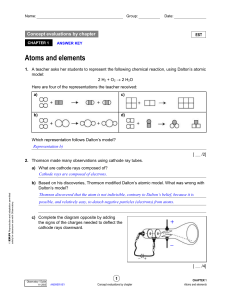
Atomic mass
... different numbers of neutrons. Hydrogen has three isotopes: Protium – 0 neutrons Deuterium – 1 neutron Tritium – 2 neutrons ...
... different numbers of neutrons. Hydrogen has three isotopes: Protium – 0 neutrons Deuterium – 1 neutron Tritium – 2 neutrons ...
Early Atomic Theory
... Most of the particles passed through the gold foil, but some were deflected and some even bounced back! This suggested the gold atoms must have a densely, positively charged nucleus to affect the path of an α particle (a positively charged He atom). ...
... Most of the particles passed through the gold foil, but some were deflected and some even bounced back! This suggested the gold atoms must have a densely, positively charged nucleus to affect the path of an α particle (a positively charged He atom). ...
Mystery Isotopes
... Teaching Phase: How will the teacher present the concept or skill to students? Teacher will discuss atoms with students. "Atoms are the smallest unit of matter...but what are the parts of an atom? How are they arranged? Let's draw a Bohr's model of the isotope Oxygen-18." Teacher will model how to a ...
... Teaching Phase: How will the teacher present the concept or skill to students? Teacher will discuss atoms with students. "Atoms are the smallest unit of matter...but what are the parts of an atom? How are they arranged? Let's draw a Bohr's model of the isotope Oxygen-18." Teacher will model how to a ...
Atomic Structure
... By adding an electric field he found that the moving pieces were negative Also placed a paddle wheel in the center – and it turned! So they must have mass! ...
... By adding an electric field he found that the moving pieces were negative Also placed a paddle wheel in the center – and it turned! So they must have mass! ...
Nuclear Chemistry
... Nuclear Chemistry The strong force, also known as strong nuclear force, is stronger than the electrostatic repulsion. It holds the nucleus together. ...
... Nuclear Chemistry The strong force, also known as strong nuclear force, is stronger than the electrostatic repulsion. It holds the nucleus together. ...
Henry Moseley, the Atomic Number, and Synthesis
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
EXPERIMENT 4 – The Periodic Table
... The sum of the number of protons and neutrons in the nucleus of an atom is called the mass number (A) of the atom (remember A for “all”): Mass Number = Number of Protons + Number of Neutrons Protons attract the electrons because they have opposite charges. In a neutral atom, the number of protons is ...
... The sum of the number of protons and neutrons in the nucleus of an atom is called the mass number (A) of the atom (remember A for “all”): Mass Number = Number of Protons + Number of Neutrons Protons attract the electrons because they have opposite charges. In a neutral atom, the number of protons is ...
Henry Moseley, the Atomic Number, and Synthesis
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
EXPERIMENT 4 – The Periodic Table
... The sum of the number of protons and neutrons in the nucleus of an atom is called the mass number (A) of the atom (remember A for “all”): Mass Number = Number of Protons + Number of Neutrons Protons attract the electrons because they have opposite charges. In a neutral atom, the number of protons is ...
... The sum of the number of protons and neutrons in the nucleus of an atom is called the mass number (A) of the atom (remember A for “all”): Mass Number = Number of Protons + Number of Neutrons Protons attract the electrons because they have opposite charges. In a neutral atom, the number of protons is ...
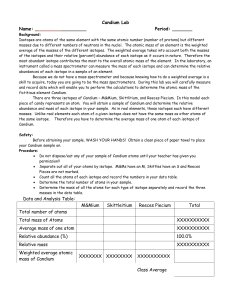
Chapter 18 Comparing Atoms Lab
... many marbles, and of as many colors as they need but must take at least as many total marbles as they put in. For example, a player can trade 2 yellows for 1 yellow, 1 blue, and 1 red. 11. Which particles are found in an atom’s nucleus? Which particles are found outside the nucleus? 12. What four el ...
... many marbles, and of as many colors as they need but must take at least as many total marbles as they put in. For example, a player can trade 2 yellows for 1 yellow, 1 blue, and 1 red. 11. Which particles are found in an atom’s nucleus? Which particles are found outside the nucleus? 12. What four el ...
Masses of Atoms
... Atomic Number ~ number of protons in the atom of an element Atomic Mass ~ number of neutrons AND number of protons Isotope ~ atoms of the same element, with different numbers of neutrons Carbon - 12 (6 protons, 6 neutrons) Carbon - 14 (6 protons, 8 neutrons) ...
... Atomic Number ~ number of protons in the atom of an element Atomic Mass ~ number of neutrons AND number of protons Isotope ~ atoms of the same element, with different numbers of neutrons Carbon - 12 (6 protons, 6 neutrons) Carbon - 14 (6 protons, 8 neutrons) ...
Unit 3 Notes only
... These elements are called: RADIOACTIVE When these elements decay their nucleus changes into another element and they give off RADIATION. ...
... These elements are called: RADIOACTIVE When these elements decay their nucleus changes into another element and they give off RADIATION. ...
Atomic Structure
... 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element. ...
... 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element. ...
Atomic Structure Notepacket
... But suppose I have a recipe to make a compound. I need 100 hydrogen atoms and 50 oxygen atoms-you cannot count atoms or pluck them out with atomic tweezers! So instead they mass them(weigh them) Qt. What accounts for the mass of the atom? ...
... But suppose I have a recipe to make a compound. I need 100 hydrogen atoms and 50 oxygen atoms-you cannot count atoms or pluck them out with atomic tweezers! So instead they mass them(weigh them) Qt. What accounts for the mass of the atom? ...
Calculating the number of Protons, Neutrons, and Electrons.
... Atomic Number = Protons and Electron Calculate Neutrons = Rounded Atomic Mass – Atomic Number EXAMPLE: 15.999 rounds to 16 so ...
... Atomic Number = Protons and Electron Calculate Neutrons = Rounded Atomic Mass – Atomic Number EXAMPLE: 15.999 rounds to 16 so ...
11129_evl_ch1_ste_corr
... electron shells. Some of them (boron, nitrogen, fluorine and neon) have two electron shells; others (sodium and magnesium) have three. ...
... electron shells. Some of them (boron, nitrogen, fluorine and neon) have two electron shells; others (sodium and magnesium) have three. ...
Chapter 17 - murraysphysical
... Name of element followed by mass number identifies the isotopes. ...
... Name of element followed by mass number identifies the isotopes. ...
The History of the Periodic Table
... other workers had reported. For the rest of the 19th century, atomic masses were continually revised and improved and new elements were rapidly being discovered By 1817, it was recognized that some elements could be placed into groups, using their physical and chemical properties. Elements with clos ...
... other workers had reported. For the rest of the 19th century, atomic masses were continually revised and improved and new elements were rapidly being discovered By 1817, it was recognized that some elements could be placed into groups, using their physical and chemical properties. Elements with clos ...
Atoms
... • It cannot be broken down any further without a chemical or nuclear reaction. – It will then become a different substance. • Uranium in a nuclear bomb ...
... • It cannot be broken down any further without a chemical or nuclear reaction. – It will then become a different substance. • Uranium in a nuclear bomb ...
atoms - Fort Bend ISD
... Sizing up the Atom Elements are able to be subdivided into smaller and smaller particles – these are the atoms, and they still have properties of that element If you could line up 100,000,000 copper atoms in a single file, they would be approximately 1 cm long Despite their small size, individu ...
... Sizing up the Atom Elements are able to be subdivided into smaller and smaller particles – these are the atoms, and they still have properties of that element If you could line up 100,000,000 copper atoms in a single file, they would be approximately 1 cm long Despite their small size, individu ...
Test Review Chapter 1
... b. atomic number. c. the number of electrons in the highest energy level. d. the total number of electrons. ____ 30. The atomic number of oxygen, 8, indicates that there are eight a. protons in the nucleus of an oxygen atom. b. oxygen nuclides. c. neutrons outside the oxygen atom's nucleus. d. energ ...
... b. atomic number. c. the number of electrons in the highest energy level. d. the total number of electrons. ____ 30. The atomic number of oxygen, 8, indicates that there are eight a. protons in the nucleus of an oxygen atom. b. oxygen nuclides. c. neutrons outside the oxygen atom's nucleus. d. energ ...
SUBATOMIC PARTICLES The three main subatomic particles found
... of rare isotopes that may not be included in the percentages when calculating atomic mass. ...
... of rare isotopes that may not be included in the percentages when calculating atomic mass. ...
Isotopes
... ESQ: How are isotopes similar? Different? How do you calculate the average atomic mass of an isotope? ...
... ESQ: How are isotopes similar? Different? How do you calculate the average atomic mass of an isotope? ...
Einsteinium

Einsteinium is a synthetic element with symbol Es and atomic number 99. It is the seventh transuranic element, and an actinide.Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952, and named after Albert Einstein. Its most common isotope einsteinium-253 (half life 20.47 days) is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but at much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside of basic scientific research. In particular, einsteinium was used to synthesize, for the first time, 17 atoms of the new element mendelevium in 1955.Einsteinium is a soft, silvery, paramagnetic metal. Its chemistry is typical of the late actinides, with a preponderance of the +3 oxidation state; the +2 oxidation state is also accessible, especially in solids. The high radioactivity of einsteinium-253 produces a visible glow and rapidly damages its crystalline metal lattice, with released heat of about 1000 watts per gram. Difficulty in studying its properties is due to einsteinium-253's conversion to berkelium and then californium at a rate of about 3% per day. The isotope of einsteinium with the longest half life, einsteinium-252 (half life 471.7 days) would be more suitable for investigation of physical properties, but it has proven far more difficult to produce and is available only in minute quantities, and not in bulk. Einsteinium is the element with the highest atomic number which has been observed in macroscopic quantities in its pure form, and this was the common short-lived isotope einsteinium-253.Like all synthetic transuranic elements, isotopes of einsteinium are very radioactive and are considered highly dangerous to health on ingestion.