SNC1D- Grade 9- Unit: Chemistry March 03,2009 Periodic Table
... They have a single valence electron which they give up easily, forming ions with a 1+ charge. Hydrogen is generally included in this group, because it has a single valence electron. However, it does not have any of the other metallic properties, and generally behaves as a nonmetal when forming compo ...
... They have a single valence electron which they give up easily, forming ions with a 1+ charge. Hydrogen is generally included in this group, because it has a single valence electron. However, it does not have any of the other metallic properties, and generally behaves as a nonmetal when forming compo ...
7th Grade Atomic Structure and Periodic Table of Elements
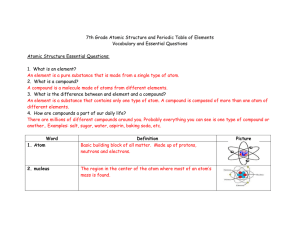
... A compound is a molecule made of atoms from different elements. 3. What is the difference between and element and a compound? An element is a substance that contains only one type of atom. A compound is composed of more than one atom of different elements. 4. How are compounds a part of our daily li ...
... A compound is a molecule made of atoms from different elements. 3. What is the difference between and element and a compound? An element is a substance that contains only one type of atom. A compound is composed of more than one atom of different elements. 4. How are compounds a part of our daily li ...
Chapter 4 Atomic Structure
... identical. Atoms of any one element are different from those of any other element. ...
... identical. Atoms of any one element are different from those of any other element. ...
Lesson 1 & 2 Periodic table trends and formation
... element, averaged over all the isotopes of the element. (Note: students may not have studied isotopes yet, and may not be ready to grapple with the distinction between atomic mass and mass number. At this point it is sufficient that they simply understand atomic mass as resulting from the number of ...
... element, averaged over all the isotopes of the element. (Note: students may not have studied isotopes yet, and may not be ready to grapple with the distinction between atomic mass and mass number. At this point it is sufficient that they simply understand atomic mass as resulting from the number of ...
Atomic number, atomic mass and isotopes
... Appendix B and check out the atomic numbers and atomic masses of your favourite elements. Isotopes. Isotopes are atoms of the same element that have different atomic masses; they have the same number of protons, but different numbers of neutrons. Look at the element chlorine in Appendix B. Its atomi ...
... Appendix B and check out the atomic numbers and atomic masses of your favourite elements. Isotopes. Isotopes are atoms of the same element that have different atomic masses; they have the same number of protons, but different numbers of neutrons. Look at the element chlorine in Appendix B. Its atomi ...
Document
... Democritus and the Atom section and explain how the student in figure 3 is illustrating Democritus’s ...
... Democritus and the Atom section and explain how the student in figure 3 is illustrating Democritus’s ...
Atomic Structure Notes file
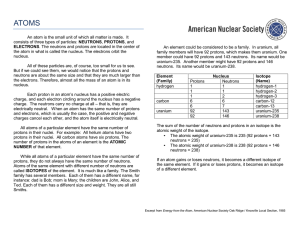
... The atoms of an element can differ in mass from each other because they have differing numbers of neutrons. Those with more neutrons will weigh more and be more massive. The atomic mass (often referred to as atomic weight) of an element is calculated by adding together the number of protons and the ...
... The atoms of an element can differ in mass from each other because they have differing numbers of neutrons. Those with more neutrons will weigh more and be more massive. The atomic mass (often referred to as atomic weight) of an element is calculated by adding together the number of protons and the ...
04 Atom notes
... Circle the letter of each sentence that is true about the nuclear theory of atoms suggested by Rutherford’s experimental results. a. An atom is mostly empty space. b. All the positive charge of an atom is concentrated in a small central region called the nucleus. c. The nucleus is composed of proton ...
... Circle the letter of each sentence that is true about the nuclear theory of atoms suggested by Rutherford’s experimental results. a. An atom is mostly empty space. b. All the positive charge of an atom is concentrated in a small central region called the nucleus. c. The nucleus is composed of proton ...
Chapter 4 Atomic Structure
... identical. Atoms of any one element are different from those of any other element. ...
... identical. Atoms of any one element are different from those of any other element. ...
An atom is the small unit of which all matter is made. It consists of
... the atom in what is called the nucleus. The electrons orbit the nucleus. All of these particles are, of course, too small for us to see. But if we could see them, we would notice that the protons and neutrons are about the same size and that they are much larger than the electrons. Therefore, almost ...
... the atom in what is called the nucleus. The electrons orbit the nucleus. All of these particles are, of course, too small for us to see. But if we could see them, we would notice that the protons and neutrons are about the same size and that they are much larger than the electrons. Therefore, almost ...
File - GarzScience!
... • A weighted average mass of the atoms in a naturally occurring sample of the element • Mass of an atom in atomic mass units (amu) • Equal to 1/12th of the mass of carbon • Weighted average mass reflects both the mass and the relative abundance of the isotopes as they occur in nature • This is the n ...
... • A weighted average mass of the atoms in a naturally occurring sample of the element • Mass of an atom in atomic mass units (amu) • Equal to 1/12th of the mass of carbon • Weighted average mass reflects both the mass and the relative abundance of the isotopes as they occur in nature • This is the n ...
Atoms - Chemistry Land
... or location is quite flexible. In this shape there are two lobes. The electron occupies both. Somehow is gets back and forth without passing through the middle space. Or it is in both locations at the same time. Either way it is not a simple particle. Also, it’s location is not fixed. The probabilit ...
... or location is quite flexible. In this shape there are two lobes. The electron occupies both. Somehow is gets back and forth without passing through the middle space. Or it is in both locations at the same time. Either way it is not a simple particle. Also, it’s location is not fixed. The probabilit ...
mass number - KCPE-KCSE
... mass of one atom of carbon-12 Most elements have more than one isotope. The Ar of the element is the average mass of the isotopes taking into account the abundance of each isotope. This is why the Ar of an element is frequently not a whole number. ...
... mass of one atom of carbon-12 Most elements have more than one isotope. The Ar of the element is the average mass of the isotopes taking into account the abundance of each isotope. This is why the Ar of an element is frequently not a whole number. ...
Atoms! - Holtmeyerhouse
... they are like the glue helping hold together. When get too many protons ( above 83) not enough neutrons to hold. So all elements above 83 are radioactive. (state some) Also isotopes of stable atoms that have too many neutrons makes an atom unstable! ...
... they are like the glue helping hold together. When get too many protons ( above 83) not enough neutrons to hold. So all elements above 83 are radioactive. (state some) Also isotopes of stable atoms that have too many neutrons makes an atom unstable! ...
In actual laboratories, isotopes in a sample can be
... Background: Elements are composed of atoms. These atoms are composed of protons, neutrons, and electrons. Essentially, the protons and neutrons that are present in the nucleus determine the mass of an atom. The mass of the electron is so small that chemists generally ignore it in most applications. ...
... Background: Elements are composed of atoms. These atoms are composed of protons, neutrons, and electrons. Essentially, the protons and neutrons that are present in the nucleus determine the mass of an atom. The mass of the electron is so small that chemists generally ignore it in most applications. ...
ON THE INSIDE
... AGreek philosopher called Democritus, who lived over 2000 years ago, taught people that all things were made of grains which could not be divided. He called these grains atoms because in Greek atom means indivisible. Today, atom is the common name for the tiny particles of matter that cannot be furt ...
... AGreek philosopher called Democritus, who lived over 2000 years ago, taught people that all things were made of grains which could not be divided. He called these grains atoms because in Greek atom means indivisible. Today, atom is the common name for the tiny particles of matter that cannot be furt ...
Defining the Atom
... 1. Isotopes of an element have a) the same mass number. b) different atomic numbers. c) the same number of protons but different numbers of neutrons. d) the same number of protons but different numbers of electrons. ...
... 1. Isotopes of an element have a) the same mass number. b) different atomic numbers. c) the same number of protons but different numbers of neutrons. d) the same number of protons but different numbers of electrons. ...
Any substance that cannot be decomposed into
... found either chemically free, such as the oxygen in air, or combined with other elements, such as the hydrogen and oxygen in water. About 20 additional elements have been produced in the laboratory through the techniques of nuclear physics. (See also atomic particles; chemistry.) Some substances now ...
... found either chemically free, such as the oxygen in air, or combined with other elements, such as the hydrogen and oxygen in water. About 20 additional elements have been produced in the laboratory through the techniques of nuclear physics. (See also atomic particles; chemistry.) Some substances now ...
IPS Unit 2 Worksheet Packet
... ____ 11. An atom has a small, positively charged nucleus surrounded by a large region in which scientists can predict where an electron is likely to be found. ____ 12. An atom is made of positively charged, pudding like material through which negatively charged particles are scattered. ____ 13. In a ...
... ____ 11. An atom has a small, positively charged nucleus surrounded by a large region in which scientists can predict where an electron is likely to be found. ____ 12. An atom is made of positively charged, pudding like material through which negatively charged particles are scattered. ____ 13. In a ...
Chapter 4 Atomic Structure
... compound, the masses of one element combined with a fixed mass of the second are in the ratio of small whole numbers. Same elements to combine in different ratios to give different substances. ...
... compound, the masses of one element combined with a fixed mass of the second are in the ratio of small whole numbers. Same elements to combine in different ratios to give different substances. ...
Einsteinium

Einsteinium is a synthetic element with symbol Es and atomic number 99. It is the seventh transuranic element, and an actinide.Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952, and named after Albert Einstein. Its most common isotope einsteinium-253 (half life 20.47 days) is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but at much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside of basic scientific research. In particular, einsteinium was used to synthesize, for the first time, 17 atoms of the new element mendelevium in 1955.Einsteinium is a soft, silvery, paramagnetic metal. Its chemistry is typical of the late actinides, with a preponderance of the +3 oxidation state; the +2 oxidation state is also accessible, especially in solids. The high radioactivity of einsteinium-253 produces a visible glow and rapidly damages its crystalline metal lattice, with released heat of about 1000 watts per gram. Difficulty in studying its properties is due to einsteinium-253's conversion to berkelium and then californium at a rate of about 3% per day. The isotope of einsteinium with the longest half life, einsteinium-252 (half life 471.7 days) would be more suitable for investigation of physical properties, but it has proven far more difficult to produce and is available only in minute quantities, and not in bulk. Einsteinium is the element with the highest atomic number which has been observed in macroscopic quantities in its pure form, and this was the common short-lived isotope einsteinium-253.Like all synthetic transuranic elements, isotopes of einsteinium are very radioactive and are considered highly dangerous to health on ingestion.