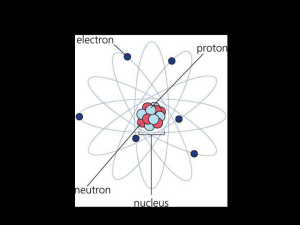
Atoms
... The element antimony (Sb) has naturally occurring isotopes with mass numbers of 121 and 123. The relative abundance and atomic masses are 57.12 % for mass = 120.90 amu, and 47.29% for mass = 122.90 amu. Calculate the atomic mass of antimony. ...
... The element antimony (Sb) has naturally occurring isotopes with mass numbers of 121 and 123. The relative abundance and atomic masses are 57.12 % for mass = 120.90 amu, and 47.29% for mass = 122.90 amu. Calculate the atomic mass of antimony. ...
The Modern Atomic Model
... • A Proton and a Neutron have about the same mass (1 amu). • Electrons mass are around 2,000 times less than protons and neutrons. • Protons and Neutrons contribute to most of the atom’s mass. ...
... • A Proton and a Neutron have about the same mass (1 amu). • Electrons mass are around 2,000 times less than protons and neutrons. • Protons and Neutrons contribute to most of the atom’s mass. ...
The atomic number tells how many protons Protons make an atom
... Atoms of the same element with different atomic masses are called isotopes Gold has a mass of 196.97 That means MOST gold atoms have 197 p+ and no, but some rare atoms will have only 196. They ALL have 79 p+. Most have 118no, but a few may have 117 no. ...
... Atoms of the same element with different atomic masses are called isotopes Gold has a mass of 196.97 That means MOST gold atoms have 197 p+ and no, but some rare atoms will have only 196. They ALL have 79 p+. Most have 118no, but a few may have 117 no. ...
Chapter 4 Atomic Structure
... across it from element to element. Each vertical column of the periodic table is called a group, or family. Elements within a group have similar chemical and physical properties. ...
... across it from element to element. Each vertical column of the periodic table is called a group, or family. Elements within a group have similar chemical and physical properties. ...
Mixtures, Pure Substance and Isotopes
... • Two main types of Isotopes are stable or unstable • Unstable Isotopes: known as radioisotopes and are subject to radioactive decay. – This ‘decay’ means that over time, these isotopes turn into another isotope of the same element ...
... • Two main types of Isotopes are stable or unstable • Unstable Isotopes: known as radioisotopes and are subject to radioactive decay. – This ‘decay’ means that over time, these isotopes turn into another isotope of the same element ...
Atomic Theory PPT
... atoms All atoms of the same element are identical in mass and properties All atoms of different elements are different in mass and properties ...
... atoms All atoms of the same element are identical in mass and properties All atoms of different elements are different in mass and properties ...
The Atom: Idea to Theory
... – Atoms of any one element differ in properties from atoms of another element Mullis ...
... – Atoms of any one element differ in properties from atoms of another element Mullis ...
CHAPTER 2: ATOMS, IONS, AND COMPOUNDS
... called atomos (meaning “not to cut) or atoms. Empedocles (490-430 B.C.): suggested all matter was composed of four basic elements: air, water, fire, and earth. Aristotle (384-321 B.C.): accepted Empedocles idea and added a fifth element, heavenly ether, which is perfect, eternal, and incorruptible. ...
... called atomos (meaning “not to cut) or atoms. Empedocles (490-430 B.C.): suggested all matter was composed of four basic elements: air, water, fire, and earth. Aristotle (384-321 B.C.): accepted Empedocles idea and added a fifth element, heavenly ether, which is perfect, eternal, and incorruptible. ...
Document
... Dalton’s Atomic Theory - Summary 1. matter is composed, indivisible particles (atoms) 2. all atoms of a particular element are identical 3. different elements have different atoms 4. atoms combine in certain whole-number ratios 5. In a chemical reaction, atoms are merely rearranged to form new comp ...
... Dalton’s Atomic Theory - Summary 1. matter is composed, indivisible particles (atoms) 2. all atoms of a particular element are identical 3. different elements have different atoms 4. atoms combine in certain whole-number ratios 5. In a chemical reaction, atoms are merely rearranged to form new comp ...
Atomic Structure and Isotopes
... Dalton’s Atomic Theory: > All elements are composed of atoms. > Atoms of the same elements are the same. > Atoms of different elements can combine together to make compounds. > Chemical reactions occur when atoms are separated, joined, or rearranged. ...
... Dalton’s Atomic Theory: > All elements are composed of atoms. > Atoms of the same elements are the same. > Atoms of different elements can combine together to make compounds. > Chemical reactions occur when atoms are separated, joined, or rearranged. ...
atoms - s3.amazonaws.com
... identical. Atoms of any one element are different from those of any other element. ...
... identical. Atoms of any one element are different from those of any other element. ...
Practice problems for chapter 1, 3 and 5 1) A small amount of salt
... 1) A small amount of salt dissolved in water is an example of a __________. 2) Which one of the following is a pure substance? A) concrete B) wood C) salt water D) elemental copper E) milk 3) For which of the following can the composition vary? A) pure substance B) element C) both homogeneous and he ...
... 1) A small amount of salt dissolved in water is an example of a __________. 2) Which one of the following is a pure substance? A) concrete B) wood C) salt water D) elemental copper E) milk 3) For which of the following can the composition vary? A) pure substance B) element C) both homogeneous and he ...
1030133Notes 4.3
... Isotopes Atoms of the same element with a different number of neutrons (different mass #) Still have same # of protons (that determines the element) Same atomic number ...
... Isotopes Atoms of the same element with a different number of neutrons (different mass #) Still have same # of protons (that determines the element) Same atomic number ...
atoms - KMKunz
... • All atoms of a given element are alike in mass and other properties, but atoms of one element differ from the atoms of every other element • Compounds are formed when atoms of different elements unite in fixed proportions • A chemical reaction involves a rearrangement of atoms. No atoms are create ...
... • All atoms of a given element are alike in mass and other properties, but atoms of one element differ from the atoms of every other element • Compounds are formed when atoms of different elements unite in fixed proportions • A chemical reaction involves a rearrangement of atoms. No atoms are create ...
Trends in the Periodic Table
... 1. __________________ An atom of this element has 1 proton and 1 electron 2. __________________ An atom of this element has 8 protons 3. __________________ An atom of this element has 10 more protons than an atom of oxygen 4. __________________ This element has an atomic number of 12 5. ____________ ...
... 1. __________________ An atom of this element has 1 proton and 1 electron 2. __________________ An atom of this element has 8 protons 3. __________________ An atom of this element has 10 more protons than an atom of oxygen 4. __________________ This element has an atomic number of 12 5. ____________ ...
Atoms - SD308.org
... Using experimental methods, Dalton transformed Democritus’s ideas on atoms into a scientific theory 1. All matter is composed of tiny indivisible particles called atoms 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element. 3. At ...
... Using experimental methods, Dalton transformed Democritus’s ideas on atoms into a scientific theory 1. All matter is composed of tiny indivisible particles called atoms 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element. 3. At ...
Distinguishing Among Atoms Worksheet
... 13. Is the following sentence true or false? The atomic mass of an element is always a whole number of atomic mass units. ___________________ 14. Circle the letter of each statement that is true about the average atomic mass of an element and the relative abundance of its isotopes. a. In nature, mos ...
... 13. Is the following sentence true or false? The atomic mass of an element is always a whole number of atomic mass units. ___________________ 14. Circle the letter of each statement that is true about the average atomic mass of an element and the relative abundance of its isotopes. a. In nature, mos ...
Atomic Mass and Isotopes
... If we know there are different Isotopes of 1 element, how can we arrange the elements on the periodic table by mass correctly? Remember when we didn’t mass our filter paper in the Separation Lab? How did we compensate for the fact that our filter paper would be similar to another sheet of filter ...
... If we know there are different Isotopes of 1 element, how can we arrange the elements on the periodic table by mass correctly? Remember when we didn’t mass our filter paper in the Separation Lab? How did we compensate for the fact that our filter paper would be similar to another sheet of filter ...
Everything is made of atoms.
... * There are actually a lot more known subatomic particles than those listed here ...
... * There are actually a lot more known subatomic particles than those listed here ...
atom atomic symbol atomic number # protons atomic mass
... (2) 5 pts - Refer to a Periodic Table and the Key below to fill out this table for each element. Turn Lithium atom into an ion and note the information. Turn Beryllium into an isotope and record what you did. ...
... (2) 5 pts - Refer to a Periodic Table and the Key below to fill out this table for each element. Turn Lithium atom into an ion and note the information. Turn Beryllium into an isotope and record what you did. ...
Chapter 4
... physically mix together or can chemically combine in simple wholenumber ratios to form compounds. Chemical reactions occur when atoms are separated, joined or rearranged. Atoms of one element can never be changed into atoms of another element as a result of a chemical reaction. ...
... physically mix together or can chemically combine in simple wholenumber ratios to form compounds. Chemical reactions occur when atoms are separated, joined or rearranged. Atoms of one element can never be changed into atoms of another element as a result of a chemical reaction. ...
Oct 14th ,2015
... (Pick up periodic table up front) Objectives: St 4: Matter: Understand the structure of an atom in terms of its subatomic particles; isotopes and ions; differentiate between the classification and separation of matter (mixtures, pure substances…) ...
... (Pick up periodic table up front) Objectives: St 4: Matter: Understand the structure of an atom in terms of its subatomic particles; isotopes and ions; differentiate between the classification and separation of matter (mixtures, pure substances…) ...
Section 6.2 Notes - oologah.k12.ok.us
... 4. Chemical reactions occur when atoms are separated, joined, or rearranged. Atoms of one element are never changed into atoms of another element as a result of a chemical reaction ...
... 4. Chemical reactions occur when atoms are separated, joined, or rearranged. Atoms of one element are never changed into atoms of another element as a result of a chemical reaction ...
ATOM ATOMIC SYMBOL ATOMIC NUMBER
... 1. Color in the Molecule Color Key molecules with colored pencils as indicated. 2. Determine the number of elements in each molecule, and write it down. 3. Draw and color the molecule with the correct number of elements. ...
... 1. Color in the Molecule Color Key molecules with colored pencils as indicated. 2. Determine the number of elements in each molecule, and write it down. 3. Draw and color the molecule with the correct number of elements. ...
Einsteinium

Einsteinium is a synthetic element with symbol Es and atomic number 99. It is the seventh transuranic element, and an actinide.Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952, and named after Albert Einstein. Its most common isotope einsteinium-253 (half life 20.47 days) is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but at much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside of basic scientific research. In particular, einsteinium was used to synthesize, for the first time, 17 atoms of the new element mendelevium in 1955.Einsteinium is a soft, silvery, paramagnetic metal. Its chemistry is typical of the late actinides, with a preponderance of the +3 oxidation state; the +2 oxidation state is also accessible, especially in solids. The high radioactivity of einsteinium-253 produces a visible glow and rapidly damages its crystalline metal lattice, with released heat of about 1000 watts per gram. Difficulty in studying its properties is due to einsteinium-253's conversion to berkelium and then californium at a rate of about 3% per day. The isotope of einsteinium with the longest half life, einsteinium-252 (half life 471.7 days) would be more suitable for investigation of physical properties, but it has proven far more difficult to produce and is available only in minute quantities, and not in bulk. Einsteinium is the element with the highest atomic number which has been observed in macroscopic quantities in its pure form, and this was the common short-lived isotope einsteinium-253.Like all synthetic transuranic elements, isotopes of einsteinium are very radioactive and are considered highly dangerous to health on ingestion.