* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download The atomic number tells how many protons Protons make an atom
Survey
Document related concepts
Transcript
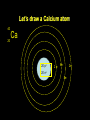
The atomic number tells how many protons Protons make an atom what it is! 79 protons make gold. If I added another proton it would actually turn into Mercury! The atomic number tells how many protons Protons are all the same. If I try to put two together, they push away from each other. It seems that neutrons can act to help glue protons together. The atomic mass tells how many protons AND neutrons there are in the nucleus The number of neutrons do NOT make a difference in the identity of an atom Atoms of the same element will ALWAYS have the same # of protons, but MAY have different neutrons Atoms of the same element with different atomic masses are called isotopes Gold has a mass of 196.97 That means MOST gold atoms have 197 p+ and no, but some rare atoms will have only 196. They ALL have 79 p+. Most have 118no, but a few may have 117 no. In other words, there are two isotopes of gold Atomic mass = p+ + no Atomic # = number of protons If you want to know how many neutrons Atomic mass – atomic # = no Now that we can determine the number of protons, neutrons in the center of the atom, let’s move to the outside of the atom! Electrons travel around the outside of the nucleus in orbitals or shells! Each shell can only hold a certain number of electrons before they are FULL! K = 2 L=8 M=8 N = 18 O = 18 P = 32 Q =32 Let’s draw a Calcium atom 40 Ca 20 20 p+ 2 e- 8e- 2e- 20 no 8e-