Chapter 4- Elements and the Periodic Table
... what atoms are made of hasn't been easy. Because atoms are so small, studying them is a bit like trying to solve the mystery of the sealed box in the Standards Warm-Up activity. Ideas about the shape and structure of atoms have changed many times. The first people to think about the nature of matter ...
... what atoms are made of hasn't been easy. Because atoms are so small, studying them is a bit like trying to solve the mystery of the sealed box in the Standards Warm-Up activity. Ideas about the shape and structure of atoms have changed many times. The first people to think about the nature of matter ...
File
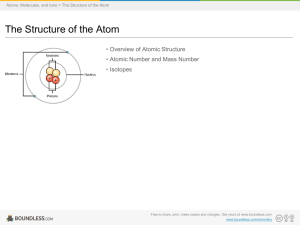
... he propose based on his evidence? Answer: It’s easy to pluck electrons from an atom the further away they are. Solar system model ...
... he propose based on his evidence? Answer: It’s easy to pluck electrons from an atom the further away they are. Solar system model ...
Boundless Study Slides
... • Despite having different numbers of neutrons, isotopes of the same element have very similar physical properties. • Some isotopes are unstable and will undergo radioactive decay to become other elements. • The predictable half-life of different decaying isotopes allows scientists to date material ...
... • Despite having different numbers of neutrons, isotopes of the same element have very similar physical properties. • Some isotopes are unstable and will undergo radioactive decay to become other elements. • The predictable half-life of different decaying isotopes allows scientists to date material ...
Atoms and Molecules
... Q: How is the Periodic Table arranged with respect to the number of protons an atom has? A: The Periodic Table is arranged in increasing Atomic Number, which corresponds to an increasing number of protons in each element. Q: How many elements are there? A: There are 117 known elements. 90 of them ar ...
... Q: How is the Periodic Table arranged with respect to the number of protons an atom has? A: The Periodic Table is arranged in increasing Atomic Number, which corresponds to an increasing number of protons in each element. Q: How many elements are there? A: There are 117 known elements. 90 of them ar ...
Atoms and Molecules
... Note: In 1869, Dmitri Mendeleyev was credited with putting together the Periodic Table of Elements. He listed all of the known elements and grouped them together based on their properties. Mendeleyev was able to organize the table in its present form even though many of the elements hadn’t been disc ...
... Note: In 1869, Dmitri Mendeleyev was credited with putting together the Periodic Table of Elements. He listed all of the known elements and grouped them together based on their properties. Mendeleyev was able to organize the table in its present form even though many of the elements hadn’t been disc ...
The Atoms Family
... red chrome-plated Harley-Davidson. He rides so fast that no one can be sure where he is at any time. Elliott is much smaller than Patty and Nelda and he is always angry because these bigger relatives will not let him in the Arcade. He has a frown on his face, eyes that are squinted with anger, and a ...
... red chrome-plated Harley-Davidson. He rides so fast that no one can be sure where he is at any time. Elliott is much smaller than Patty and Nelda and he is always angry because these bigger relatives will not let him in the Arcade. He has a frown on his face, eyes that are squinted with anger, and a ...
Atomic Mass
... The atomic number tells you the number of protons in one an atom of an element. It also tells you the number of electrons in a neutral atom of that element. The atomic number gives the identity of an element as well as its location on the periodic table. No two different elements will have the same ...
... The atomic number tells you the number of protons in one an atom of an element. It also tells you the number of electrons in a neutral atom of that element. The atomic number gives the identity of an element as well as its location on the periodic table. No two different elements will have the same ...
DEFINING THE ATOM - BradyMathScience
... ________ 13. Dalton theorized that atoms are indivisible and that all atoms of an element are identical. Scientists now know that a. Dalton’s theories are completely correct. b. atoms of an element can have different numbers of protons. c. atoms are all divisible. d. all atoms of an element are not ...
... ________ 13. Dalton theorized that atoms are indivisible and that all atoms of an element are identical. Scientists now know that a. Dalton’s theories are completely correct. b. atoms of an element can have different numbers of protons. c. atoms are all divisible. d. all atoms of an element are not ...
Плеханов В
... isotope fractionation is greater than for Zn isotope [14]. It is shown that hydrogen isotope distribution at thermal desorption from PdHxDy depends on difference between energies of corresponding transient state of atom-atom recombination reaction [15]. At liquid-ice phase transition the intermolecu ...
... isotope fractionation is greater than for Zn isotope [14]. It is shown that hydrogen isotope distribution at thermal desorption from PdHxDy depends on difference between energies of corresponding transient state of atom-atom recombination reaction [15]. At liquid-ice phase transition the intermolecu ...
Unit 2.4 Understanding the Elements Listed on the Periodic Table
... Consider the element helium. Its atomic number is 2, so it has two protons in its nucleus. Its nucleus also contains two neutrons. Since 2 + 2 = 4, we know that the mass number of the helium atom is 4. Finally, the helium atom also contains two electrons since the number of electrons must equal the ...
... Consider the element helium. Its atomic number is 2, so it has two protons in its nucleus. Its nucleus also contains two neutrons. Since 2 + 2 = 4, we know that the mass number of the helium atom is 4. Finally, the helium atom also contains two electrons since the number of electrons must equal the ...
Chapter 17 Resource: Properties of Atoms and the Periodic Table
... You have learned that the mass of atoms is measured in atomic mass units. This unit is too small to use in everyday measurement. It would be simpler to have a number of atoms that would have a mass in grams that is equal to the mass of one atom in atomic mass units. The same number would fit all ele ...
... You have learned that the mass of atoms is measured in atomic mass units. This unit is too small to use in everyday measurement. It would be simpler to have a number of atoms that would have a mass in grams that is equal to the mass of one atom in atomic mass units. The same number would fit all ele ...
Atomic Structure Practice Test
... ____ 10. The smallest unit of an element that can exist either alone or in combination with other such particles of the same or different elements is the a. electron. b. proton. c. neutron. d. atom. ____ 11. The atomic number of oxygen, 8, indicates that there are eight a. protons in the nucleus. c. ...
... ____ 10. The smallest unit of an element that can exist either alone or in combination with other such particles of the same or different elements is the a. electron. b. proton. c. neutron. d. atom. ____ 11. The atomic number of oxygen, 8, indicates that there are eight a. protons in the nucleus. c. ...
Chem101 - Lecture 2 Elements Elements as Pure
... 75.78% of all naturally occurring chlorine, the other has 20 neutrons and represents the remaining 24.22% of all naturally occurring chlorine. - Magnesium (Mg) has three naturally occurring isotopes: one has 12 neutrons and represents 78.99% of all naturally occurring magnesium, another has 13 neutr ...
... 75.78% of all naturally occurring chlorine, the other has 20 neutrons and represents the remaining 24.22% of all naturally occurring chlorine. - Magnesium (Mg) has three naturally occurring isotopes: one has 12 neutrons and represents 78.99% of all naturally occurring magnesium, another has 13 neutr ...
Atomic Structure Practice Test
... ____ 10. The smallest unit of an element that can exist either alone or in combination with other such particles of the same or different elements is the a. electron. b. proton. c. neutron. d. atom. ____ 11. The atomic number of oxygen, 8, indicates that there are eight a. protons in the nucleus. c. ...
... ____ 10. The smallest unit of an element that can exist either alone or in combination with other such particles of the same or different elements is the a. electron. b. proton. c. neutron. d. atom. ____ 11. The atomic number of oxygen, 8, indicates that there are eight a. protons in the nucleus. c. ...
Activity 9 What Determines and Limits an Atom`s Mass?
... The stability of an atom varies with the elements. Light elements become more stable as the atomic mass (the number of nucleons) increases.The most stable element is iron (atomic number 26) with an atomic mass of 56. Elements with larger atomic masses become less stable. In general, elements with nu ...
... The stability of an atom varies with the elements. Light elements become more stable as the atomic mass (the number of nucleons) increases.The most stable element is iron (atomic number 26) with an atomic mass of 56. Elements with larger atomic masses become less stable. In general, elements with nu ...
Unit 2 Atomic Theories and Structures Packet
... 12)______________________ Atoms with the same number of protons and electrons but a different number of neutrons. 13)______________________ This scientist used the oil drop experiment to discover the mass of the electron. 14)______________________ A positively charged particle. 15)_________________ ...
... 12)______________________ Atoms with the same number of protons and electrons but a different number of neutrons. 13)______________________ This scientist used the oil drop experiment to discover the mass of the electron. 14)______________________ A positively charged particle. 15)_________________ ...
Unit 2 - Solon City Schools
... 12)______________________ Atoms with the same number of protons and electrons but a different number of neutrons. 13)______________________ This scientist used the oil drop experiment to discover the mass of the electron. 14)______________________ A positively charged particle. 15)_________________ ...
... 12)______________________ Atoms with the same number of protons and electrons but a different number of neutrons. 13)______________________ This scientist used the oil drop experiment to discover the mass of the electron. 14)______________________ A positively charged particle. 15)_________________ ...
Topic 2.1 Atomic Structure Notes Topic 2.1 Atomic
... Consider an atom that has 9 protons, 9 electrons, and 10 neutrons. What is its atomic number, atomic mass, name, and symbol? Z=9 (atomic number = # protons) A=19 (atomic mass = protons + neutrons) Fluorine-19 (name and mass) ...
... Consider an atom that has 9 protons, 9 electrons, and 10 neutrons. What is its atomic number, atomic mass, name, and symbol? Z=9 (atomic number = # protons) A=19 (atomic mass = protons + neutrons) Fluorine-19 (name and mass) ...
Scandium and Yttrium - Mercyhurst University
... reaction.4 At room temperature, scandium metal turns a light yellow or pink color in air.1 It is not obvious what is causing the color as Sc3+ would be expected to be colorless (white) due to the lack of d-electrons. Purification and Properties The most interesting thing about these two elements is ...
... reaction.4 At room temperature, scandium metal turns a light yellow or pink color in air.1 It is not obvious what is causing the color as Sc3+ would be expected to be colorless (white) due to the lack of d-electrons. Purification and Properties The most interesting thing about these two elements is ...
Word - My eCoach
... d. he saw white light. __D__ 15. Experiments with cathode rays led to the discovery of the a. proton. b. nucleus. c. neutron. d. electron. __A__ 16. Because any element used in the cathode produced electrons, scientists concluded that a. all atoms contained electrons. c. atoms were indivisible. b. o ...
... d. he saw white light. __D__ 15. Experiments with cathode rays led to the discovery of the a. proton. b. nucleus. c. neutron. d. electron. __A__ 16. Because any element used in the cathode produced electrons, scientists concluded that a. all atoms contained electrons. c. atoms were indivisible. b. o ...
Atomic Structure Practice Test
... ____ 16. Because any element used in the cathode produced electrons, scientists concluded that a. all atoms contained electrons. c. atoms were indivisible. b. only metals contained electrons. d. atoms carried a negative charge. ____ 17. If you change the number of protons in an atom, what is affecte ...
... ____ 16. Because any element used in the cathode produced electrons, scientists concluded that a. all atoms contained electrons. c. atoms were indivisible. b. only metals contained electrons. d. atoms carried a negative charge. ____ 17. If you change the number of protons in an atom, what is affecte ...
Atomic Structure Practice Test Multiple Choice Identify the choice
... ____ 16. Because any element used in the cathode produced electrons, scientists concluded that a. all atoms contained electrons. c. atoms were indivisible. b. only metals contained electrons. d. atoms carried a negative charge. ____ 17. If you change the number of protons in an atom, what is affecte ...
... ____ 16. Because any element used in the cathode produced electrons, scientists concluded that a. all atoms contained electrons. c. atoms were indivisible. b. only metals contained electrons. d. atoms carried a negative charge. ____ 17. If you change the number of protons in an atom, what is affecte ...
answer key - El Camino College
... reactions the old bonds between atoms are broken down and new bonds are formed. Atoms, however, can be created or destroyed in nuclear reactions: radioactive decays, nuclear fission and fusion. ...
... reactions the old bonds between atoms are broken down and new bonds are formed. Atoms, however, can be created or destroyed in nuclear reactions: radioactive decays, nuclear fission and fusion. ...
wahideh chemistry eportfolio hw
... medicine, and everyday life. Physical Properties: Sodium is a silvery-white metal with a waxy appearance. It is soft enough to be cut with a knife. The surface is bright and shiny when first cut, but quickly becomes dull as sodium reacts with oxygen in the air. Sodium’s melting point is 208.1°F (97. ...
... medicine, and everyday life. Physical Properties: Sodium is a silvery-white metal with a waxy appearance. It is soft enough to be cut with a knife. The surface is bright and shiny when first cut, but quickly becomes dull as sodium reacts with oxygen in the air. Sodium’s melting point is 208.1°F (97. ...
Atom
... 64. _______________ was the ancient philosopher who first proposed the existence of atoms. 65. _______________ showed that matter is neither ___________________ nor _________________ in chemical reactions, and is known as the “Father of Chemistry.” 66. _______________ created the first atomic theory ...
... 64. _______________ was the ancient philosopher who first proposed the existence of atoms. 65. _______________ showed that matter is neither ___________________ nor _________________ in chemical reactions, and is known as the “Father of Chemistry.” 66. _______________ created the first atomic theory ...
Einsteinium

Einsteinium is a synthetic element with symbol Es and atomic number 99. It is the seventh transuranic element, and an actinide.Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952, and named after Albert Einstein. Its most common isotope einsteinium-253 (half life 20.47 days) is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but at much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside of basic scientific research. In particular, einsteinium was used to synthesize, for the first time, 17 atoms of the new element mendelevium in 1955.Einsteinium is a soft, silvery, paramagnetic metal. Its chemistry is typical of the late actinides, with a preponderance of the +3 oxidation state; the +2 oxidation state is also accessible, especially in solids. The high radioactivity of einsteinium-253 produces a visible glow and rapidly damages its crystalline metal lattice, with released heat of about 1000 watts per gram. Difficulty in studying its properties is due to einsteinium-253's conversion to berkelium and then californium at a rate of about 3% per day. The isotope of einsteinium with the longest half life, einsteinium-252 (half life 471.7 days) would be more suitable for investigation of physical properties, but it has proven far more difficult to produce and is available only in minute quantities, and not in bulk. Einsteinium is the element with the highest atomic number which has been observed in macroscopic quantities in its pure form, and this was the common short-lived isotope einsteinium-253.Like all synthetic transuranic elements, isotopes of einsteinium are very radioactive and are considered highly dangerous to health on ingestion.