Test 2 Review Test 2 Review (15-16)_2
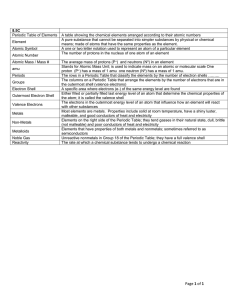
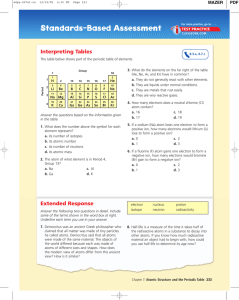
... (18) ____________ How many of these elements are gases at 0 degrees Celsius? (19) ____________ How many of these elements are metalloids? (20) ____________ How many of these elements are NON-metals and solids? (21) ____________ Write the symbol of the element that would have the most similar propert ...
... (18) ____________ How many of these elements are gases at 0 degrees Celsius? (19) ____________ How many of these elements are metalloids? (20) ____________ How many of these elements are NON-metals and solids? (21) ____________ Write the symbol of the element that would have the most similar propert ...
Atomic Structure and the Periodic Table Vocabulary
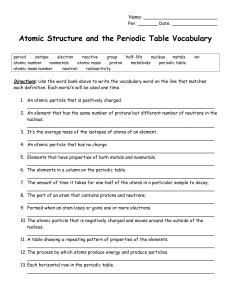
... 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __________________________________________________________________ 4. ...
... 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __________________________________________________________________ 4. ...
CH.2
... differentiate between alpha, beta, and gamma radiation with respect to penetrating power, shielding, and composition. (B2) differentiate between the major atom components (proton, neutron and electron) in terms of location, size, and charge. (B2) distinguish between a group and a period. (B2) ...
... differentiate between alpha, beta, and gamma radiation with respect to penetrating power, shielding, and composition. (B2) differentiate between the major atom components (proton, neutron and electron) in terms of location, size, and charge. (B2) distinguish between a group and a period. (B2) ...
1.2 Atomic Theory
... The average atomic mass for magnesium found on the periodic table is a weighted average of the three isotopes: 24.31 g of Mg Radioactivity: spontaneous decay of nuclei, releasing energy and subatomic particles Radioisotopes: an unstable isotope of an element, which undergoes radioactive decay ...
... The average atomic mass for magnesium found on the periodic table is a weighted average of the three isotopes: 24.31 g of Mg Radioactivity: spontaneous decay of nuclei, releasing energy and subatomic particles Radioisotopes: an unstable isotope of an element, which undergoes radioactive decay ...
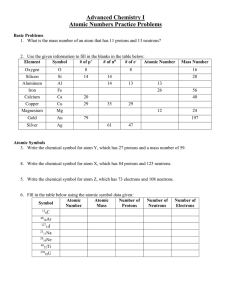
Name Test Review Chemistry Unit 2: The Atom 1. Fill in the blank
... 72.2% and the abundance of X is 27.8%, what is the average atomic mass of X? What is element X? 5. Silicon has three naturally occurring isotopes. What is the average atomic mass of silicon? ...
... 72.2% and the abundance of X is 27.8%, what is the average atomic mass of X? What is element X? 5. Silicon has three naturally occurring isotopes. What is the average atomic mass of silicon? ...
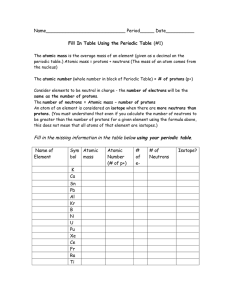
Periodic Table Fill in Table 1
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
Name Test Review Chapters 4 and 25 Honors Chemistry 1. Fill in
... 17. During beta decay, describe what happens to the atomic number of the element. 18. What is the most penetrating form of radioactive particles? What can stop this form of radioactivity? ...
... 17. During beta decay, describe what happens to the atomic number of the element. 18. What is the most penetrating form of radioactive particles? What can stop this form of radioactivity? ...
Ch4StudyGuide
... Although the nucleus is almost 100% of the mass of an atom, what determines the volume? ...
... Although the nucleus is almost 100% of the mass of an atom, what determines the volume? ...
1st Term Review
... 1. Name the scientist(s) responsible for each of the following. You may use a scientist more than once. a) The plum pudding model of the atom b) Modern atomic theory c) Modern periodic law d) Discovery of the nucleus e) Discovery of the neutron f) Gold foil experiment g) Father of the modern periodi ...
... 1. Name the scientist(s) responsible for each of the following. You may use a scientist more than once. a) The plum pudding model of the atom b) Modern atomic theory c) Modern periodic law d) Discovery of the nucleus e) Discovery of the neutron f) Gold foil experiment g) Father of the modern periodi ...
Chemistry Review: Antoine Lavoisier (1743
... Isotopes: atoms of the same element that differs in mass. Ex there are 3 isotopes of hydrogen, with mass numbers of 1, 2 and 3. These atoms have different masses because they have different numbers of neutrons. However, since they have the same number of protons, they are the same element. Most el ...
... Isotopes: atoms of the same element that differs in mass. Ex there are 3 isotopes of hydrogen, with mass numbers of 1, 2 and 3. These atoms have different masses because they have different numbers of neutrons. However, since they have the same number of protons, they are the same element. Most el ...
AP Chem Test 5-7 Practice Exam - mvhs
... 2. The ΔH for the exothermic solution process when solid sodium hydroxide dissolves in water is 44.4 kJ/mol. When a 13.9-g sample of NaOH dissolves in 250.0 g of water in a coffee-cup calorimeter, the temperature change is _______. Assume that the solution has the same specific heat as liquid water, ...
... 2. The ΔH for the exothermic solution process when solid sodium hydroxide dissolves in water is 44.4 kJ/mol. When a 13.9-g sample of NaOH dissolves in 250.0 g of water in a coffee-cup calorimeter, the temperature change is _______. Assume that the solution has the same specific heat as liquid water, ...
Quiz: The Atom (Open Notes)
... 10. T or F A neutron carries a positive charge. 11. T or F A proton has a charge that is equal in force but opposite in charge to each electron. 12. T or F Protons and electrons are about equal in mass. 13. T or F The mass of an atom depends on the number of protons and neutrons in its nucleus. 14. ...
... 10. T or F A neutron carries a positive charge. 11. T or F A proton has a charge that is equal in force but opposite in charge to each electron. 12. T or F Protons and electrons are about equal in mass. 13. T or F The mass of an atom depends on the number of protons and neutrons in its nucleus. 14. ...
Radioisotopes
... having a different atomic mass (mass number) • Isotopes of an element have nuclei with the same number of protons (the same atomic number) but different numbers of neutrons. • Therefore, isotopes have different mass numbers, which give the total number of nucleons, the number of protons plus neutron ...
... having a different atomic mass (mass number) • Isotopes of an element have nuclei with the same number of protons (the same atomic number) but different numbers of neutrons. • Therefore, isotopes have different mass numbers, which give the total number of nucleons, the number of protons plus neutron ...
Slide 1
... The multimedia approach of the standards is refreshing, and the focus on giving students a clear way to access the vocabulary of the standard is essential. This work reflects the new paradigms of teaching and learning that is being infused into educational classrooms of the 21st century. ...
... The multimedia approach of the standards is refreshing, and the focus on giving students a clear way to access the vocabulary of the standard is essential. This work reflects the new paradigms of teaching and learning that is being infused into educational classrooms of the 21st century. ...
Quiz review
... Horizontal rows of the periodic table are called this. Vertical columns of the periodic table are called ‘groups’ or this. Which element in period 3 has 6 valence electrons? Which element in period 5 has only 1 electron in its 5s sublevel? Which element in period 3 has a full octet? What family of e ...
... Horizontal rows of the periodic table are called this. Vertical columns of the periodic table are called ‘groups’ or this. Which element in period 3 has 6 valence electrons? Which element in period 5 has only 1 electron in its 5s sublevel? Which element in period 3 has a full octet? What family of e ...
Atomic Numbers Practice Problems
... 3. Write the chemical symbol for atom Y, which has 27 protons and a mass number of 59. ...
... 3. Write the chemical symbol for atom Y, which has 27 protons and a mass number of 59. ...
Section 1 Review
... and that is located in the nucleus of an atom proton a subatomic particle that has a positive charge and that is located in the nucleus of an atom; the number of protons of the nucleus is the atomic number, which determines the identity of an element ...
... and that is located in the nucleus of an atom proton a subatomic particle that has a positive charge and that is located in the nucleus of an atom; the number of protons of the nucleus is the atomic number, which determines the identity of an element ...
Ch. 2 note packet
... CHAPTER #2 - Atoms, Molecules, and Ions 2.1 The Early History of Chemistry Ancient Greeks - thought matter was composed to 4 substances – earth, air, fire, water (_________ vs. _________) Alchemy - ______________________________ (discovered many elements; learned to prepare mineral acids) Metallurgy ...
... CHAPTER #2 - Atoms, Molecules, and Ions 2.1 The Early History of Chemistry Ancient Greeks - thought matter was composed to 4 substances – earth, air, fire, water (_________ vs. _________) Alchemy - ______________________________ (discovered many elements; learned to prepare mineral acids) Metallurgy ...
Atomic Structure - hrsbstaff.ednet.ns.ca
... So, what’s up with all these isotopes anyway? In nature elements are not made up of atoms that are all exactly the same! Some will be heavier than others, even though they are still the same type of atom. C-12 and C-14 are both Carbon, with all the usual Carbon properties, but the C-14 has two more ...
... So, what’s up with all these isotopes anyway? In nature elements are not made up of atoms that are all exactly the same! Some will be heavier than others, even though they are still the same type of atom. C-12 and C-14 are both Carbon, with all the usual Carbon properties, but the C-14 has two more ...
Pre-Knowledge: Chemistry and Physics Vocabulary Atomic Number
... element will, by definition, have the same number of protons in their nuclei but can vary in the number of neutrons. Since neutrons do not carry any charge, adding or subtracting them from the nucleus of an atom only affects the mass of the atom. Isotopes of an element will have identical or closely ...
... element will, by definition, have the same number of protons in their nuclei but can vary in the number of neutrons. Since neutrons do not carry any charge, adding or subtracting them from the nucleus of an atom only affects the mass of the atom. Isotopes of an element will have identical or closely ...
Page 233 - ClassZone
... the world differed because each was made of atoms of different sizes and shapes. How does the modern view of atoms differ from this ancient view? How is it similar? ...
... the world differed because each was made of atoms of different sizes and shapes. How does the modern view of atoms differ from this ancient view? How is it similar? ...
L.O.
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
The Atom Chapter 2
... • Heteronuclear Diatomic Molecule: a molecule made of two atoms that are different elements – NO ...
... • Heteronuclear Diatomic Molecule: a molecule made of two atoms that are different elements – NO ...
Lawrencium

Lawrencium is a synthetic chemical element with chemical symbol Lr (formerly Lw) and atomic number 103. It is named in honor of Ernest Lawrence, inventor of the cyclotron, a device that was used to discover many artificial radioactive elements. A radioactive metal, lawrencium is the eleventh transuranic element and is also the final member of the actinide series. Like all elements with atomic number over 100, lawrencium can only be produced in particle accelerators by bombarding lighter elements with charged particles. Twelve isotopes of lawrencium are currently known; the most stable is 266Lr with a half-life of 11 hours, but the shorter-lived 260Lr (half-life 2.7 minutes) is most commonly used in chemistry because it can be produced on a larger scale.Chemistry experiments have confirmed that lawrencium indeed behaves as a heavier homolog to lutetium in the periodic table, and is a trivalent element. It thus could also be classified as the first of the 7th-period transition metals: however, its electron configuration is anomalous for its position in the periodic table, having an s2p configuration instead of the s2d configuration of its homolog lutetium. This means that lawrencium may be less volatile than expected for its position in the periodic table and have a volatility comparable to that of lead.In the 1950s, 1960s, and 1970s, many claims of the synthesis of lawrencium of varying quality were made from laboratories in the Soviet Union and the United States. The priority of the discovery and therefore the naming of the element was disputed between Soviet and American scientists, and while the International Union of Pure and Applied Chemistry (IUPAC) established lawrencium as the official name for the element and gave the American team credit for the discovery, this was reevaluated in 1997, giving both teams shared credit for the discovery but not changing the element's name.