Chapter 11 and 12-2 Review/Study Guide for Test
... 1. List the major discovery or accomplishment associated with of each of the following scientists: a. Democritus – named the atom, stated it was indivisible b. Dalton – atom is positively charged sphere - said atoms of different elements are different, atoms of the same element are the same c. Thoms ...
... 1. List the major discovery or accomplishment associated with of each of the following scientists: a. Democritus – named the atom, stated it was indivisible b. Dalton – atom is positively charged sphere - said atoms of different elements are different, atoms of the same element are the same c. Thoms ...
Isotopes
... • Atoms of the same element with different atomic masses are called isotopes. • Atoms of the same element have the same properties. • But what causes the chemical properties of an atom? • So atoms of different isotopes of an element must have the same number of protons and electrons. • Whatever cau ...
... • Atoms of the same element with different atomic masses are called isotopes. • Atoms of the same element have the same properties. • But what causes the chemical properties of an atom? • So atoms of different isotopes of an element must have the same number of protons and electrons. • Whatever cau ...
Atomic Number
... -Elements within the same group have similar properties EX. Au, Ag, Cu -Each horizontal row is called a ____________________ -Properties of the elements gradually change when you move through a period -Elements get smaller when you move from _________________ to ______________. ...
... -Elements within the same group have similar properties EX. Au, Ag, Cu -Each horizontal row is called a ____________________ -Properties of the elements gradually change when you move through a period -Elements get smaller when you move from _________________ to ______________. ...
Chapter 18
... same element that have different numbers of neutrons • Average atomic mass—the weighted average mass of an element’s mixture of isotopes (used because most elements have more than one isotope) ...
... same element that have different numbers of neutrons • Average atomic mass—the weighted average mass of an element’s mixture of isotopes (used because most elements have more than one isotope) ...
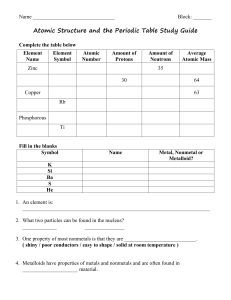
Name: Chapter 4 and 5 Study Guide Who was the Greek
... a. Its atomic mass b. Attractions between its atoms c. Its number of valence electrons d. The ratio of protons and neutrons 26. Which of the halogens is the most reactive? ...
... a. Its atomic mass b. Attractions between its atoms c. Its number of valence electrons d. The ratio of protons and neutrons 26. Which of the halogens is the most reactive? ...
Extension 18.2: Isotopes
... Recall that we have defined the nucleus of an atom by specifying the number of protons, Z (the atomic number) and the number of neutrons, N. These added together give the total number of nucleons in the nucleus, A (the mass number). So, as we have said, A = Z + N. ...
... Recall that we have defined the nucleus of an atom by specifying the number of protons, Z (the atomic number) and the number of neutrons, N. These added together give the total number of nucleons in the nucleus, A (the mass number). So, as we have said, A = Z + N. ...
Extra Credit Test Review
... 14. Elements in a group on the periodic table share similar properties because their atoms have the same number of _______________ in their outer energy levels. ...
... 14. Elements in a group on the periodic table share similar properties because their atoms have the same number of _______________ in their outer energy levels. ...
Atomic Structure ppt
... It is found that the element boron has two isotopes. B-10 (11.811amu) is 19.91% abundant and B-11 (10.845amu) is 80.09% abundant. What is the average atomic mass of B? 10.946 amu ...
... It is found that the element boron has two isotopes. B-10 (11.811amu) is 19.91% abundant and B-11 (10.845amu) is 80.09% abundant. What is the average atomic mass of B? 10.946 amu ...
Atomic Structure Power Point
... All matter is composed of atoms. Atoms cannot be created or destroyed. Atoms of elements combine to form compounds. Each element is made up of different kinds of atoms. ...
... All matter is composed of atoms. Atoms cannot be created or destroyed. Atoms of elements combine to form compounds. Each element is made up of different kinds of atoms. ...
VOCABULARY name, date, hour: Fill in the number of each term
... ___ substance that is a mixture of two or more metals ___ columns of the periodic table; also known as groups ___ number of protons carried by the nucleus of an atom ___ element with an imbalance in the number of neutrons and protons ___ uncharged particle found in the nucleus of an atom ___ physica ...
... ___ substance that is a mixture of two or more metals ___ columns of the periodic table; also known as groups ___ number of protons carried by the nucleus of an atom ___ element with an imbalance in the number of neutrons and protons ___ uncharged particle found in the nucleus of an atom ___ physica ...
Isotopes
... Soddy won the Nobel Prize in Chemistry in 1921 for his work with isotopes and radioactive materials. ...
... Soddy won the Nobel Prize in Chemistry in 1921 for his work with isotopes and radioactive materials. ...
atomic structure - IGCSE STUDY BANK
... The electrons are arranged in energy levels or shells around the nucleus and with increasing distance from the nucleus. Each electron in an atom is in a particular energy level (or shell) and the electrons must occupy the lowest available energy level (or shell) available nearest the nucleus. When t ...
... The electrons are arranged in energy levels or shells around the nucleus and with increasing distance from the nucleus. Each electron in an atom is in a particular energy level (or shell) and the electrons must occupy the lowest available energy level (or shell) available nearest the nucleus. When t ...
Lecture 2
... protons in an atom (as in a nuclear reaction) changes the element. • While atoms of the same element must have the same atomic number, they may have different mass numbers. If so, they are referred to as isotopes. Most elements have more than one naturally occurring isotope: ...
... protons in an atom (as in a nuclear reaction) changes the element. • While atoms of the same element must have the same atomic number, they may have different mass numbers. If so, they are referred to as isotopes. Most elements have more than one naturally occurring isotope: ...
Test Review: Unit 1 - Ms. Hill`s Pre
... a. Fusion: The combination of smaller molecule into larger ones. This happens on the sun. b. Fission: The splitting of large molecules into smaller radioactive daughter isotopes (“Mean Girls”) we do this in nuclear reactor and bombs! c. The big picture….both nuclear reaction result in the release of ...
... a. Fusion: The combination of smaller molecule into larger ones. This happens on the sun. b. Fission: The splitting of large molecules into smaller radioactive daughter isotopes (“Mean Girls”) we do this in nuclear reactor and bombs! c. The big picture….both nuclear reaction result in the release of ...
Periodic Table
... • When writing isotopes, the atomic number (or number of protons) will appear at the __________ • The mass number (number of protons plus neutrons will appear at the __________ • The element symbol will appear to the __________ • The different number of neutrons has NO bearing on chemical reactivity ...
... • When writing isotopes, the atomic number (or number of protons) will appear at the __________ • The mass number (number of protons plus neutrons will appear at the __________ • The element symbol will appear to the __________ • The different number of neutrons has NO bearing on chemical reactivity ...
SS18A - Atoms, Isotopes and Ions
... In addition to the atomic number, every atom can also be described by its mass number. The mass number is equal to the number of protons and neutrons in the nucleus of an atom. Recall that atoms of the same element have the same number of protons. Atoms of the same element can have different numbers ...
... In addition to the atomic number, every atom can also be described by its mass number. The mass number is equal to the number of protons and neutrons in the nucleus of an atom. Recall that atoms of the same element have the same number of protons. Atoms of the same element can have different numbers ...
Homework 1B1 - 3 - Uddingston Grammar School
... 7. Sodium has 10 electrons. (a) Complete the diagram to show how the electrons are arranged. You may wish to use the data booklet to help you. ...
... 7. Sodium has 10 electrons. (a) Complete the diagram to show how the electrons are arranged. You may wish to use the data booklet to help you. ...
14_1_atoms and isotopes FPS3
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
Structure of the Atom
... • The mass number is the total number of protons and neutrons in the nucleus. • Atoms can have different numbers of neutrons. • Atoms that have different numbers of neutrons are called isotopes. ...
... • The mass number is the total number of protons and neutrons in the nucleus. • Atoms can have different numbers of neutrons. • Atoms that have different numbers of neutrons are called isotopes. ...
12.1 Atoms and Isotopes
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
The Atom - Riverside City College
... • Heteronuclear Diatomic Molecule: a molecule made of two atoms that are different elements – NO ...
... • Heteronuclear Diatomic Molecule: a molecule made of two atoms that are different elements – NO ...
Element Blocks Project
... For this activity each person will be assigned an element to research. Your assignment is to produce an element block that will have six sides, each having different information about your element. Elements will be assigned randomly. Your teacher will show you how to make the block after you have re ...
... For this activity each person will be assigned an element to research. Your assignment is to produce an element block that will have six sides, each having different information about your element. Elements will be assigned randomly. Your teacher will show you how to make the block after you have re ...
Lawrencium

Lawrencium is a synthetic chemical element with chemical symbol Lr (formerly Lw) and atomic number 103. It is named in honor of Ernest Lawrence, inventor of the cyclotron, a device that was used to discover many artificial radioactive elements. A radioactive metal, lawrencium is the eleventh transuranic element and is also the final member of the actinide series. Like all elements with atomic number over 100, lawrencium can only be produced in particle accelerators by bombarding lighter elements with charged particles. Twelve isotopes of lawrencium are currently known; the most stable is 266Lr with a half-life of 11 hours, but the shorter-lived 260Lr (half-life 2.7 minutes) is most commonly used in chemistry because it can be produced on a larger scale.Chemistry experiments have confirmed that lawrencium indeed behaves as a heavier homolog to lutetium in the periodic table, and is a trivalent element. It thus could also be classified as the first of the 7th-period transition metals: however, its electron configuration is anomalous for its position in the periodic table, having an s2p configuration instead of the s2d configuration of its homolog lutetium. This means that lawrencium may be less volatile than expected for its position in the periodic table and have a volatility comparable to that of lead.In the 1950s, 1960s, and 1970s, many claims of the synthesis of lawrencium of varying quality were made from laboratories in the Soviet Union and the United States. The priority of the discovery and therefore the naming of the element was disputed between Soviet and American scientists, and while the International Union of Pure and Applied Chemistry (IUPAC) established lawrencium as the official name for the element and gave the American team credit for the discovery, this was reevaluated in 1997, giving both teams shared credit for the discovery but not changing the element's name.