Document
... In 1916, Gilbert Lewis used this fact to explain why atoms form certain kinds of ions and molecules The Octet Rule: in forming compounds, atoms tend to achieve a noble gas configuration; 8 in the outer level is stable ...
... In 1916, Gilbert Lewis used this fact to explain why atoms form certain kinds of ions and molecules The Octet Rule: in forming compounds, atoms tend to achieve a noble gas configuration; 8 in the outer level is stable ...
Activities 2
... E) Covalent bonds involve the transfer of electrons between atoms; ionic bonds involve the sharing of electrons between atoms. 4) Which bond or interaction between atoms would be most difficult to disrupt when the interacting atoms are put into water and heated slightly? A) covalent bond B) hydrogen ...
... E) Covalent bonds involve the transfer of electrons between atoms; ionic bonds involve the sharing of electrons between atoms. 4) Which bond or interaction between atoms would be most difficult to disrupt when the interacting atoms are put into water and heated slightly? A) covalent bond B) hydrogen ...
Studies Regarding the Nickel Electrodeposition from
... appearance of Zn coatings from ionic liquids, several brighteners have been investigated, such as acetonitrile, ethylene diamine and ammonia (Abbott et al. 2010). Ammonia was initially chosen as previous studies have determined that Zn is effectively complexed by this ligand in aqueous sulphate elec ...
... appearance of Zn coatings from ionic liquids, several brighteners have been investigated, such as acetonitrile, ethylene diamine and ammonia (Abbott et al. 2010). Ammonia was initially chosen as previous studies have determined that Zn is effectively complexed by this ligand in aqueous sulphate elec ...
Chapter 1
... B. Why do salts (NaCl, say) easily dissolve in water? C. Discuss: identify as many properties of water (H2O) as possible D. Why does pure water have a pH = 7? E. Explain how buffers can stabilize the pH of a ...
... B. Why do salts (NaCl, say) easily dissolve in water? C. Discuss: identify as many properties of water (H2O) as possible D. Why does pure water have a pH = 7? E. Explain how buffers can stabilize the pH of a ...
Name_______________________ Answers to Final Exam Study
... 4. Which of these steps in the digestive process is a physical change? A) Saliva changing carbohydrates to sugars B) The hormone insulin metabolizing C) Enzymes breading down proteins to amino acids D) Liquids being absorbed in the large intestine 5. Bones are made up of many minerals, one of which ...
... 4. Which of these steps in the digestive process is a physical change? A) Saliva changing carbohydrates to sugars B) The hormone insulin metabolizing C) Enzymes breading down proteins to amino acids D) Liquids being absorbed in the large intestine 5. Bones are made up of many minerals, one of which ...
Solubility Main article: Solvation The ability of one compound to
... interactions are unfavorable, then the free energy decreases with increasing solute concentration. At some point the energy loss outweighs the entropy gain, and no more solute particles can be dissolved; the solution is said to be saturated. However, the point at which a solution can become saturate ...
... interactions are unfavorable, then the free energy decreases with increasing solute concentration. At some point the energy loss outweighs the entropy gain, and no more solute particles can be dissolved; the solution is said to be saturated. However, the point at which a solution can become saturate ...
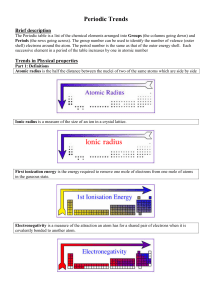
Topic 3 Periodicity notes SL - Chemical Minds
... attraction between nucleus and electrons in the outer shell so therefore less energy is required to remove an electron from the outer shell (ionization energy). The melting points (of metals) decrease because the increase in radius of the positive metallic ion results in a weaker attraction between ...
... attraction between nucleus and electrons in the outer shell so therefore less energy is required to remove an electron from the outer shell (ionization energy). The melting points (of metals) decrease because the increase in radius of the positive metallic ion results in a weaker attraction between ...
Word
... b. Water boils above 100 0C at higher pressures c. Water boils below 100 0C at lower pressures C. Condensation 1. The conversion of a gas to a liquid by the removal of energy IV. Freezing and Melting A. Freezing Point 1. The temperature at which the solid and liquid are in equilibrium at 1 atm 2. Fo ...
... b. Water boils above 100 0C at higher pressures c. Water boils below 100 0C at lower pressures C. Condensation 1. The conversion of a gas to a liquid by the removal of energy IV. Freezing and Melting A. Freezing Point 1. The temperature at which the solid and liquid are in equilibrium at 1 atm 2. Fo ...
Solution - ZOMUedu
... ○ Stock solution = a solution of known concentration that is used to make more dilute solutions Precipitation ○ Precipitation = when aqueous solutions of ionic compounds are poured together and a solid forms (solid = precipitate) ○ We can predict the products ■ The anion and cation switch partners ■ ...
... ○ Stock solution = a solution of known concentration that is used to make more dilute solutions Precipitation ○ Precipitation = when aqueous solutions of ionic compounds are poured together and a solid forms (solid = precipitate) ○ We can predict the products ■ The anion and cation switch partners ■ ...
Name:_____________ Chemistry 114 Second Hour Exam
... HF will form intermolecular hydrogen bonds, HCl won’t; this will give HF stronger intermolecular interactions and give it a higher boiling point. I2 has a lower vapor pressure than Cl2 at room temperature. I2 is larger that Cl2, this will make its London force larger to give it stronger intermolecul ...
... HF will form intermolecular hydrogen bonds, HCl won’t; this will give HF stronger intermolecular interactions and give it a higher boiling point. I2 has a lower vapor pressure than Cl2 at room temperature. I2 is larger that Cl2, this will make its London force larger to give it stronger intermolecul ...
Acids and bases
... 9.12 Ionic liquids (also called molten or fused salts ) A eutectic is a mixture of two substances and is characterized by a sharp melting point lower than that of either of the components; a eutectic behaves as though it were a single substance. For example, the melting point of NaCl is 1073 K, but ...
... 9.12 Ionic liquids (also called molten or fused salts ) A eutectic is a mixture of two substances and is characterized by a sharp melting point lower than that of either of the components; a eutectic behaves as though it were a single substance. For example, the melting point of NaCl is 1073 K, but ...
Chemical Bonds ch6 p.161
... +ion and -ion break apart in water so will conduct electricity in water. Some do not dissolve because the pull between the charges are greater than the attraction of H2O molecule ...
... +ion and -ion break apart in water so will conduct electricity in water. Some do not dissolve because the pull between the charges are greater than the attraction of H2O molecule ...
Solute - St John Brebeuf
... because it breaks into ________ A substance made up of a NONMETAL and a NONMETAL is COVALENT, and will NOT form a conducting solution in water because it stays as a molecular compound. ...
... because it breaks into ________ A substance made up of a NONMETAL and a NONMETAL is COVALENT, and will NOT form a conducting solution in water because it stays as a molecular compound. ...
Chem 321 Lecture 11 - Chemical Activities
... shielded). Consequently, the formal charge an ion projects to other ions is less than it normally would be, so it interacts with oppositely-charged ions less attractively - its effective concentration is lower. The activity coefficient is a measure of how effectively an ion can interact in solution. ...
... shielded). Consequently, the formal charge an ion projects to other ions is less than it normally would be, so it interacts with oppositely-charged ions less attractively - its effective concentration is lower. The activity coefficient is a measure of how effectively an ion can interact in solution. ...
Chemical Bonding Notes for 2016
... Ionic Bonding • Occurs between a metal and a non-metal. • Electrons are moved from one atom to another. • The charged particle that results is called an ...
... Ionic Bonding • Occurs between a metal and a non-metal. • Electrons are moved from one atom to another. • The charged particle that results is called an ...
Writing Ionic Formulas continued
... 13. Many ionic compounds that contain calcium have been in use for so long that they are often identified by their mineral or common names, which were given to them before their chemical composition was determined. Write the ionic formulas for the following calcium compounds, whose common or mineral ...
... 13. Many ionic compounds that contain calcium have been in use for so long that they are often identified by their mineral or common names, which were given to them before their chemical composition was determined. Write the ionic formulas for the following calcium compounds, whose common or mineral ...
ln2_storage_pre
... On the other hand, the gas cylinders are probably at room temperature. This is way above the critical temperature for both fluids, so you will not get a liquid no matter how much pressure you put on it. The gases in the cylinders are supercritical fluids, though when you get that far above the criti ...
... On the other hand, the gas cylinders are probably at room temperature. This is way above the critical temperature for both fluids, so you will not get a liquid no matter how much pressure you put on it. The gases in the cylinders are supercritical fluids, though when you get that far above the criti ...
Study Guide for Exam 2_old
... Study Guide for Exam 2 You should be able to answer the following questions, solve problems involving the following concepts, or understand the following concepts so that you can describe them and answer questions about them. Periodic trends regarding atomic and ionic radii. What is meant by valence ...
... Study Guide for Exam 2 You should be able to answer the following questions, solve problems involving the following concepts, or understand the following concepts so that you can describe them and answer questions about them. Periodic trends regarding atomic and ionic radii. What is meant by valence ...
Word - chemmybear.com
... Atoms tend to lose, gain, or ___________ electrons to complete their valence shells. When a chlorine atom gains an electron, it fills its valence shell forming a negative chloride________. Whenever ionic solids are formed, __________ is involved. An ionic material is composed of positive ions bonded ...
... Atoms tend to lose, gain, or ___________ electrons to complete their valence shells. When a chlorine atom gains an electron, it fills its valence shell forming a negative chloride________. Whenever ionic solids are formed, __________ is involved. An ionic material is composed of positive ions bonded ...
Metathesis Problems (and Some Solutions) Identified Through
... • Miscible fluids: fluids that dissolve with each other in all proportions • All nonreactive gases are generally miscible – Air is a gaseous solution (N2, O2, CO2, etc.) • Liquid solutions: dissolving a solid, liquid, or gas into a liquid – Ethanol and water are miscible, and when mixed make a liqui ...
... • Miscible fluids: fluids that dissolve with each other in all proportions • All nonreactive gases are generally miscible – Air is a gaseous solution (N2, O2, CO2, etc.) • Liquid solutions: dissolving a solid, liquid, or gas into a liquid – Ethanol and water are miscible, and when mixed make a liqui ...
Spatial Structure of Electrical Diffuse Layers in Highly Concentrated
... fundamentally different from traditional (dilute) electrolytes and demonstrate the limited mechanistic understanding of highly concentrated electrolytes. Moreover, electrolyte behavior placed in close proximity of two charged surfaces becomes even more unclear due to the possible overlap between diffu ...
... fundamentally different from traditional (dilute) electrolytes and demonstrate the limited mechanistic understanding of highly concentrated electrolytes. Moreover, electrolyte behavior placed in close proximity of two charged surfaces becomes even more unclear due to the possible overlap between diffu ...
CHEM1100 Practice Exam 2 You have 120 minutes to complete this
... rule are rare. Salts containing the ammonium ion (NH4+) are also soluble. 2. Salts containing nitrate ion (NO3-) are generally soluble. 3. Salts containing Cl–, Br–, I– are generally soluble. Important exceptions to this rule are halide salts of Ag+, Pb2+, and (Hg2)2+. 4. Most silver salts are insol ...
... rule are rare. Salts containing the ammonium ion (NH4+) are also soluble. 2. Salts containing nitrate ion (NO3-) are generally soluble. 3. Salts containing Cl–, Br–, I– are generally soluble. Important exceptions to this rule are halide salts of Ag+, Pb2+, and (Hg2)2+. 4. Most silver salts are insol ...
Improvement of the Sorption Performance of Nanosilica
... better charge distribution and larger ion size compared to classical inorganic salts result in melting points below 100 °C. By variation of anions and cations, solubility, reactivity, and coordination properties of the ILs can be changed according to the special requirements of the given application ...
... better charge distribution and larger ion size compared to classical inorganic salts result in melting points below 100 °C. By variation of anions and cations, solubility, reactivity, and coordination properties of the ILs can be changed according to the special requirements of the given application ...
Ionic liquid
An ionic liquid (IL) is a salt in the liquid state. In some contexts, the term has been restricted to salts whose melting point is below some arbitrary temperature, such as 100 °C (212 °F). While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions and short-lived ion pairs. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid salts, or ionic glasses. Ionic liquids have many applications, such as powerful solvents and electrically conducting fluids (electrolytes). Salts that are liquid at near-ambient temperature are important for electric battery applications, and have been used as sealants due to their very low vapor pressure.Any salt that melts without decomposing or vaporizing usually yields an ionic liquid. Sodium chloride (NaCl), for example, melts at 801 °C (1,474 °F) into a liquid that consists largely of sodium cations (Na+) and chloride anions (Cl−). Conversely, when an ionic liquid is cooled, it often forms an ionic solid—which may be either crystalline or glassy.The ionic bond is usually stronger than the Van der Waals forces between the molecules of ordinary liquids. For that reason, common salts tend to melt at higher temperatures than other solid molecules. Some salts are liquid at or below room temperature. Examples include compounds based on the 1-Ethyl-3-methylimidazolium (EMIM) cation and include: EMIM:Cl, EMIM dicyanamide, (C2H5)(CH3)C3H3N+2·N(CN)−2, that melts at −21 °C (−6 °F); and 1-butyl-3,5-dimethylpyridinium bromide which becomes a glass below −24 °C (−11 °F).Low-temperature ionic liquids can be compared to ionic solutions, liquids that contain both ions and neutral molecules, and in particular to the so-called deep eutectic solvents, mixtures of ionic and non-ionic solid substances which have much lower melting points than the pure compounds. Certain mixtures of nitrate salts can have melting points below 100 °C.The term ""ionic liquid"" in the general sense was used as early as 1943.